Catalysts
Dr. Davinder Bhachu
Teacher

What Is A Catalyst?
A catalyst is a substance that speeds up a reaction without being chemically used up.
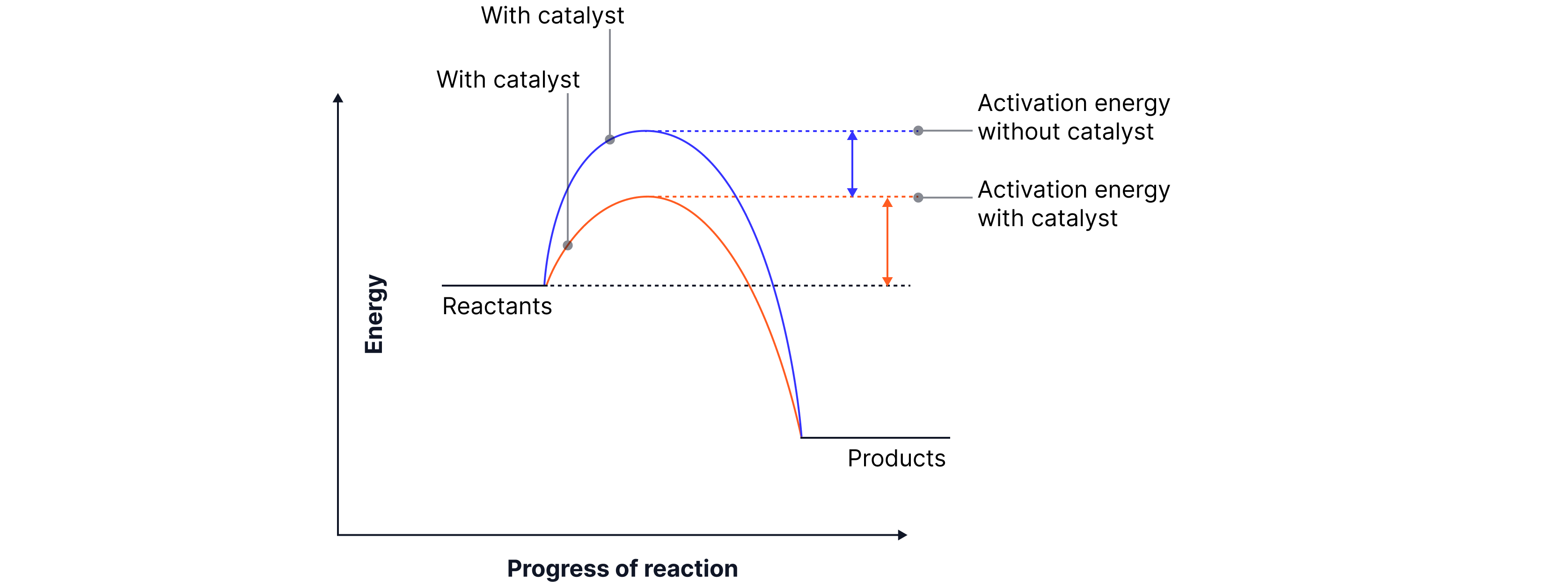
How they work
Provide a different reaction pathway,
One that requires a lower activation energy.
Lower activation energy means more likely for collisions to be successful, therefore faster reaction rate
Cyclist Analogy for Activation Energy and Catalysts
Imagine a cyclist travelling from one side of a hill to the other:
Without a Catalyst - The Steep Hill
The cyclist must pedal up and over a tall, steep hill.
This hill represents the activation energy - the minimum amount of energy needed for the reaction to start.
If the cyclist doesn’t push hard enough, they won’t reach the top and will roll back down.
In the same way, particles that collide without enough energy don’t react and simply bounce apart.
With a Catalyst - The Tunnel
Now imagine there’s a tunnel dug straight through the hill.
The tunnel offers a lower, easier route that requires much less effort from the cyclist.
This tunnel represents the alternative reaction pathway created by a catalyst.
The journey becomes quicker and requires less energy, even though the cyclist still ends up on the same side.
Why the Analogy Works
Hill = activation energy barrier
Tunnel = catalyst pathway
Cyclist = reacting particles
Getting to the other side = products forming
The height of the hill doesn’t change - but the route taken does.
Biological Catalysts: Enzymes
Key points
Enzymes are protein catalysts found in living organisms.
They are highly specific, usually acting on only one type of molecule.
Examples include:
Enzymes in the stomach breaking down proteins,
Enzymes in yeast converting sugars to carbon dioxide during fermentation.
Check for Understanding
Recall
a) What is a catalyst?
Answer:
Substance that speeds up a reaction; not chemically used up
b) What effect does a catalyst have on activation energy?
Answer:
Lowers activation energy
c) What type of biological molecule is an enzyme?
Answer:
Protein
Application
d) Explain how a catalyst increases the rate of a reaction using collision theory.
Answer:
Provides alternative pathway; lower activation energy; more collisions have sufficient energy; more successful collisions per second
e) Use the cyclist analogy to explain how a catalyst works.
Answer:
Hill represents activation energy; tunnel represents alternative pathway; easier/lower energy route; products still formed
f) Explain why enzymes are described as highly specific.
Answer:
Each enzyme has a specific active site shape; only one substrate fits; forms enzyme–substrate complex
Challenge (HT)
g) Explain why a catalyst increases reaction rate without changing the overall energy change (ΔH) of the reaction.
Answer:
Catalyst lowers activation energy only; reactant and product energy levels remain unchanged; overall energy difference (ΔH) stays the same
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!