Cracking and alkenes
Lajoy Tucker
Teacher

Contents
Cracking and alkenes
Cracking is a chemical process used to break down large hydrocarbon molecules (from crude oil) into smaller, more useful ones.
Large hydrocarbons are often thick, viscous, and not very useful as fuels, whereas smaller hydrocarbons are more volatile and burn more cleanly.
Cracking helps meet the high demand for small-chain hydrocarbons, such as petrol (gasoline) and alkenes used to make plastics and other chemicals.
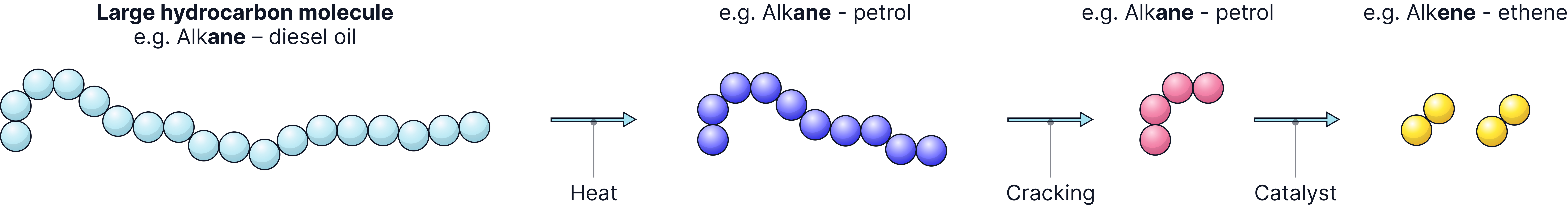
Why Cracking Is Important
The demand for short-chain hydrocarbons (like petrol) is greater than the supply directly available from crude oil.
Cracking converts long-chain alkanes (from heavy fractions) into shorter-chain alkanes and alkenes, which have more commercial value.
Examples of uses:
Short-chain alkanes: fuels such as petrol, LPG, and jet fuel.
Alkenes: used as starting materials to make polymers, alcohols, and solvents.
The Cracking Process
There are two main methods of cracking hydrocarbons:
Type of Cracking | Conditions Used | Description |
|---|---|---|
Catalytic cracking | High temperature and a catalyst (often aluminium oxide or zeolite) | Hydrocarbon vapours are passed over a hot catalyst, breaking large molecules into smaller ones. |
Steam cracking | Very high temperature and steam | Hydrocarbon vapours are mixed with steam and heated strongly, breaking them into smaller molecules including alkenes. |
Products of Cracking
Cracking produces a mixture of alkanes and alkenes.
Alkenes are unsaturated hydrocarbons with at least one C=C double bond.
General formula for alkenes:
Example:
Decane Octane Ethene
Here, octane is an alkane (fuel) and ethene is an alkene (used in polymers).
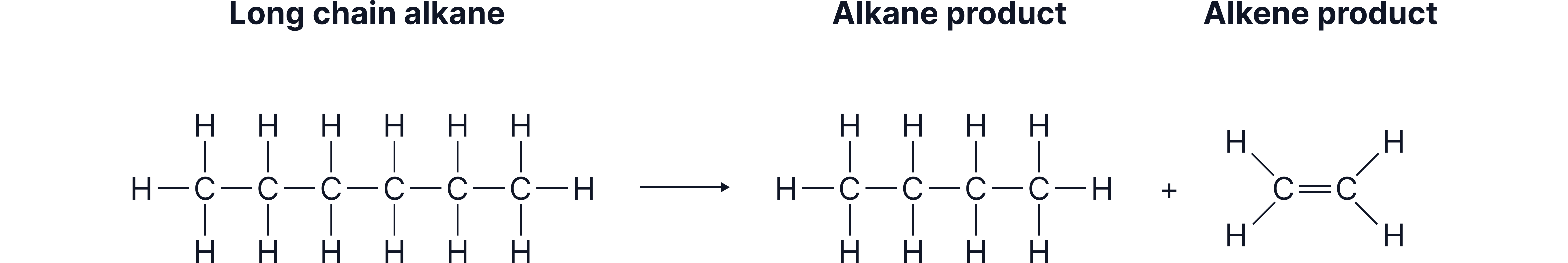
Cracking Equations
Cracking equations can be easily balanced by considering the conservation of mass. The number of atoms of each element must be the same on both sides of the equation.
Example:
_______
There are 10 C atoms and 22 H atoms on the left hand side.
There are only 8 C atoms and 18 H atoms on the right hand side.
2 C atoms and 4 H atoms are required to balance this equation.
The missing species is
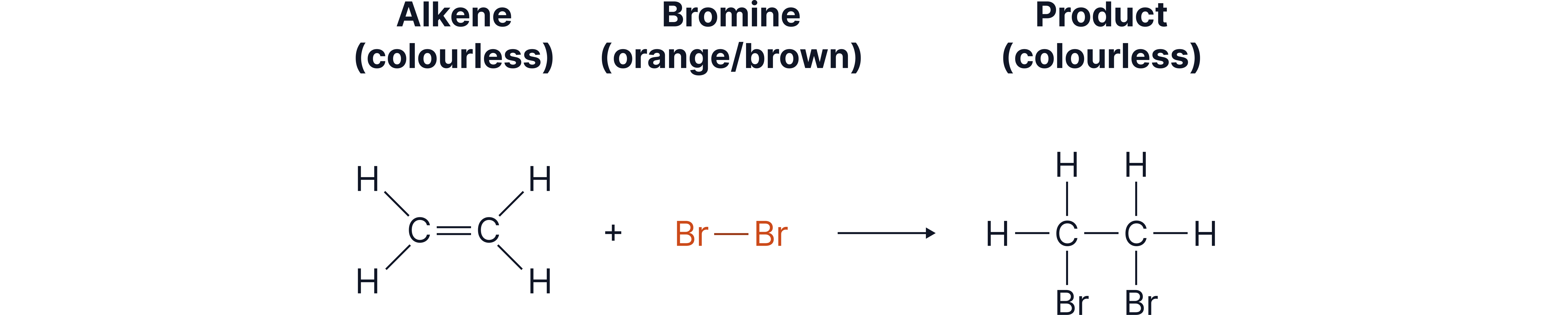
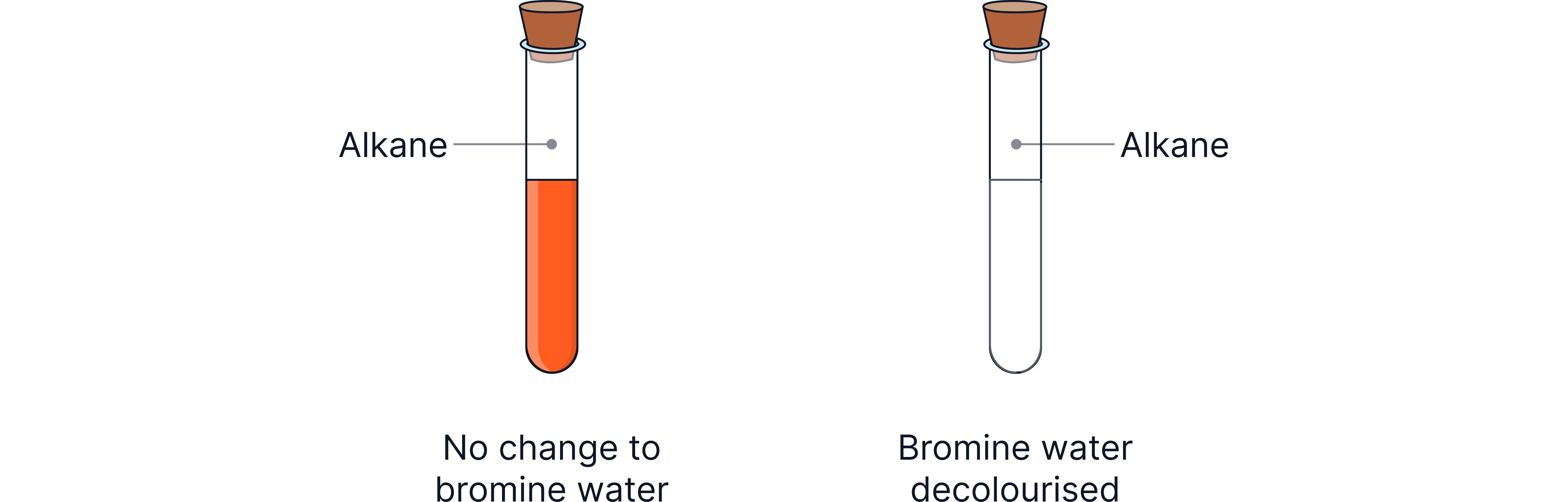
Testing for Alkenes
Alkenes are more reactive than alkanes due to the C=C double bond.
They react with bromine water, which is used as a chemical test.
Substance Tested | Observation with Bromine Water | Explanation |
|---|---|---|
Alkane | No change (bromine water stays orange/brown) | Alkanes are saturated and do not react. |
Alkene | Colour changes from orange/brown → colourless | Alkenes add bromine across the double bond, forming a dibromo compound. |
Summary
Cracking breaks large hydrocarbons into smaller alkanes and alkenes.
Catalytic cracking uses a catalyst and heat (~500°C).
Steam cracking uses steam at very high temperature (~800°C).
Alkenes are formed, tested using bromine water (orange → colourless).
Short-chain hydrocarbons are used as fuels, and alkenes are used to make polymers and other chemicals.
Check for Understanding
Recall
a) What is cracking?
Answer
Breaking long-chain hydrocarbons into shorter, more useful alkanes and an alkene is also formed.
b) State the general formula for alkenes.
Answer
CₙH₂ₙ
c) Why is cracking economically important?
Answer
Produces fuels and alkenes in higher demand for industry.
Application
d) Describe the test for an alkene.
Answer
Bromine water; changes from orange to colourless.
e) Compare steam cracking and catalytic cracking.
Answer
Steam: high temperatures and uses steam; Catalytic: lower temperatures required; catalyst (aluminiun oxide/silicon dioxide) used.
Challenge
f) C₁₄H₃₀ cracks to form C₉H₂₀ and one alkene. Deduce the alkene formula.
Answer
C₅H₁₀