Electrolysis of molten ionic compounds
Lajoy Tucker
Teacher

Contents
What Is Electrolysis?
Definition:
Electrolysis is the splitting (decomposition) of an ionic compound using electricity to form its elements.
An electric current passes through an electrolyte - a molten or aqueous ionic compound - causing ions to move and undergo redox reactions at the electrodes.
What Are Electrolytes?
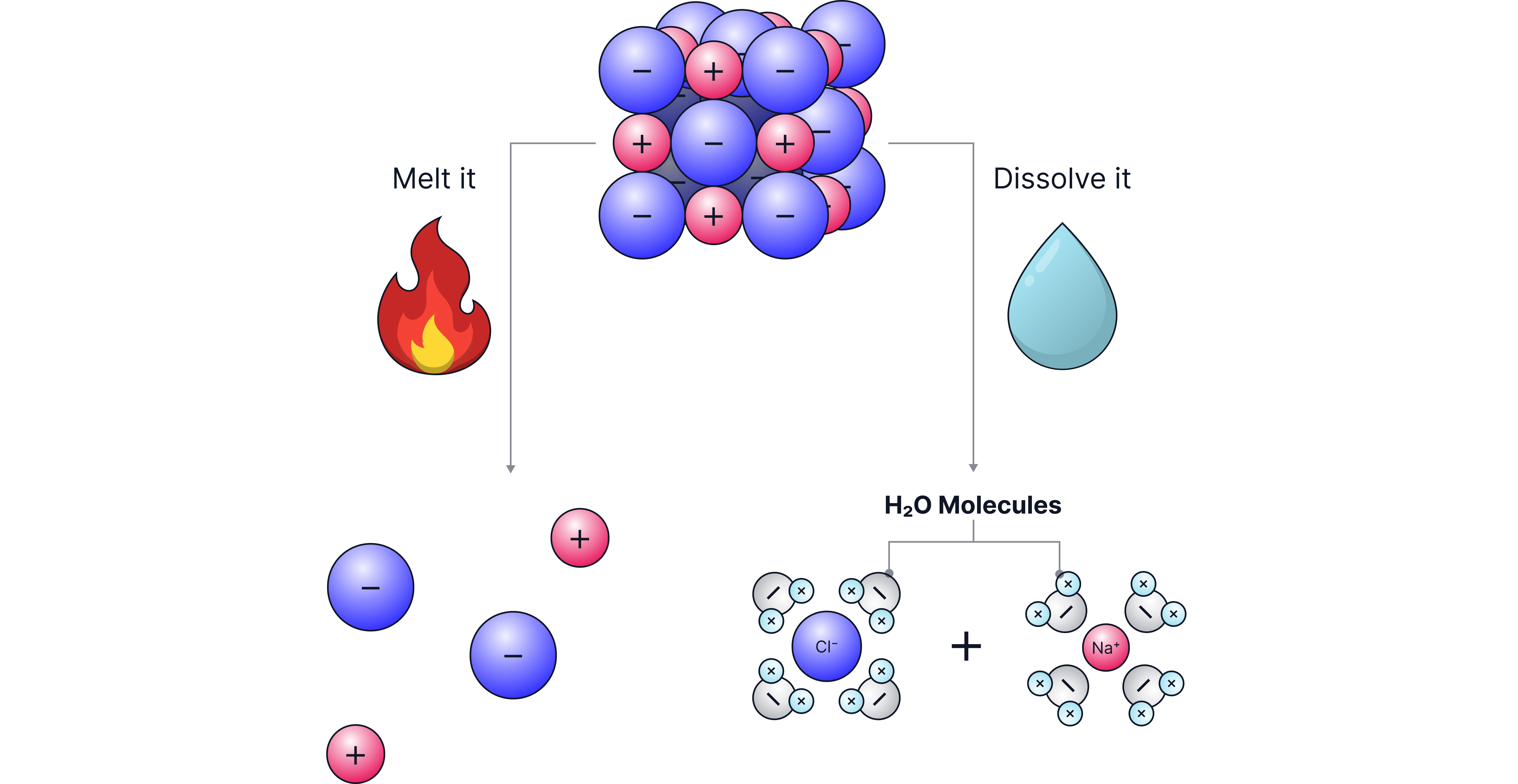
Electrolyte: An ionic compound that can conduct electricity when molten or in solution.
In solids, ions are fixed in a lattice and cannot move, so no current flows.
When molten or dissolved, the ions are free to move and carry charge through the structure.
State
Conducts electricity?
Reason
Solid ionic compound
No
Ions fixed in lattice
Molten ionic compound
Yes
Ions free to move
Aqueous solution
Yes
Ions free to move in water
Basic Electrolysis Setup
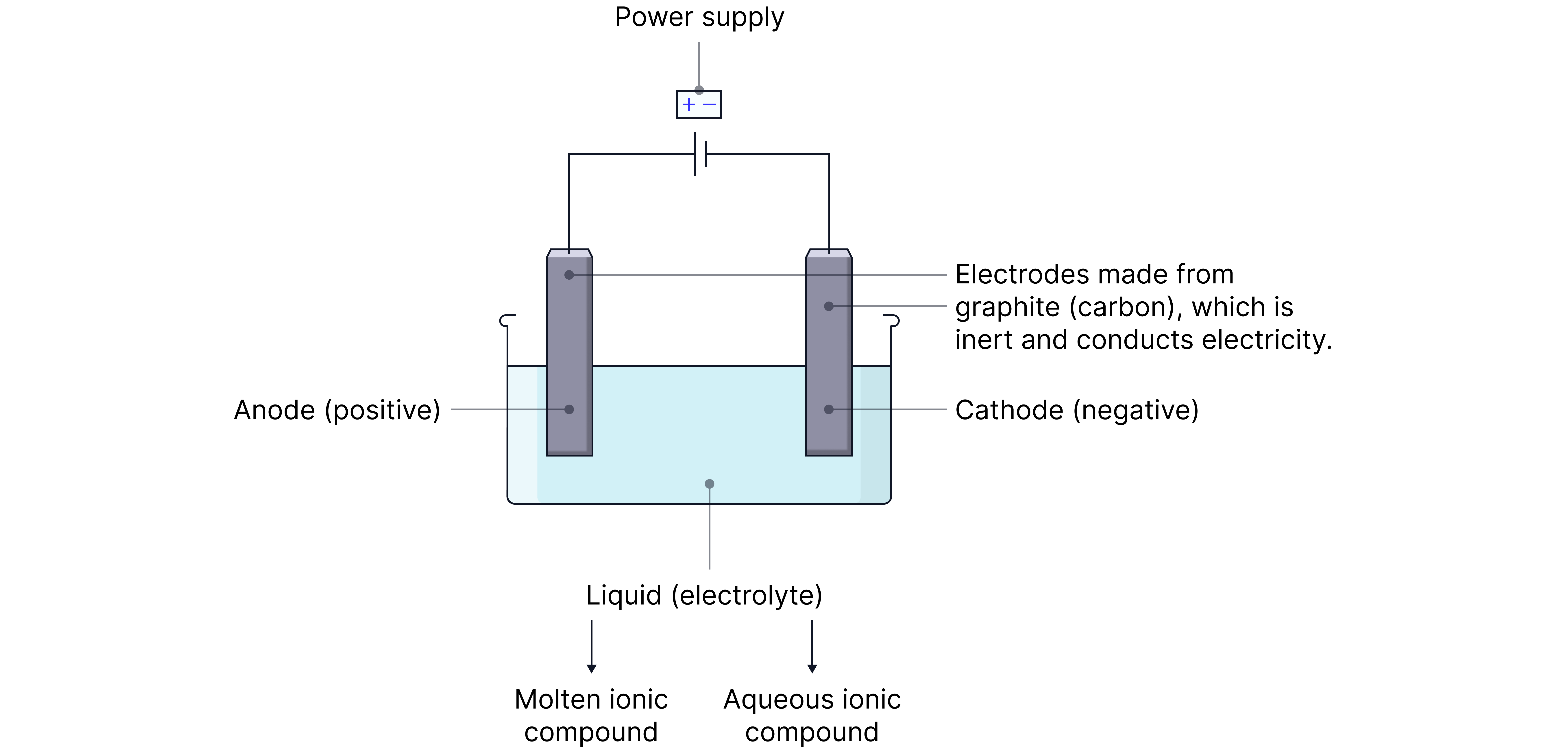
Labels Explained:
Electrolyte: molten or aqueous ionic compound containing free ions.
Electrodes: made of graphite (inert, conducts electricity).
Anode : Positive electrode attracts anions (negative ions).
Cathode : Negative electrode attracts cations (positive ions).
Power supply: drives the movement of electrons through the circuit.
PANIC is used to remember the charges on the ELECTRODES
P - Positive
A - Anode
N - Negative
I – Is
C - Cathode
Ionic Movement and Reactions (HT – Only)
Ions are attracted to the oppositely charged electrode because opposites attract!
Electrode | Charge | Ion Type | Reaction Type | Example |
Cathode | Negative | Cations | Reduction (gain ) | |
Anode | Positive | Anions | Oxidation (lose ) |
OIL RIG:
Oxidation Is Loss (of electrons)
Reduction Is Gain (of electrons)
The Reactivity Series and Extraction of Metals
Metals are extracted from their ores depending on their position in the reactivity series.
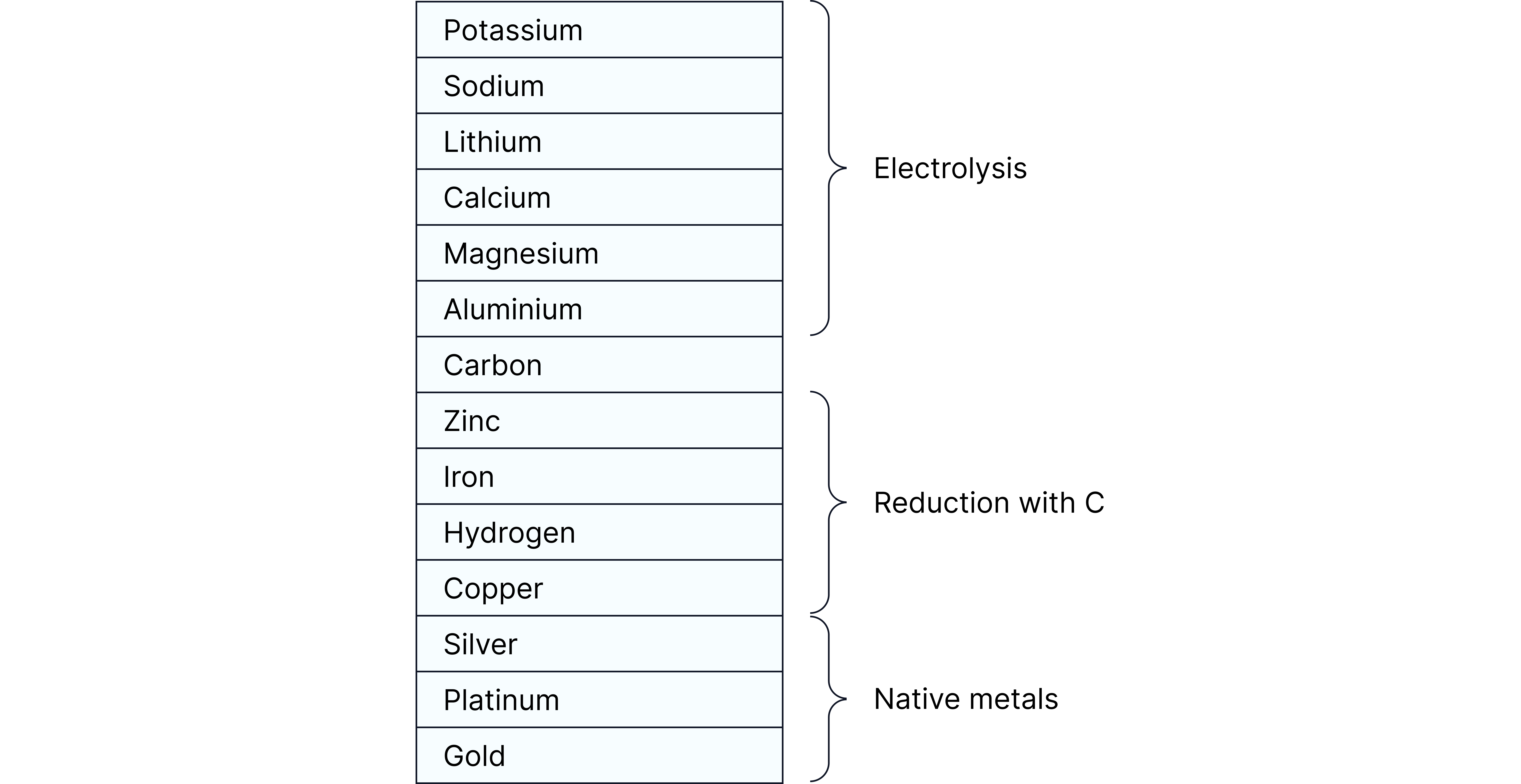
Key idea:
Metals above carbon in the reactivity series (e.g. K, Na, Ca, Mg, Al) must be extracted by electrolysis.
Metals below carbon (e.g. Zn, Fe, Cu) are extracted by chemical reduction using carbon.
Why Electrolysis Is Used for Reactive Metals
Highly reactive metals form stable compounds that carbon cannot reduce.
Electricity is used to decompose molten compounds into elements.
This requires a lot of energy, making electrolysis expensive.
Example:
Example 1: Electrolysis of Molten Lead Bromide ()
At the Cathode ():
Lead ions gain electrons to become pure lead
HT Only:
At the Anode ():
Bromide ions lose electrons to become gaseous bromine.
HT Only:
Products: Lead (metal) and Bromine (gas)
Important Example: Electrolysis of Aluminium Oxide (Al₂O₃)
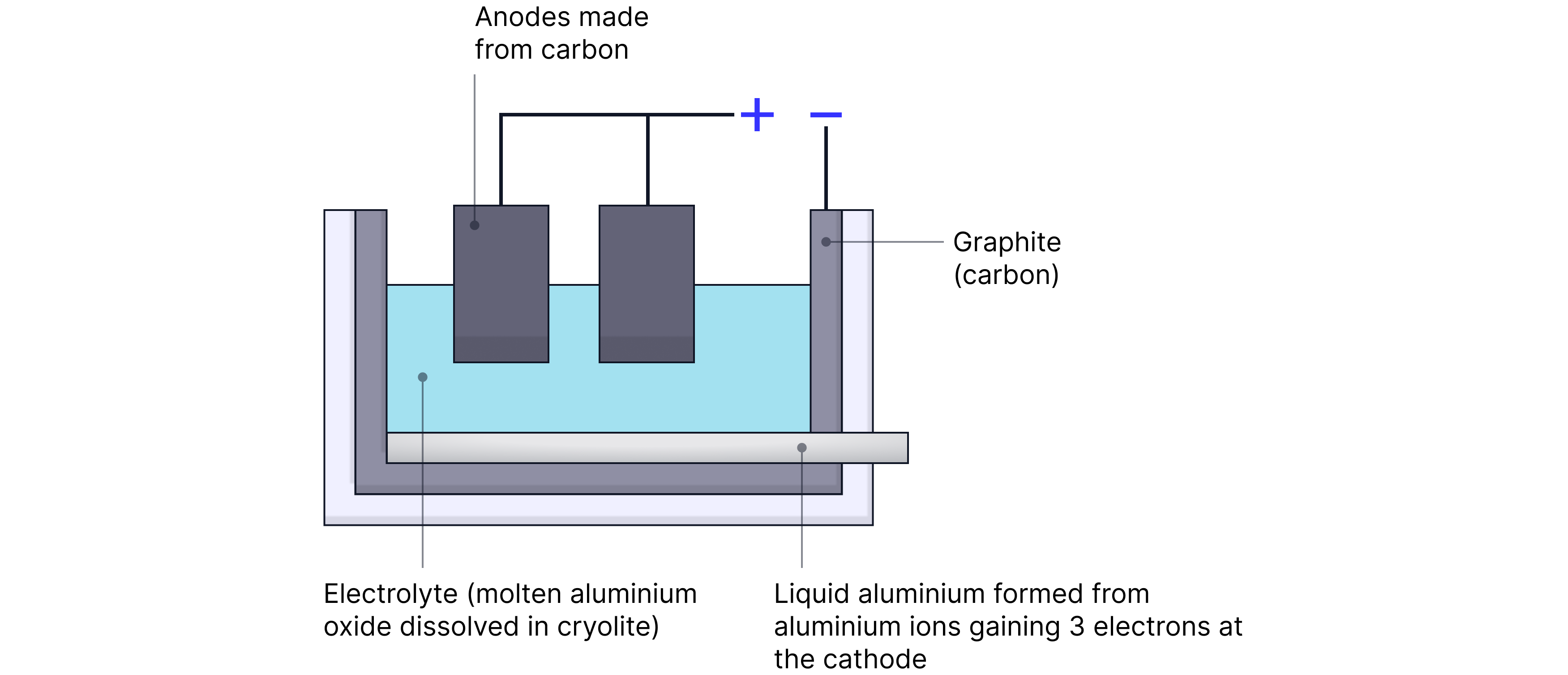
Step 1: Melting and Dissolving
Aluminium oxide is dissolved in molten cryolite to lower its melting point, reducing energy cost.
Step 2: Electrolysis Reactions
At the Cathode (-):
Aluminum ions gain electrons to become pure aluminium
At the Anode (+):
Oxude ions lose electrons to become pure oxygen
Products: Aluminium metal and Oxygen gas
Step 3: Replacement of Anode
The carbon anode reacts with oxygen to form , so it must be replaced frequently.
Summary Table — Extraction of Metals
Metal | Position | Extraction Method | Reason |
Potassium, Sodium, Calcium, Magnesium, Aluminium | Above Carbon | Electrolysis | Too reactive to reduce with carbon |
Zinc, Iron, Tin, Lead | Below Carbon | Reduction with Carbon | Less reactive |
Copper | Below Hydrogen | Reduction or found native | Forms less stable compounds |
Silver, Gold, Platinum | Below Hydrogen | Native metals | Very unreactive |
Quick Recap
Term | Meaning |
Electrolysis | Splitting of a compound using electricity |
Electrolyte | Molten or aqueous ionic compound |
Anode | Positive electrode (oxidation occurs) |
Cathode | Negative electrode (reduction occurs) |
Cryolite | Lowers melting point of to reduce costs |
Conductor used | Graphite (inert carbon) – needs replacing due to oxygen reacting with anode in the electrolysis of |
Key Ideas Summary
Concept | Foundation Tier | Higher Tier (HT Only) |
|---|---|---|
Electrolysis definition | Splitting compounds using electricity. | Writing and balancing half-equations. |
Electrolyte condition | Must be molten or dissolved. | Explain movement of ions and charges. |
At cathode | Cations gain electrons (reduction). | Write balanced half equations. |
At anode | Anions lose electrons (oxidation). | Link to OIL RIG and oxidation states. |
Metal extraction | Used for reactive metals (e.g. Al). | Explain cryolite’s role and formation. |
Practice Questions
Recall
a) What is electrolysis?
Answer
a) Electrolysis is the splitting of an ionic compound using electricity.
b) What are electrolytes made of?
Answer
b) Ions (charged particles).
c) Which ions move to the anode and which to the cathode?
Answer
c) Anions → Anode; Cations → Cathode.
d) Why must ionic compounds be molten or dissolved before electrolysis?
Answer
d) So ions can move freely and carry charge.
e) Name one reason why electrolysis is expensive.
Answer
e) It requires large amounts of energy to melt and supply current.
Apply (HT Only)
f) Write half equations for the electrolysis of molten NaCl.
Answer
f) .
g) Explain why aluminium cannot be extracted by carbon reduction.
Answer
g) Aluminium is too reactive - carbon cannot displace it.
h) State the role of cryolite in aluminium extraction.
Answer
h) Cryolite lowers melting point and saves energy.
i) Why must the anode be replaced regularly?
Answer
i) The carbon anode reacts with oxygen to form .
j) Predict the products formed during electrolysis of molten magnesium oxide.
Answer
j) and .