Extraction of metals
Emmanuel Opoku
Teacher

Contents
Why Do Metals Need to Be Extracted?
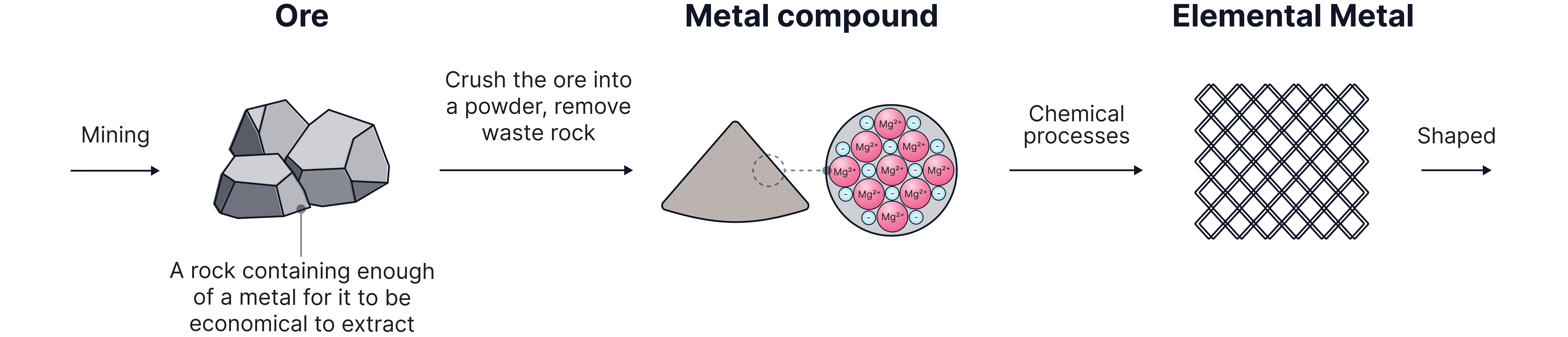
Most metals in the Earth’s crust are chemically combined with other elements, forming compounds such as oxides, sulfides, or carbonates. These compounds are found in rocks called ores.
Definition:
An ore is a rock that contains enough metal to make extraction worthwhile (economically viable).
Only a few unreactive metals, like gold, silver, and platinum, are found in their native (elemental) state because they do not react with oxygen or water.
Unreactive (Native) Metals
Some metals are so unreactive that they are found as the pure metal in the Earth – native metals.
Examples:
Gold (Au)
Silver (Ag)
Platinum (Pt)
These metals are found as metallic elements rather than as compounds because they do not corrode or oxidise easily.
No chemical extraction is required - they can be collected and refined directly.
Extracting Metals Below Carbon
Metals that are less reactive than carbon are extracted from their metal oxides by reduction with carbon.
Definition:
Reduction = loss of oxygen
Oxidation = gain of oxygen
During extraction:
The metal oxide is reduced (loses oxygen).
The carbon (or carbon monoxide) is oxidised (gains oxygen).
This type of reaction is called a redox reaction.
Worked Example: Extracting Iron from Iron(III) Oxide
Iron is extracted in a blast furnace using carbon monoxide.
Step-by-step explanation:
1. Carbon monoxide acts as a reducing agent - it removes oxygen from iron(III) oxide.
2. Iron(III) oxide is reduced to iron metal.
3. Carbon monoxide is oxidised to carbon dioxide.
In words:
Iron(III) oxide carbon monoxide iron carbon dioxide
Substance | Change | Explanation |
|---|---|---|
Iron(III) oxide | Reduced | Loses oxygen, forming iron |
Carbon monoxide | Oxidised | Gains oxygen, forming |
Remember:
Reduction = loss of oxygen
Oxidation = gain of oxygen
Alternative Reaction (Direct Reduction)
Carbon can also react directly with the metal oxide in some extractions:

In this reaction:
Carbon removes oxygen from the iron oxide.
Carbon is oxidised to carbon dioxide.
This process is cheaper than electrolysis and widely used for iron, zinc, and lead extraction.
Extracting Metals Above Carbon
Metals more reactive than carbon cannot be extracted by reduction.
They are extracted by electrolysis, which uses electricity to split molten or dissolved ionic compounds into elements (more of this in Electrolysis notes).
Summary
Metal Type | Example | Chemical Process to Extract | Reaction Type |
Very Unreactive | Gold, silver, Platinum | Found as native metal (no chemical process needed) | No reaction needed |
Less Reactive (Below Carbon) | Iron, Lead, Zinc | Reduction with carbon | Redox (carbon removes oxygen from ore) |
More reactive (Above Carbon) | Aluminium, Sodium | Electrolysis | Requires electricity |
Check Your Understanding
Recall
a) What is an ore?
Answer
a) A rock that contains enough metal to make extraction economical.
b) Define reduction and oxidation in terms of oxygen.
Answer
b) Reduction is the loss of oxygen; oxidation is the gain of oxygen.
c) Name one metal found uncombined in the Earth’s crust.
Answer
c) Gold (also acceptable: silver or platinum).
Apply
d) Write the word equation for the extraction of iron using carbon monoxide.
Answer
d) Iron(III) oxide + carbon monoxide → iron + carbon dioxide.
e) Explain why carbon can extract iron but not aluminium.
Answer
e) Iron is below carbon in the reactivity series, so carbon is reactive enough to remove oxygen; aluminium is above carbon, so it cannot.
Challenge
f) In the reaction :
– Which substance is oxidised?
– Which substance is reduced?
Explain your reasoning.
Answer
f) Carbon monoxide is oxidised (gains oxygen to form ), and iron oxide is reduced (loses oxygen to form iron).