Identification of common gases
Lajoy Tucker
Teacher

Introduction and Definition
Many gases are colourless and cannot be identified just by looking at them.
Chemists use simple laboratory tests to identify common gases such as hydrogen, oxygen, carbon dioxide and chlorine.
Each gas gives a distinctive, reliable result when tested.

Gas | Test | Positive Result | Why It Happens |
Hydrogen (H₂) | Hold a burning splint at the mouth of the test tube | Squeaky pop sound | Hydrogen burns rapidly in oxygen, creating a small explosion |
Gas | Test | Positive Result | Why It Happens |
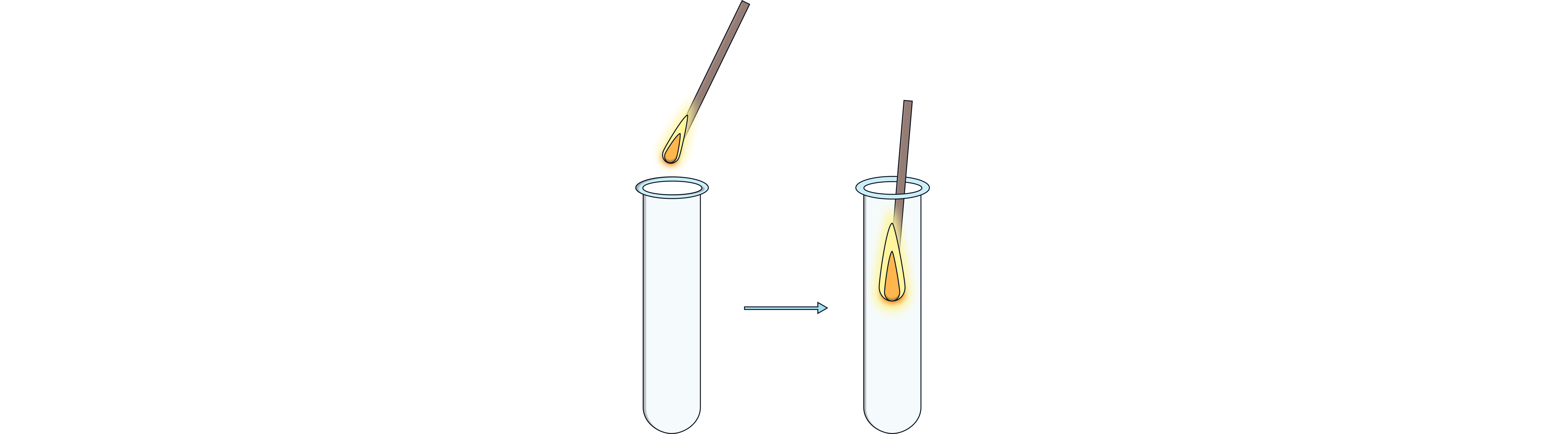
Oxygen (O₂) | Insert a glowing splint into the gas | Splint relights | Oxygen supports combustion |
Gas | Test | Positive Result | Why It Happens |
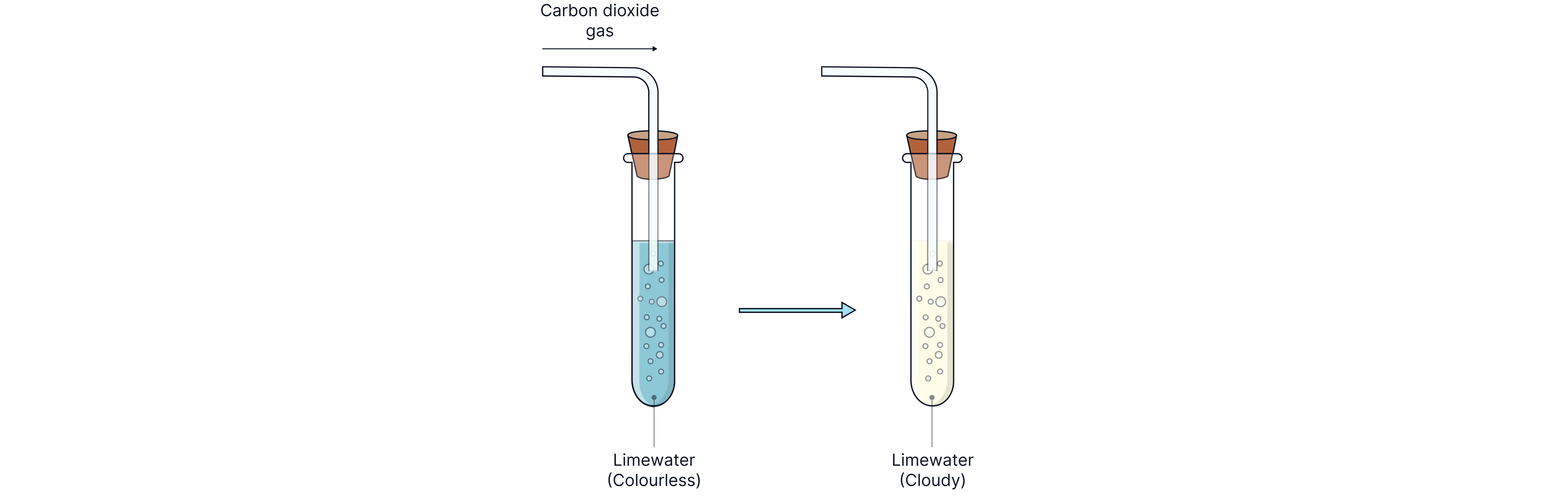
Carbon dioxide (CO₂) | Bubble the gas through limewater (calcium hydroxide solution) | Limewater turns milky/cloudy | A white precipitate of calcium carbonate forms |
Gas | Test | Positive Result | Why It Happens |
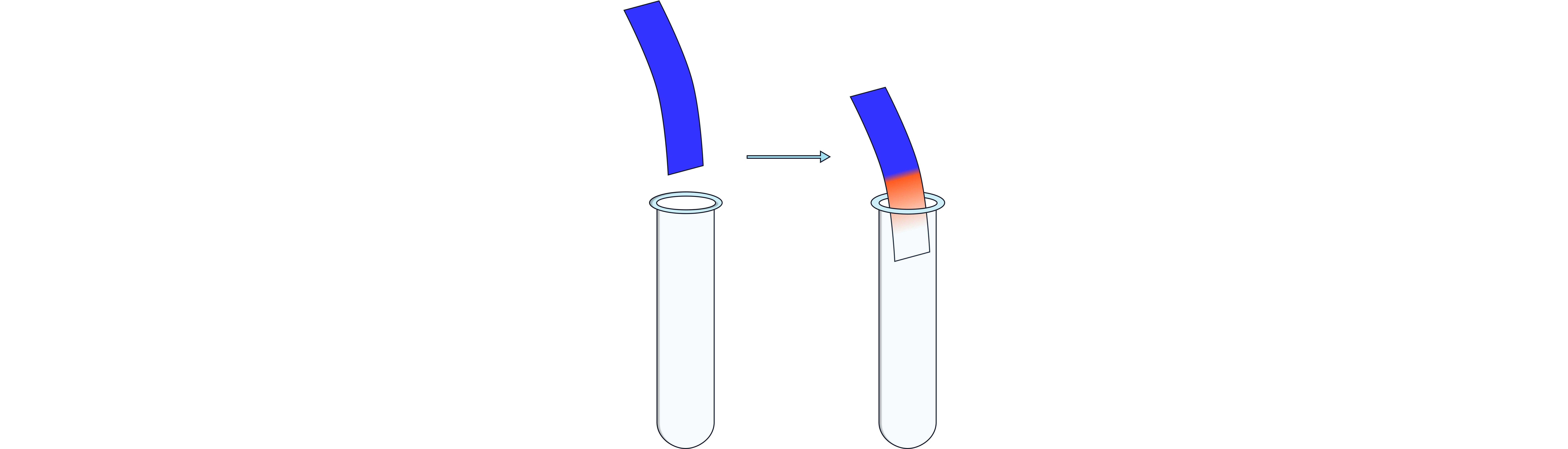
Chlorine (Cl₂) | Hold damp litmus paper in the gas | Litmus paper is bleached white | Chlorine is a bleaching agent and oxidises dyes |
Check Your Understanding
Recall
a. What is the best test for Hydrogen?
Answer:
a. Squeaky pop produced on introductions to a burning/lit splint
b. What is the test for Oxygen?
Answer:
b. Relights a glowing splint.
c. What happens when carbon dioxide is bubbled through limewater?
Answer:
c. Turns milky/cloudy.
d. What color is Chlorine gas?
Answer:
d. Green
Application
e. A gas relights a glowing splint. Identify the gas and explain.
Answer:
e. Oxygen; supports combustion.
f. A gas turns damp blue litmus paper red, but then white after some time. Identify the gas.
Answer:
f. Chlorine; bleaches litmus
g. Describe how you would confirm a gas is carbon dioxide.
Answer:
g. Bubble through limewater; turns milky/cloudy
Summary
Hydrogen: squeaky pop with a burning splint
Oxygen: relights a glowing splint
Carbon dioxide: limewater turns milky
Chlorine: damp litmus bleached white