NPK Fertilisers
Emmanuel Opoku
Teacher

Contents
NPK Fertilisers
What Are NPK Fertilisers?
NPK fertilisers are formulations containing compounds of the elements:
- Nitrogen (N) – for healthy leaf and stem growth
- Phosphorus (P) – for strong root development
- Potassium (K) – for overall plant health and resistance to disease
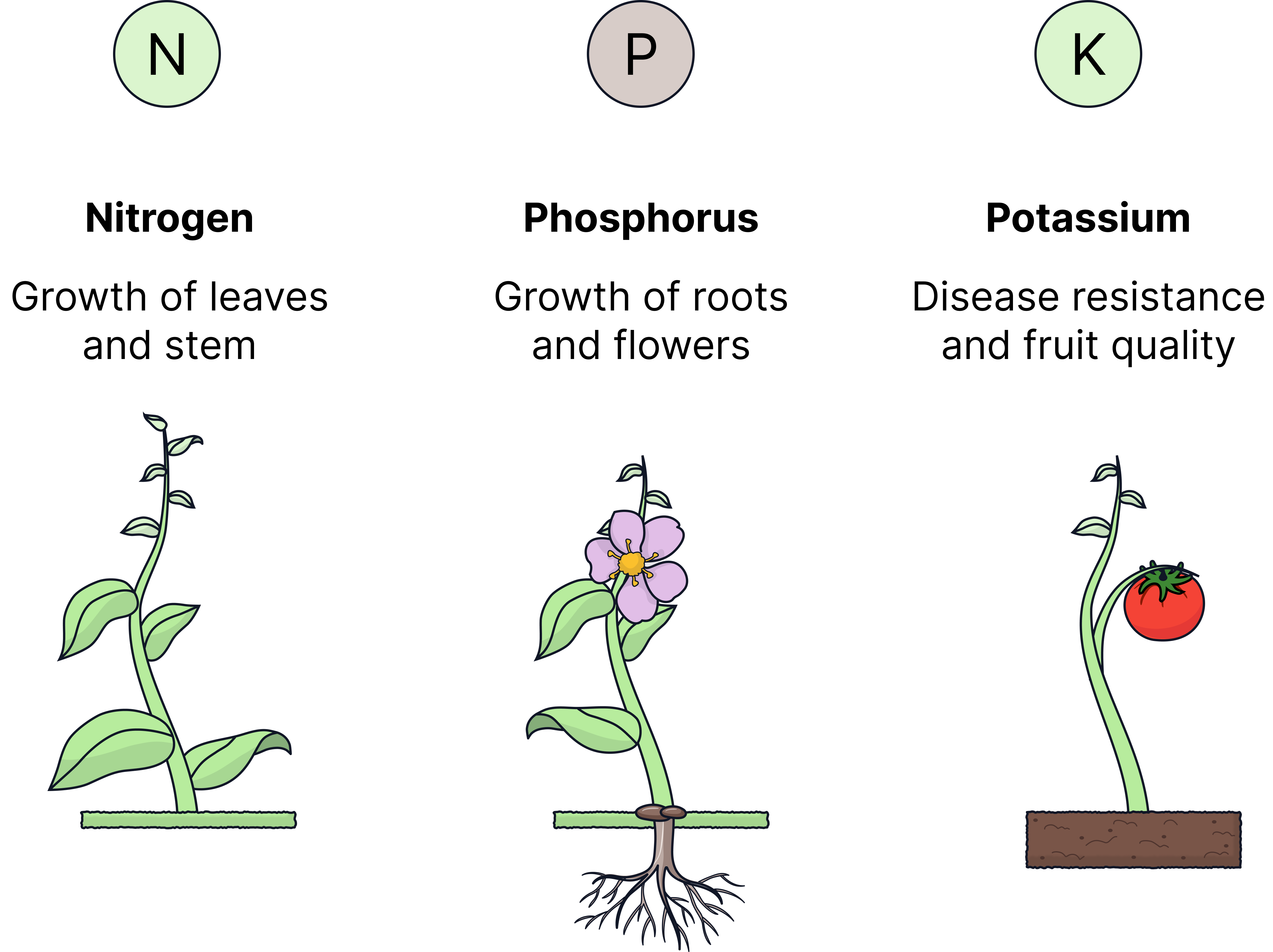
These elements are essential for plant growth. If soils lack them, crop yield and quality decrease, as plants cannot make enough proteins and DNA.
NPK stands for the three essential elements plants need: Nitrogen, Phosphorus, and Potassium.
How NPK Fertilisers Are Made
Industrial production of fertilisers involves several processes to ensure the right nutrient balance.
These fertilisers are formulation so they contain specific percentages of each element to meet different soil and crop needs.
Tip: NPK fertilisers are designed for precision farming - they give plants exactly what they need, in the right amounts, unlike natural manure which varies in nutrient content from batch to batch.
1. Nitrogen Compounds
Nitrogen compounds come from ammonia (NH₃) produced by the Haber Process.
Ammonia can react with different acids to form useful ammonium salts:
|
Reaction |
Product |
Name of Fertiliser |
|---|---|---|
|
2 NH₃ + H₂SO₄ → (NH₄)₂SO₄ |
Ammonium sulfate |
Nitrogen + sulfur source |
|
NH₃ + HNO₃ → NH₄NO₃ |
Ammonium nitrate |
High nitrogen content |
Ammonium nitrate is a key nitrogen fertiliser - it provides nitrogen from both ammonia and nitric acid, making it especially effective for boosting plant growth.
2. Potassium and Phosphorus Compounds
- Potassium chloride (KCl) and potassium sulfate (K₂SO₄) are mined directly from the ground and used as sources of potassium.
- Phosphate rock is also mined, but it cannot be used directly because it is insoluble in water.
To make it soluble and useful as a fertiliser, it must be treated with acids:
|
Treatment |
Reaction Products |
Type of Fertiliser |
|---|---|---|
|
Phosphate rock + nitric acid |
Phosphoric acid + calcium nitrate |
Soluble source of phosphorus |
|
Phosphate rock + sulfuric acid |
Calcium sulfate + calcium phosphate |
Single superphosphate |
|
Phosphate rock + phosphoric acid |
Calcium phosphate only |
Triple superphosphate |
Fertiliser Production – Lab Vs Industry
|
Feature |
Laboratory Preparation |
Industrial Production |
|---|---|---|
|
Scale |
Small (batch process) |
Continuous, large scale |
|
Reactants |
Dilute acids and ammonia solution |
Concentrated acids and ammonia gas |
|
Control |
Safer, slower, easier to manage |
Faster, more exothermic |
|
Example |
Titration and crystallisation of ammonium nitrate |
Direct reaction in large vats (heat used to evaporate water) |
In industry, safety and efficiency are key - the process must run continuously to meet agricultural demands and reduce waste and energy consumption.
Check Your Understanding
Recall
Name the three essential elements found in NPK fertilisers.
Answer:
Nitrogen, Phosphorus, and Potassium
Write the word and symbol equation for the reaction between ammonia and nitric acid.
Answer:
- Word: Ammonia + Nitric acid → Ammonium nitrate
- Symbol: NH₃ + HNO₃ → NH₄NO₃
Explain why phosphate rock cannot be used directly as a fertiliser.
Answer:
Phosphate rock is insoluble in water, so plants cannot absorb its nutrients directly
State two potassium compounds used in fertilisers.
Answer:
Potassium chloride (KCl) and Potassium sulfate (K₂SO₄)
Apply Your Knowledge
Describe how phosphate rock is treated to make a fertiliser.
Answer:
Phosphate rock is treated with acids (nitric, sulfuric, or phosphoric) to produce soluble salts that plants can absorb
Compare the production of ammonium nitrate in the lab and in industry.
Answer:
In the lab, dilute acids are used and the process involves titration and crystallisation; in industry, concentrated acids and ammonia gas are used in a continuous, exothermic process
Explain why industrial fertiliser production needs to be continuous.
Answer:
Continuous production ensures constant supply, reduces costs, and maximises efficiency for global agricultural demand
Challenge Question
Using the table below, evaluate which fertiliser type would be most suitable for large-scale commercial farming.
|
Method |
Advantages |
Disadvantages |
|---|---|---|
|
Ammonium nitrate (NH₄NO₃) |
High nitrogen content, fast-acting |
Expensive and energy-intensive to produce |
|
Ammonium sulfate ((NH₄)₂SO₄) |
Cheaper and safer to make |
Lower nitrogen content |
|
Natural manure |
Eco-friendly and renewable |
Inconsistent composition, slow nutrient release |
Answer:
- For large-scale farming, ammonium nitrate is most suitable.
- Although it’s more expensive to produce, it has a high nitrogen concentration, giving faster plant growth and higher yields.
- Ammonium sulfate is cheaper but provides less nitrogen, while manure is not practical on an industrial scale because it releases nutrients too slowly and varies in composition
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!