Relative Formula Mass
Lajoy Tucker
Teacher

Contents
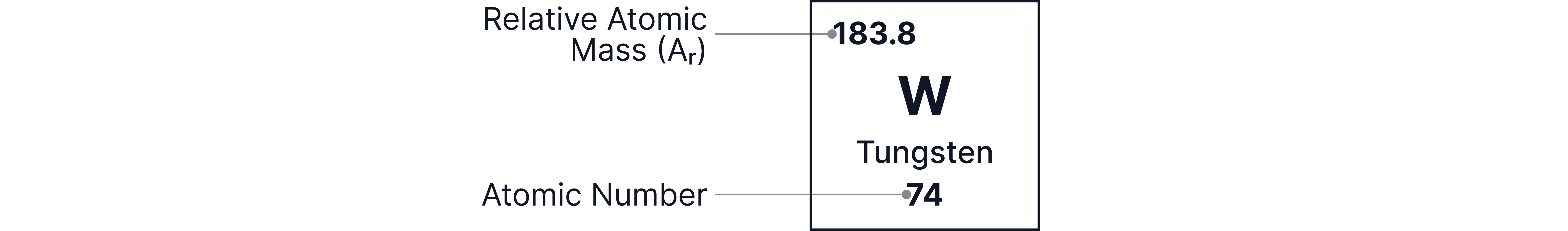
Relative Atomic Mass
The relative atomic mass of an element is the weighted average mass of an atom of an element, compared to 1/12th the mass of a carbon-12 atom.
Details on calculating relative atomic mass can be found in the Isotopes and Relative Atomic Mass note.
Relative Atomic Masses of all elements can be found on your Periodic Table
Relative Molecular Mass
Relative molecular masses are calculated as the sum of the relative atomic masses of the atoms in the molecule
Example 1: Water
The molecular formula of water is
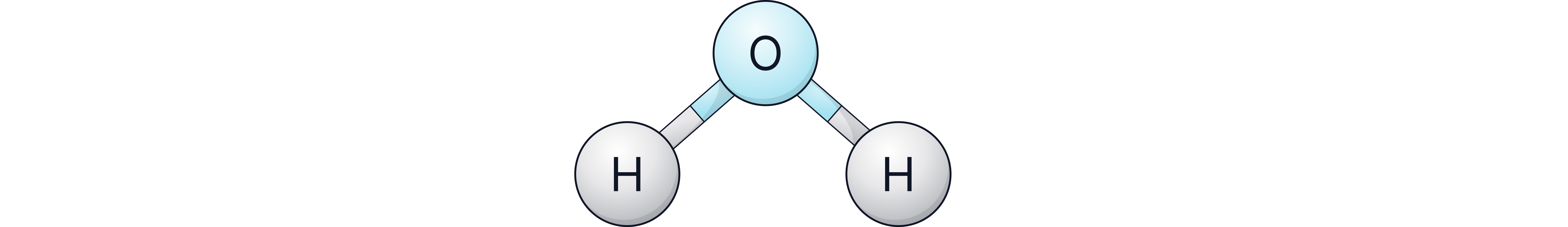
The relative atomic masses of hydrogen and oxygen are 1 and 16, respectively
of = (2×H) + O
of = (2×1) +16 = 18
The relative molecular mass of water is 18
Example 2: Trichloromethane
The molecular formula of trichloromethane is .
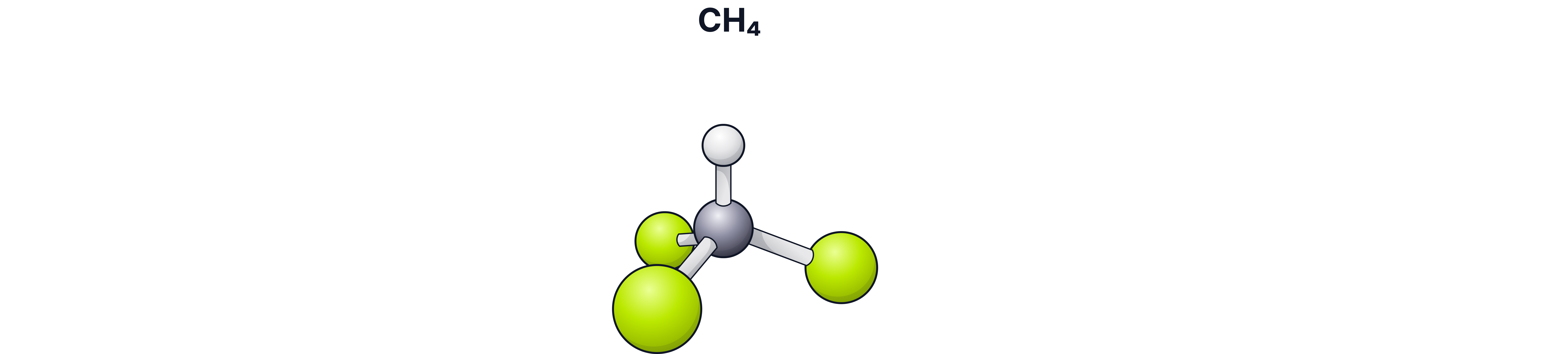
The relative atomic masses of carbon, hydrogen, and chlorine are 12, 1, and 35.5, respectively.
of = C + H + (3×Cl) = 119.5
The relative molecular mass of is 119.5.
Relative Formula Mass
The relative formula mass is essentially the same as the relative molecular mass but applies to giant structures e.g. ionic compounds, where species do not exist as discrete units.
Example: Sodium Chloride
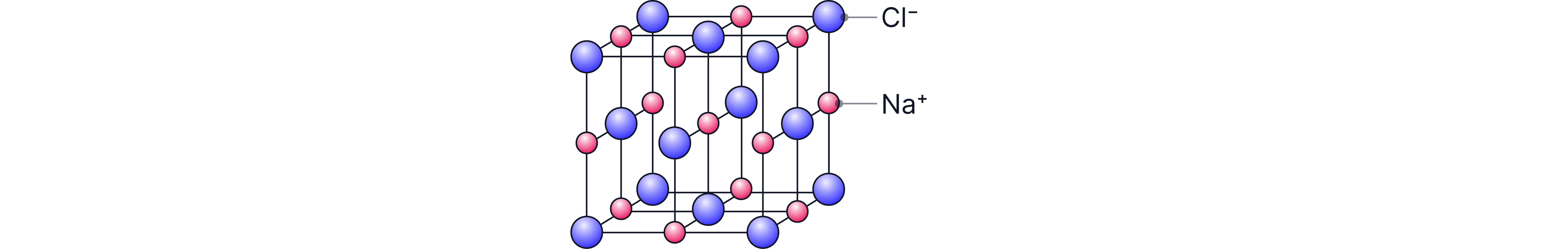
The formula of sodium chloride is .
The relative atomic masses of sodium and chlorine are 23 and 35.5, respectively
of = 23 + 35.5 = 58.5
The relative formula mass of sodium chloride is 58.5.
Example: Copper (II) Sulfate Crystals,

. is an example of a dot formula.
The ‘’ represents water of crystallisation in a hydrated ionic compound. These water molecules are a key part of the lattice structure and must be included when calculating .
Tip! Students usually remember to multiply the 2 hydrogens by 5 but forget to multiply the oxygen by 5. If you add the water up as a whole, you limit your chances of making this mistake.
Check for Understanding
Recall
a) What is meant by the relative atomic mass (Ar) of an element?
Answer:
a) The weighted average mass of an atom of an element compared with 1/12 of the mass of a carbon-12 atom.
b) How is relative molecular mass (Mr) calculated for a molecule?
Answer:
b) By adding the relative atomic masses of all the atoms in the molecule.
c) When is relative formula mass used instead of relative molecular mass?
Answer:
c) For ionic compounds / giant ionic structures where substances do not exist as individual molecules.
Application
d) Calculate the relative molecular mass (Mr) of water, H₂O.
(Ar: H = 1, O = 16)
Answer:
d)
Mr = (2 × 1) + 16
= 18
e) Calculate the relative formula mass (Mr) of sodium chloride, NaCl.
(Ar: Na = 23, Cl = 35.5)
Answer:
e)
Mr = 23 + 35.5
= 58.5
Challenge (HT)
f) Calculate the relative formula mass (Mr) of hydrated magnesium sulfate, MgSO₄·7H₂O.
(Ar: Mg = 24, S = 32, O = 16, H = 1)
Answer:
f)
Mr = Mg+ S + (4 × O) + 7(H₂O)
= 24 + 32 + (4 × 16) + 7[(2 × 1) + 16]
= 24 + 32 + 64 + 126
= 246
Key points to remember:
Relative atomic mass accounts for isotopes' masses and abundances.
Relative molecular mass is used for covalent molecules and calculated by summing the atomic masses of the atoms in the molecule.
Relative formula mass applies to ionic compounds, calculated similarly to relative molecular mass but only for the empirical formula.