The modern periodic table
Lajoy Tucker
Teacher

Arrangement of Elements
The elements in the periodic table are arranged in order of atomic number (the number of protons in the nucleus).
This order ensures that elements with similar properties appear in the same vertical columns, called groups.
The name “periodic” means that certain chemical properties repeat at regular intervals or periods.
Structure of the Periodic Table
Rows in the table are called periods. Each period represents a new electron shell being filled.
Columns are called groups. Each group contains elements that share similar chemical properties.
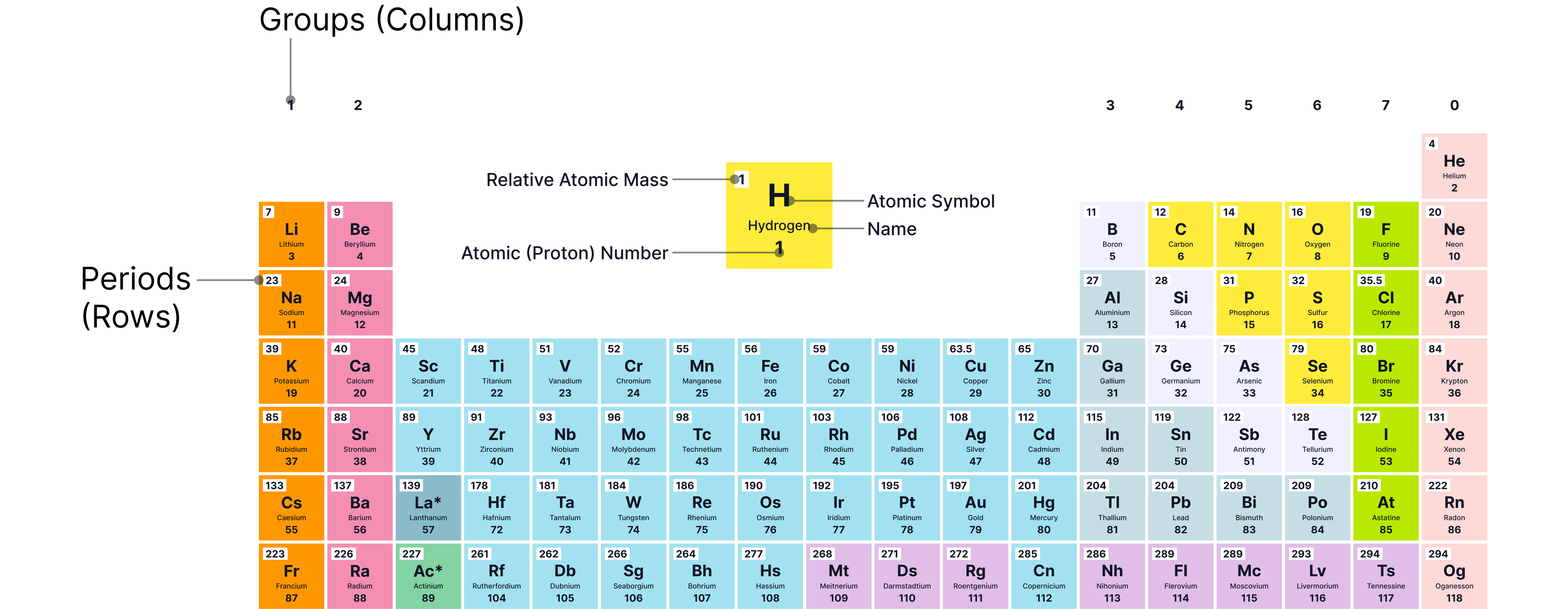
Feature | Description |
Periods (rows) | Show how many electron shells the atoms have |
Groups (columns) | Show how many electrons are in the outer shell |
Atomic number | Tells how many protons an atom has |
Link Between Position and Electron Arrangement
The position of an element in the periodic table is directly related to its electron configuration.
The group number shows the number of outer electrons.
The period number shows the number of electron shells.
Examples:
Lithium (Li) – atomic number 3 → electronic structure 2,1 → Group 1, Period 2
Oxygen (O) – atomic number 8 → electronic structure 2,6 → Group 6, Period 2
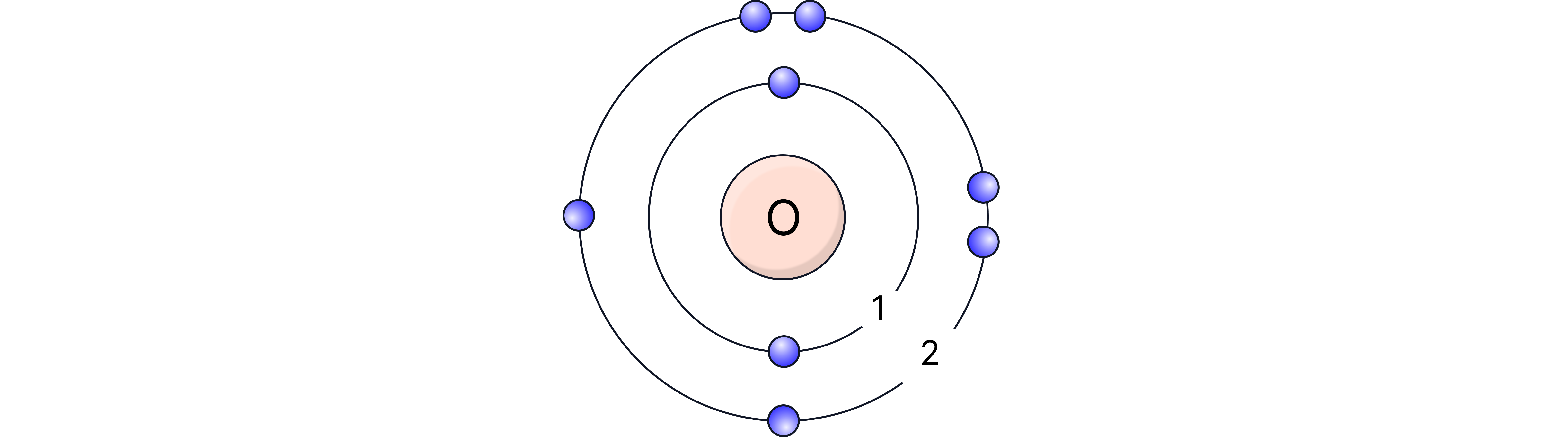
Chlorine (Cl) – atomic number 17 → electronic structure 2,8,7 → Group 7, Period 3
This pattern means we can predict an element’s reactivity and chemical behaviour based on its position in the table.
Similar Properties in Groups
Elements in the same group have similar chemical reactions because they have the same number of outer electrons.
For example:
Group 1 (alkali metals) all react with water to form a metal hydroxide and hydrogen.
Group 7 (halogens) all form salts when reacting with metals.
Group 0 (noble gases) are unreactive because their outer shells are full.
Predicting Reactivity
Reactivity trends can be predicted from the group and period:
In Group 1, reactivity increases down the group (outer electron lost more easily).
In Group 7, reactivity decreases down the group (harder to gain an electron).
The group number therefore helps to predict how elements will react and what type of compounds they form.
Check for Understanding
Recall
a) In what order are elements arranged in the modern periodic table?
Answer
Elements are arranged in order of atomic number.
b) What are the horizontal rows (periods) in the periodic table called?
Answer
Periods
c) What does the group number tell you about an atom?
Answer
The number of electrons in the outer shell.
Application
d) An element has the electronic structure 2,8,4. Identify its group and period.
Answer
Group 4 and Period 3.
e) Explain why elements in the same group have similar chemical properties.
Elements in the same group have the same number of outer electrons, so they react in similar ways.
Challenge
f) Explain how the position of an element in the periodic table can be used to predict its chemical behaviour.
Answer
The group number shows the number of outer electrons and the period shows the number of electron shells. This allows scientists to predict reactivity, chemical properties, and the types of compounds an element will form.
Summary
Elements are arranged in order of atomic number in the periodic table.
Groups contain elements with similar properties due to the same number of outer electrons.
Periods show how many electron shells are filled.
You can predict reactivity and reactions of elements using their position in the table and their electron arrangement.
The periodic table reveals repeating (periodic) patterns in chemical behaviour.