Specific Heat Capacity
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
Specific Heat Capacity
Imagine walking along a hot beach: the sand feels hot, yet the nearby seawater feels cool. Both have been heated by the Sun for the same amount of time, so why do they feel so different? The answer lies in their specific heat capacities.
When a substance is heated (and it is not changing state), the energy transferred increases the kinetic energy of its particles, which causes its temperature to rise. However, different materials require different amounts of energy to raise their temperature.
The specific heat capacity of a substance tells you how much energy is needed to raise the temperature of of a substance by . Substances with a high specific heat capacity (like water) warm up slowly because they require more energy to increase their temperature. Substances with a low specific heat capacity (like sand) heat up quickly because they need much less energy.
The energy transferred to an object to increase it's temperature by is the same amount as the energy emitted by an object when it's temperature decreases by .
We can calculate the specific heat capacity of a substance using the following equation.
Formula:
We can use this equation to calculate how much energy is needed to bring of water at up to boiling point in a kettle, if we know the specific heat capacity of water is .
First, we need to convert by dividing by , so becomes .
Next, we need to find the change in temperature. Water will boil at , so the change in temperature will be:
Finally we will use the following equation:
Worked Example:
of tea in a cup cools down from to . The specific heat capacity of tea is . Calculate the energy transferred to the surroundings by the tea.
Answer:
First, we need to convert to by dividing by :
Then, we need to calculate the change in temperature:
Calculating energy transferred:
Worked Example:
Aluminium has a specific heat capacity of .
A aluminium block, originally at , is heated by a power of for minutes.
How much energy is absorbed by the aluminium?
What will the final temperature of the aluminium be, assuming no heat dissipation to the surroundings?
Answer:
First, we need to convert minutes into seconds:
Then calculate the energy transferred:
First, we need to calculate the mass of the substance in . To do this, we need to divide the mass by , so . Then we will use the equation;
This is the change in temperature, so to find the final temperature:
Specific Heat Capacity RP
We can find the specific heat capacity of solid, metal block by heating it with an electrical heater (immersion heater) and measuring the:
The energy transferred using the Joulemeter
The mass using a scale/balance
The change in temperature using a thermometer
Method
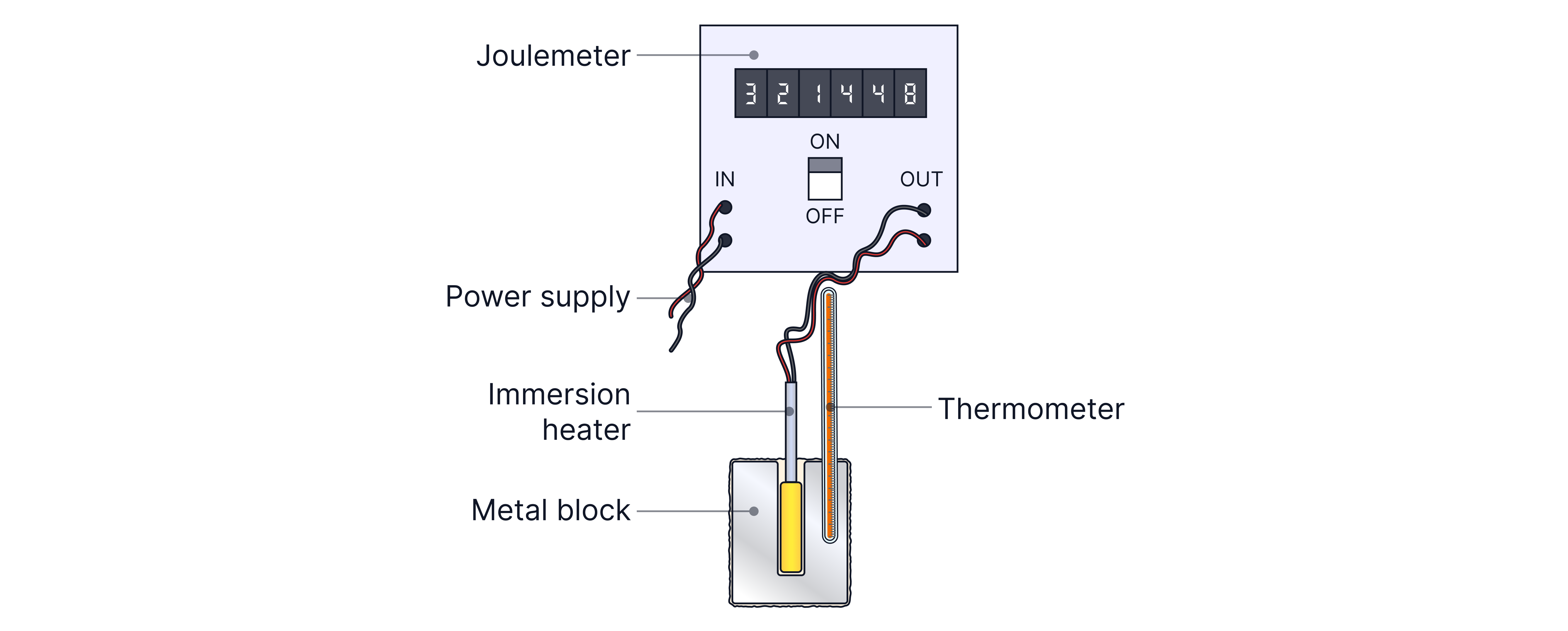
Use a balance to measure the mass of the metal block (ensure it reads zero before beginning).
Set up the apparatus as shown above.
Wrap the block in insulation to reduce energy loss to the surroundings.
Measure and record the block's initial temperature using the thermometer.
Switch on the power supply and start a stopwatch.
Record the final temperature after and calculate the temperature change.
Record the total energy transferred to the block using the joulemeter.
Use the equation to find the specific heat capacity.
In some scenarios, you will not have a joulemeter available to you. In these cases, to find the energy supplied to the block, you need to measure the potential difference across the heater with a voltmeter, the current with an ammeter, calculate the power supplied using the equation,, and then multiply by the time to obtain the total energy supplied, . We can adapt the method by recording the block's temperature every 60 seconds and calculating the energy supplied at each interval to see how the data changes.
For example, if I wanted to find the specific heat capacity of a copper block, heated using an electric immersion heater that is connected to a voltmeter reading and an ammeter reading,, I could calculate the power supplied using the equation:
Then, I calculate the energy transferred by using to get the following results:
Time / | Energy transferred / | Temperature / |
0 | 19 | |
60 | 23 | |
120 | 27 | |
180 | 31 | |
240 | 35 | |
300 | 38 |
Next, we need to calculate the change in temperature, as at a time = , the temperature is this is the initial temperature.
The total energy transferred to the block in the experiment would be so we can calculate the specific heat capacity:
If you look up the specific heat capacity of copper online, you will find it is . Our calculated value is higher as some energy was dissipated to the surroundings, not all of the energy supplied went into heating the block.
Worked Example:
A student wants to find the specific heat capacity of an aluminium block. Name the measurements the student should take and the apparatus used to take these measurements.
Answer:
Measure energy transferred using a joulemeter
Measure the mass of the block using a balance
Measure the temperature of the block every seconds for minutes using a thermometer.
Worked Example:
A student investigates the specific heat capacity of aluminium. They place a aluminium block on an insulating mat and insert a thermometer and a , immersion heater into the block. They switch on the heater for seconds. During this time, the temperature of the aluminium block rises from to .
Calculate the specific heat capacity of aluminium.
Assume that all electrical energy supplied is transferred to the block.
Answer:
Step - calculate the power of the heater:
Step - calculate the energy transferred:
Step - calculate the change in temperature:
Step - calculate the specific heat capacity:
Practice Questions
A student heats a block of aluminium using an electric heater. The temperature of the block rises from to . The specific heat capacity of aluminium is .
Calculate the temperature change of the block.
Calculate the energy transferred to the block.
State one reason why the actual temperature rise of the block might be lower than expected.
-> Check out Hannah's video explanation for more help.
Answer:
Some energy is lost to the surroundings
A student compares the heating of two liquids, A and B, using identical heaters supplying of energy to each liquid. Both liquids have a mass of . Liquid A increases in temperature by ; liquid B increases by .
Calculate the specific heat capacity of liquid A.
Calculate the specific heat capacity of liquid B.
Explain why liquid B has a lower temperature rise than liquid A, even though both received the same amount of energy.
-> Check out Hannah's video explanation for more help.
Answer:
Liquid B has a higher specific heat capacity. This means it requires more energy per kilogram per degree Celsius to increase its temperature.