Aldehydes & Ketones
Lajoy Tucker
Teacher

Contents
Aldehydes & Ketones
All carbonyls have a bond and examples consist of aldehydes and ketones. Aldehydes and ketones can be distinguished by the groups attached to the C of the bond.
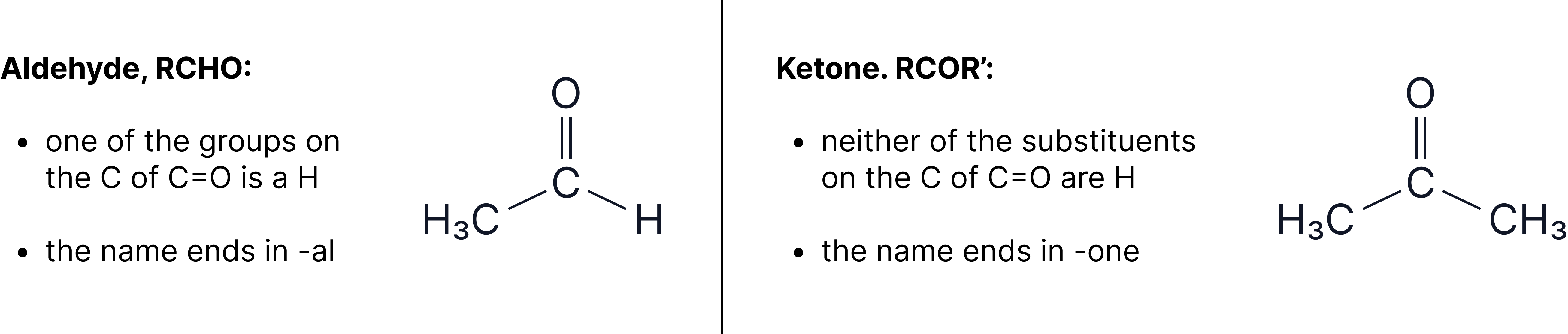
Preparation
Aldehydes are prepared by partially oxidising primary alcohols using acidified potassium dichromate is a distillation setup.
Ketones are prepared by oxidising secondary alcohols using the same oxidising agent.
Intermolecular forces
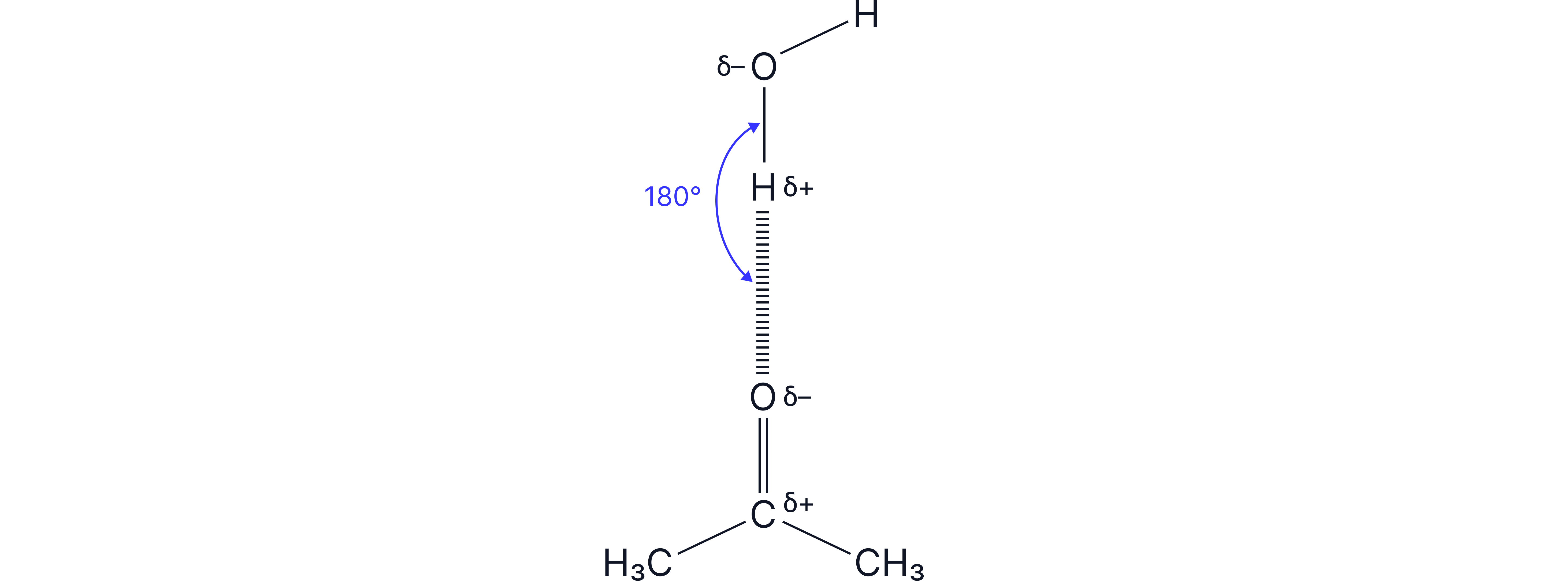
As there are no protons on the O of carbonyls, carbonyls are not able to hydrogen bond to one another, and instead they have permanent dipole interactions between them.
However, when carbonyls are in water, they are soluble to a certain degree (depending on the length of the carbon chain) since they can form hydrogen bonds with water.
Aldehydes and Ketones Explained
Test for Aldehydes and Ketone
Tollen’s Reagent | Fehling’s solution | |
Reagent | Cu2+ ion solution | |
Condition | heat | heat |
Reaction | Aldehyde → Carboxylic Acid | Aldehyde → Carboxylic Acid |
Observation with Aldehydes | Silver mirror forms | Blue solution changes to red precipitate |
Observation with Ketones | No visible reaction: solution remains colourless | No visible reaction: solution remains blue |
Oxidation
To collect the aldehyde when oxidising a prinary alcohol. Distillation must be used to remove and collect the aldehyde before it reacts further.
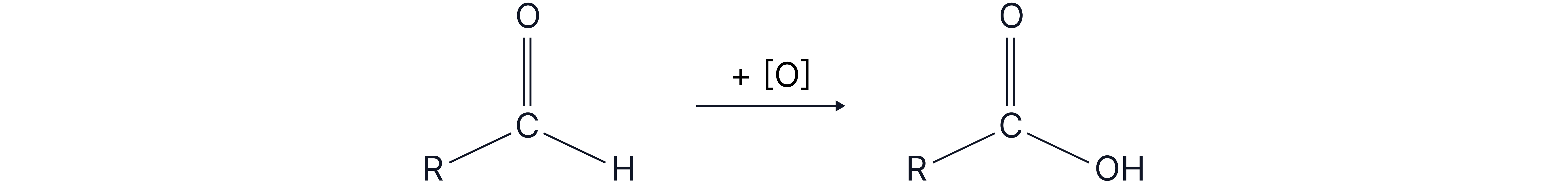
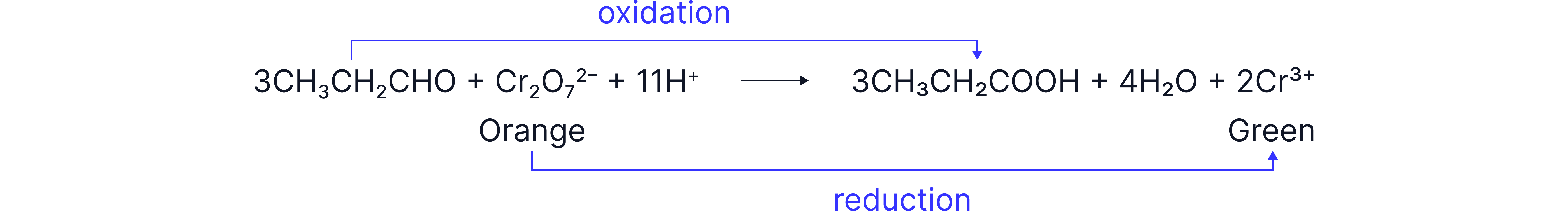
Aldehydes can be oxidesed to carboxylic acid using acidified potassium dichromate.
Regeant: and dilute
Condition: heat under reflux
Observation: orange solution turns green
Ketones cannot be oxidised further
Primary Alcohol → Aldehyde → Carboxylic Acid |
Secondary Alcohol → Ketone |
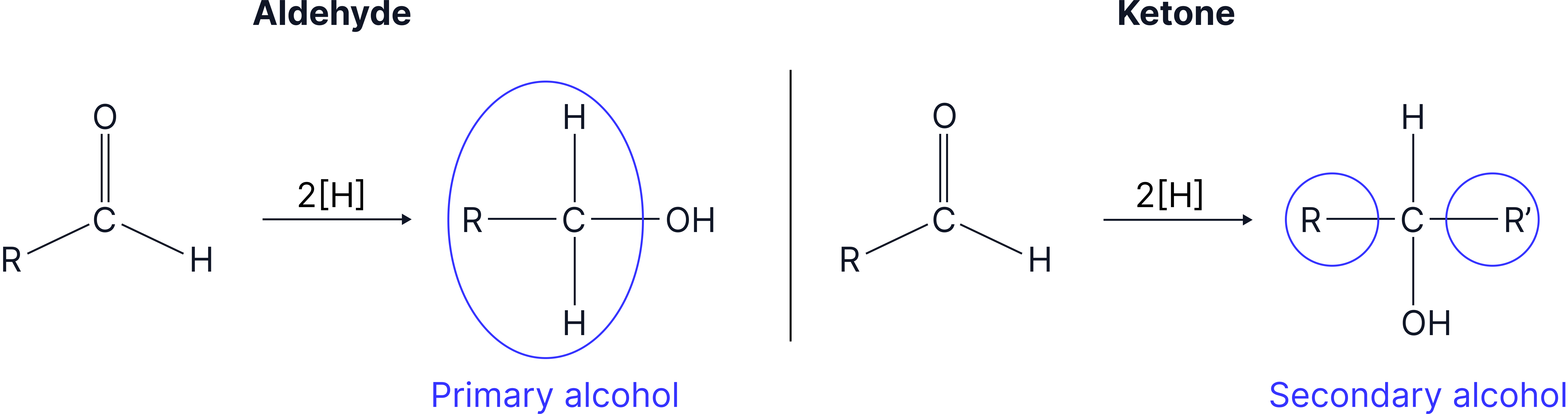
Reduction
Reduction is the opposite of oxidation , so aldehydes can be reduced to primary alcohols, and ketones will reduce to secondary alcohols. Both use in aqueous solution or in ethanol.
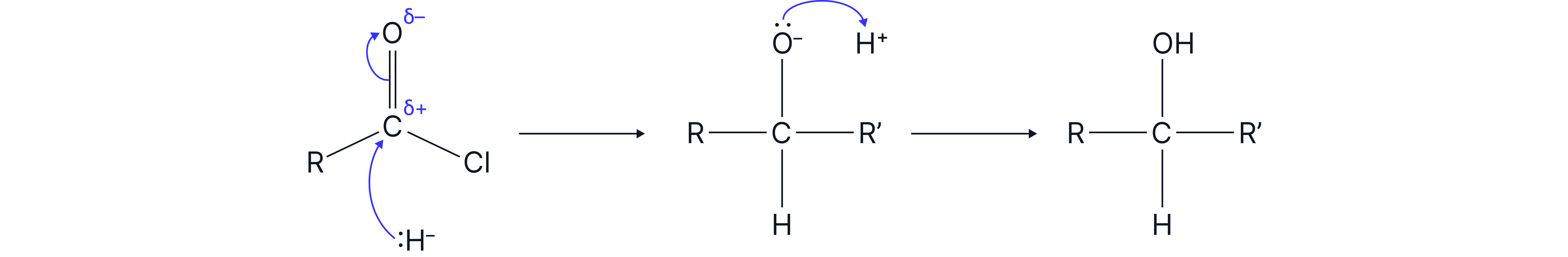
The mechanism for reduction involves H- ions wich act as a nucleophile and attack the carbonyl on . When water or a weak acid is present, the mechanism is a nucleophilic addition shown below
Practice Question
Question:
How would you differentiate between proponal and propanone?
Answer:
Add Tollens' reagent OR Fehling's solution
Proponal will produce silver mirror OR a brick-red precipitate
Propanone will show no reaction.
Key Tips and Reminders
Always use distillation when preparing aldehydes to prevent over-oxidation.
Ketones do not react with Tollens' or Fehling's solutions under normal conditions.
Make sure to quote observations accurately: "silver mirror" and "red precipitate", not just "colour change".