Chromatography
Lajoy Tucker
Teacher

Contents
Introduction
Chromatography is a technique used to separate and identify the components of a mixture. It separates substances by seeing how far or how quickly they travel with a liquid or gas (mobile phase) across a surface (stationary phase). Separation depends on the balance between solubility in the moving phase and retention by the stationary phase. The speed/distance travelled is based on the relative affinities (attractions) for the mobile and stationary phases.
Chromatography can be used to separate mixtures of organic compounds including amino acids and sugars.
Key Terms:
Stationary Phase: The phase that doesn’t move (e.g. paper in paper chromatography or silica in thin layer chromatography).
Mobile Phase: The phase that moves over the stationary phase (e.g. a solvent).
Chromatography Explained
How Polarity Affects Separation
In chromatography, "like dissolves like" is a key idea:
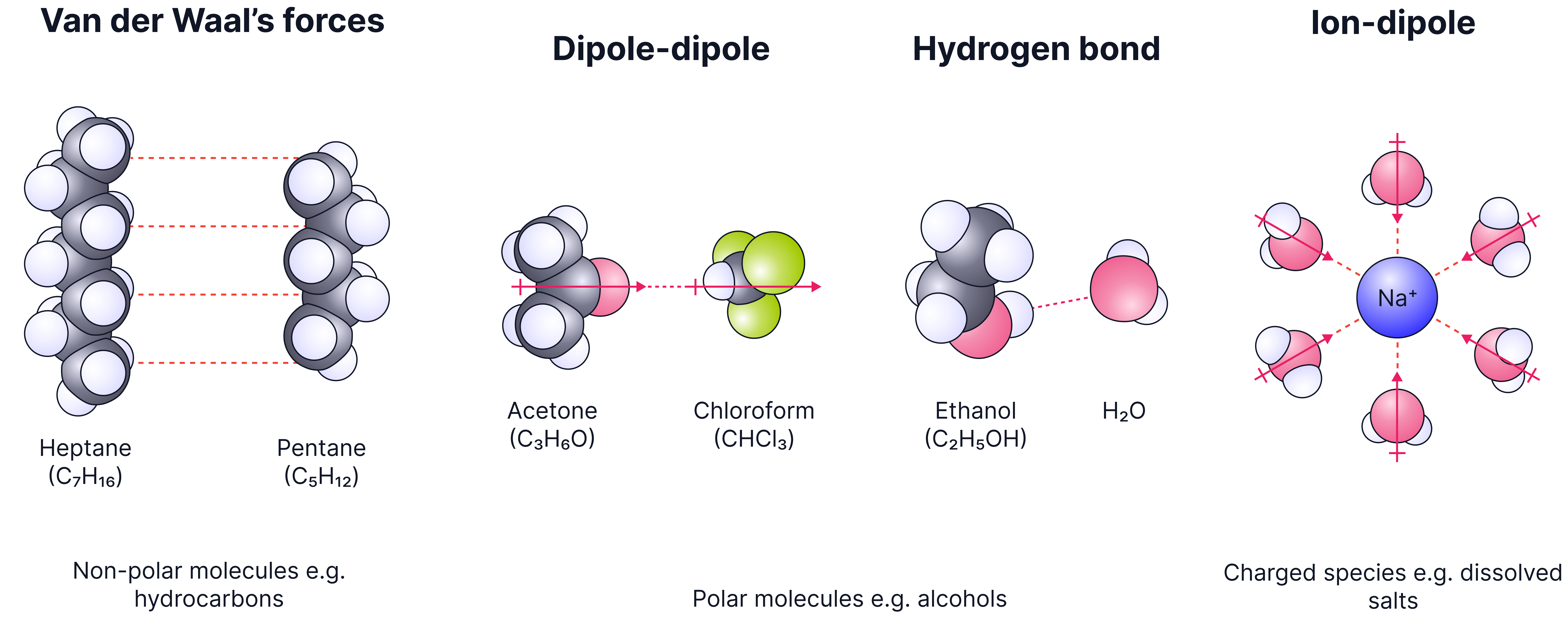
Polar compounds are more attracted to polar solvents.
Non-polar compounds are more attracted to non-polar solvents.
In TLC or paper chromatography:
The stationary phase (silica or paper) is usually polar.
The mobile phase (solvent or carrier gas) can be polar or non-polar depending on what you're separating.
For example, you're separating a mixture of two substances:
Substance A is polar
Substance B is non-polar
If your mobile phase is non-polar, Substance B will:
Travel further/faster
Meanwhile, Substance A will:
Stick more to the stationary phase (because both are polar)
Travel a shorter distance/more slowly
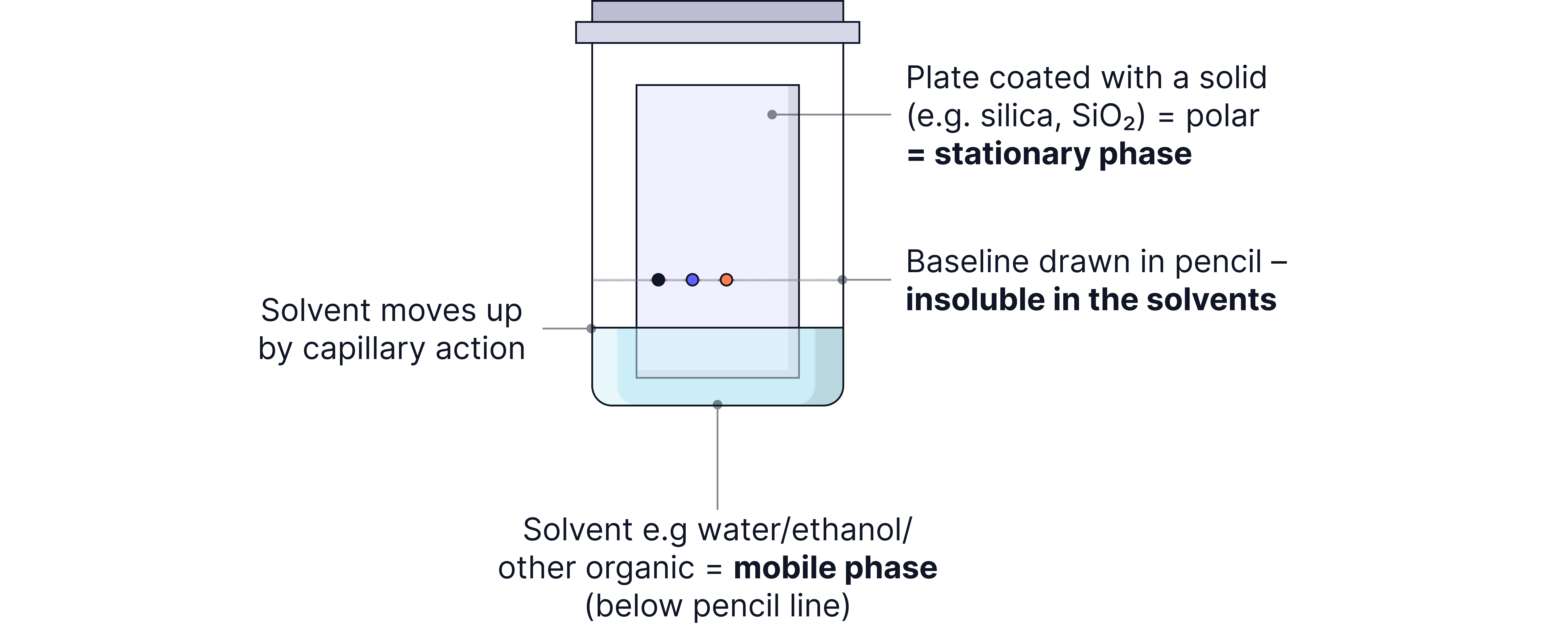
Thin-Layer Chromatography (TLC)
Stationary Phase: Thin layer of silica or alumina on a glass/plastic plate.
Mobile Phase: A solvent.
TLC Experimental Set-Up
Chromatogram
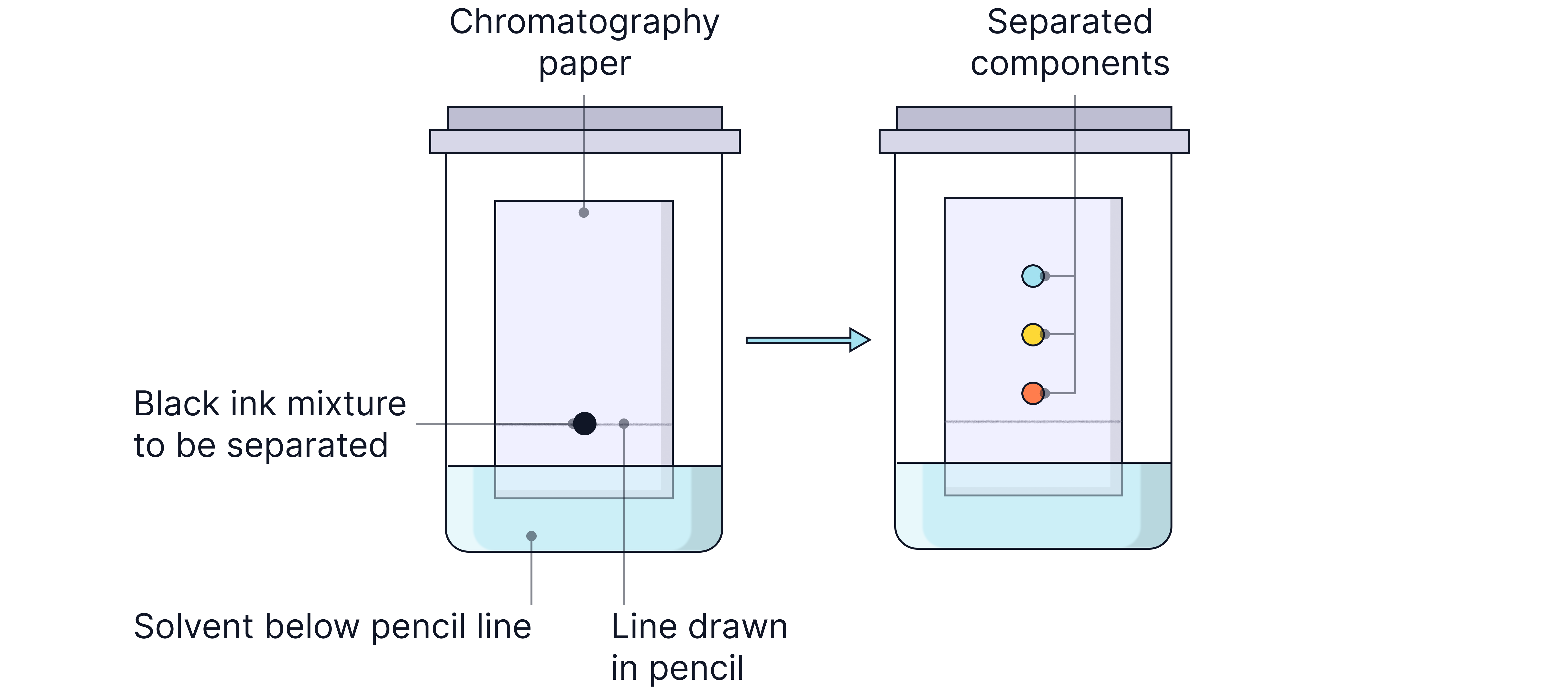
The distance a sample travels depends on its affinity to the silica in the stationary phase compared to its solubility in the mobile phase (solvent).
Retention Factor (Rf): Quantitatively describes how far a compound travels relative to the solvent front.
Rf = Distance travelled by substance / Distance travelled by solvent
The distance is always measured from the baseline where the sample was spotted.
Example: If the solvent front moved and a spot moved , then:
Interpreting Rf values
Substances that interact more strongly with the stationary phase (e.g. polar compounds binding to polar silica) travel shorter distances → lower Rf.
Substances that are more soluble in the mobile phase travel further → higher Rf.
Example: On a silica plate (polar) with hexane as the solvent (non-polar), a non-polar hydrocarbon will travel far (high Rf), while a polar alcohol may stick to the surface and barely move (low Rf).
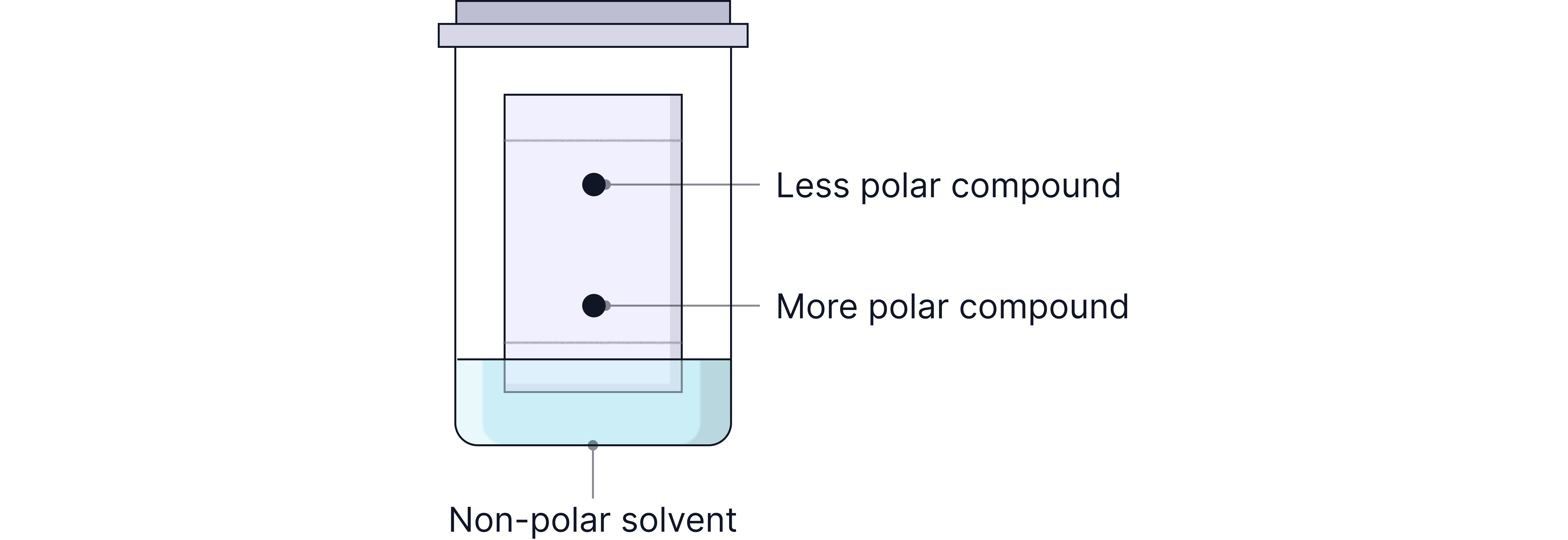
You can adjust the separation by choosing a different solvent:
A more polar solvent helps polar substances move further.
A less polar solvent slows down polar substances and speeds up non-polar ones.
Identifying substances using Rf
If pure known substances (standards) are run on the same plate under identical conditions, the Rf values of unknown spots can compared with these standards.
If they match, the unknown can be identified.
Example: An unknown amino acid with Rf = 0.45 matches the Rf of known glycine under the same solvent conditions → the unknown is likely glycine.
Amino Acids
Amino acids can be separated and identified using thin-layer chromatography. As amino acids are colourless, a locating agent such as ninhydrin or ultraviolet light is used to visualise the spots on the chromatogram.
Worked Example:
A student runs a TLC of three unknown amino acids on a silica plate using ethanol–water as the solvent. After the solvent front reaches near the top, the plate is dried and sprayed with ninhydrin, which reacts with amino acids to give purple spots.
Distances moved are measured:
Spot 1: 3.6 cm
Spot 2: 4.4 cm
Spot 3: 2.8 cm
Solvent front: 6.0 cm
Rf values:
Spot 1:
Spot 2:
Spot 3:
Known standards in the same solvent
Amino acid | Rf value |
Serine | 0.47 |
Glycine | 0.60 |
Aspartic acid | 0.40 |
Alanine | 0.73 |
Valine | 0.82 |
Identification of amino acids as:
Spot 1 = Glycine, Spot 2 = Alanine, Spot 3 = Serine
Gas Chromatography (GC)
Stationary Phase: Coated solid in a metal column or solid coated with a high boiling point liquid.
Mobile Phase: Inert carrier gas (e.g. helium or nitrogen).
Used for: Volatile substances, often coupled with mass spectrometry (GCMS).
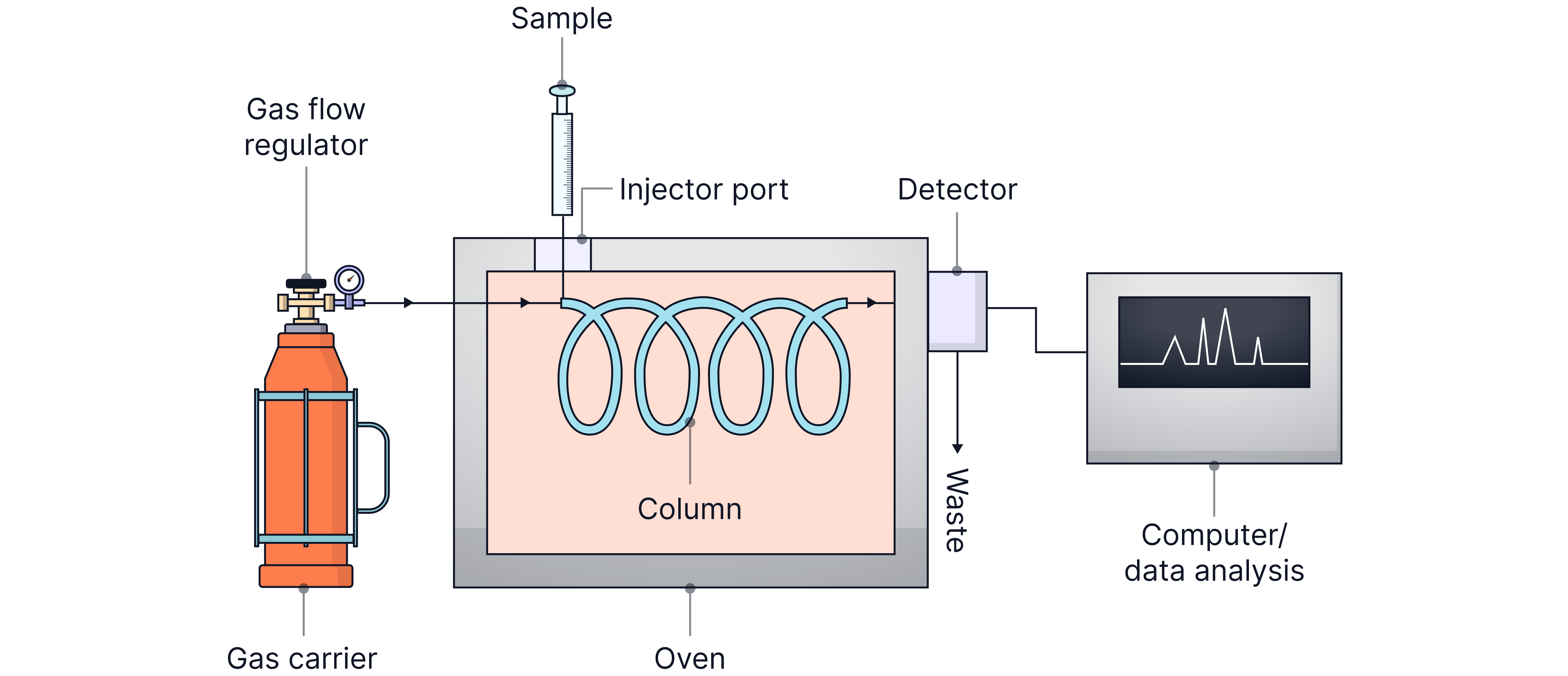
The carrier gas with the sample is passed through the column under pressure at high temperatures.
Different substances interact differently with the stationary phase – some dissolve more, some less.
Retention Times
Retention time = time a compound takes to pass through the column.
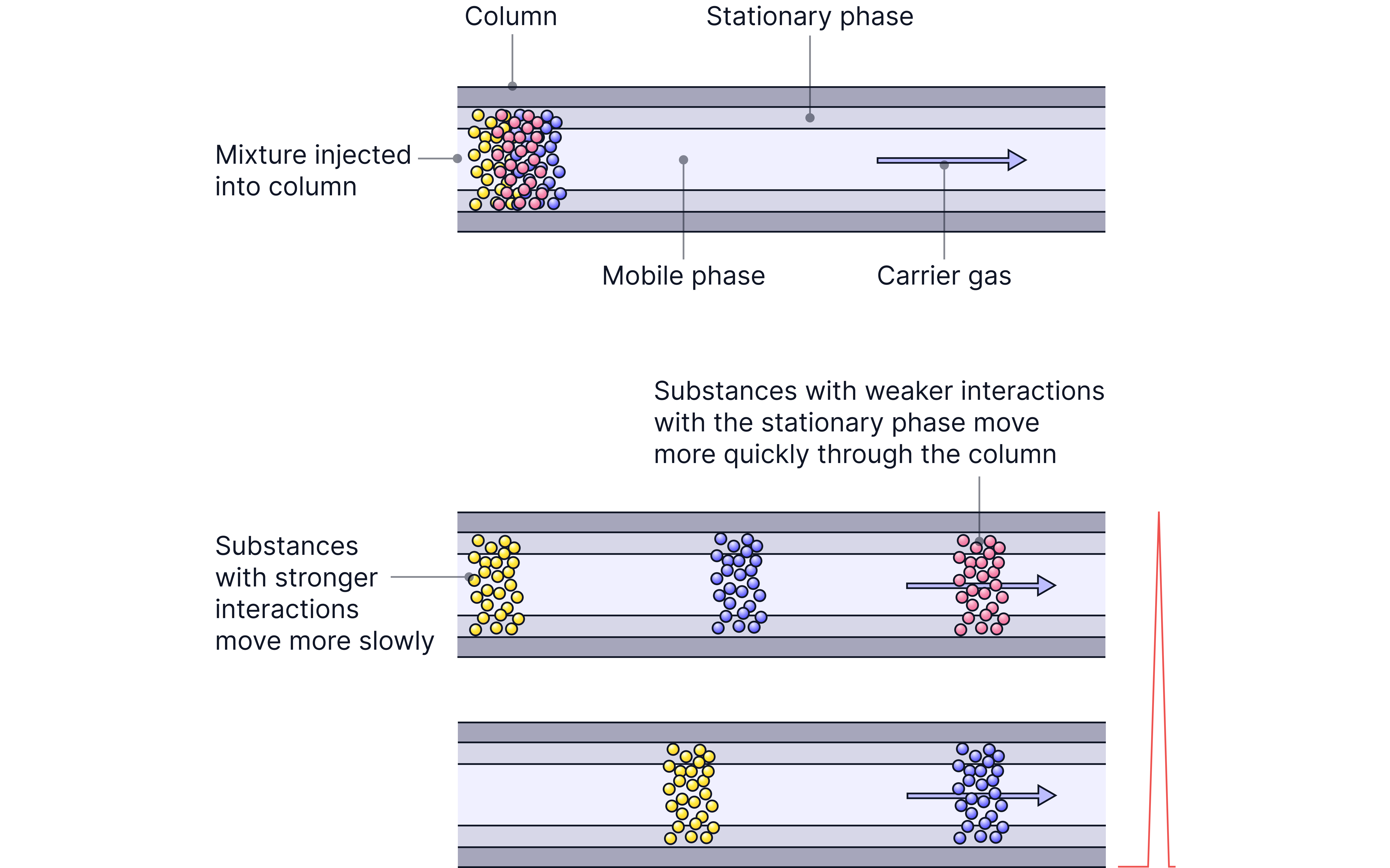
Each component leaves the column at different times. This is called the retention time.
Short retention time = weak interactions with stationary phase
Long retention time = strong interactions with stationary phase
Worked Example
A mixture of ethanol, propanol, and butanol is analysed using a polar stationary phase.
Ethanol interacts least with the stationary phase → shortest retention time.
Butanol interacts more strongly → longest retention time.
The chromatogram shows three peaks at different times, which can be matched with standards to identify each alcohol.
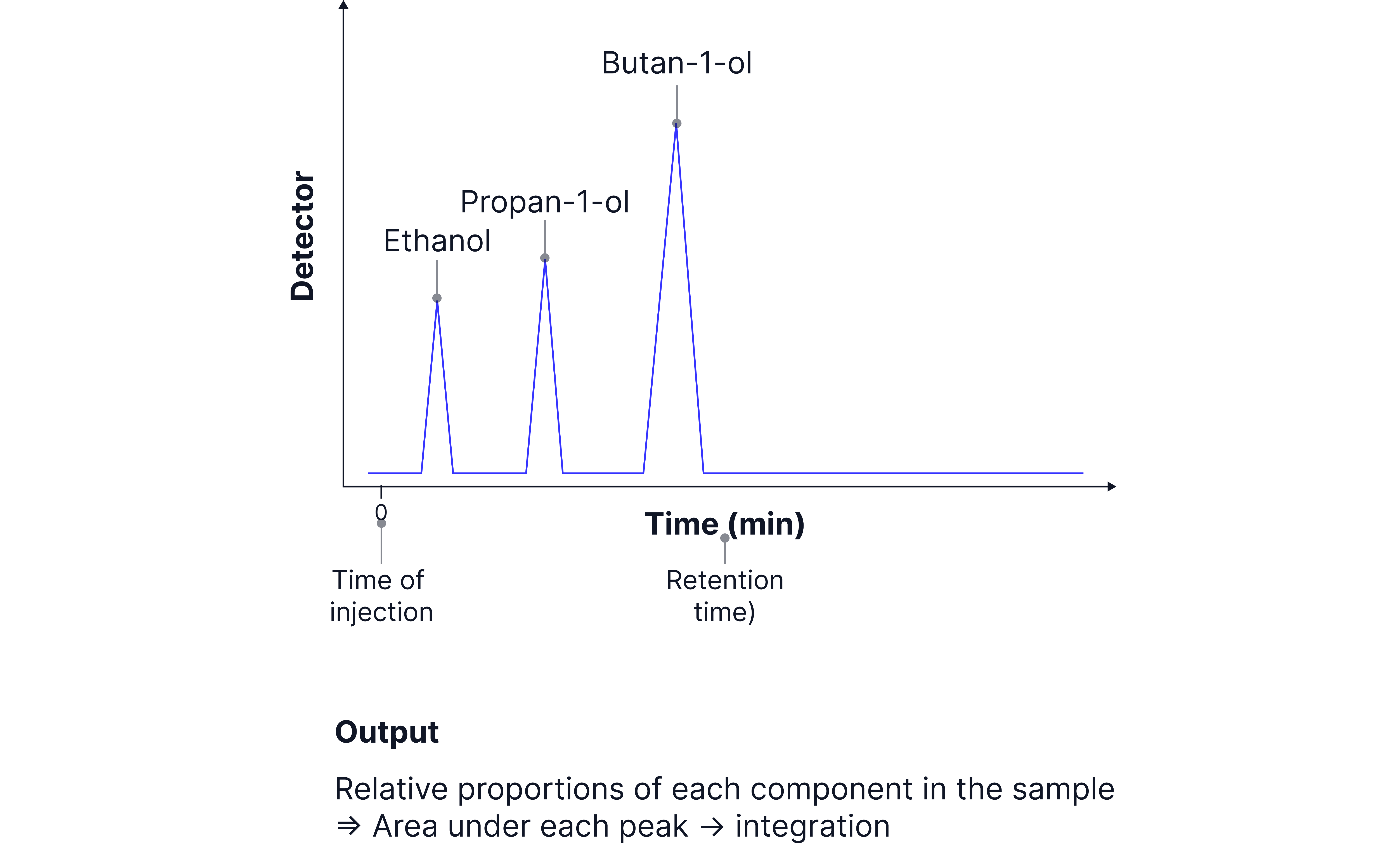
The area under each peak also provide information on how much of each component there is in the sample mixture.
For example, in the output above, there is more butan-1-ol than propan-1-ol, and more propan-1-ol than ethanol in the sample mixture.
Combining with MS (GC-MS)
GC separates, MS identifies by producing a mass spectrum allowing determination of molecular formula.
This is useful for identifying unknown compounds in complex mixtures.
Column Chromatography
Stationary Phase: A glass column packed with a solid (usually silica or alumina)
Mobile Phase: A solvent
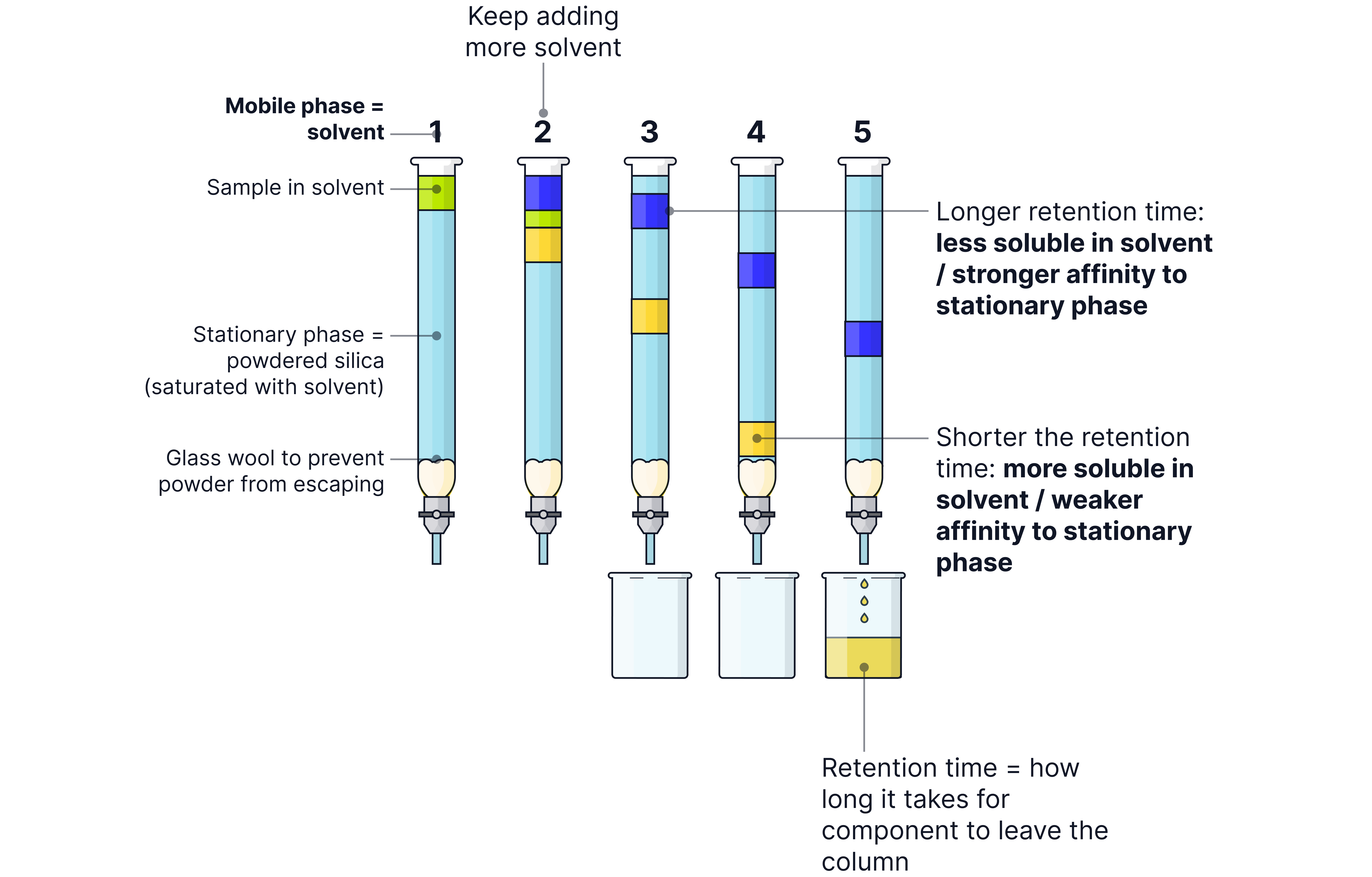
Separation
The sample mixture is placed at the top of the column.
As the solvent runs through, different components of the mixture move down the column at different speeds.
Substances that interact more strongly with the stationary phase (e.g. polar compounds binding to polar silica) move more slowly.
Substances that interact more strongly with the mobile phase dissolve and move more quickly.
This causes the mixture to separate into distinct bands down the column.
Collection and identification
As each band reaches the bottom, it can be collected in separate containers (fractions).
Retention times can also be measured similar to gas chromatography
The identity of the components can then be checked using known standards, melting points, TLC, or spectroscopy.
Practice Questions
Question 1: TLC and Rf Calculation
Triethylamine and propylamine were analysed by TLC. A silica plate was used with heptane as the solvent.
Triethylamine travels , while the solvent front travels .
(a) Calculate the Rf value of triethylamine
(b) Suggest and explain whether the Rf value of triethylamine would increase or decrease if hydrochloric acid was added to the sample
mixture
Answer:
(a)
(b) Decreases – the protonated amines would be less soluble in the non-polar heptane solvent and have a stronger affinity for the polar silica stationary phase
Question 2: A mixture contains several volatile compounds. A GC-MS is used to analyse the sample.
What information does gas chromatography provide?
Answer:
Number of components (from number of peaks), retention time of each component, and relative amounts (from peak area)
Key Tips and Reminders
Rf values are dimensionless and range between 0 and 1.
Compare Rf values to database/known values for identification.
Stronger attraction to stationary phase smaller Rf value/longer retention time
Stronger attraction to mobile phase larger Rf value/shorter retention time