Condensation Polymers
Lajoy Tucker & Dr. Davinder Bhachu
Teachers

Contents
Introduction to Condensation Polymers
Condensation polymers are formed by the reaction between monomers with two or more functional groups, resulting in the elimination of a small molecule, typically water () or hydrogen chloride .
Key Features
Condensation Polymer Types:
Polyesters
Polyamides
Proteins
DNA
Functional Groups:
Ester Link : Found in polyesters.
Amide Link : Found in polyamides.
Condensation Polymerisation Explainer Video
Polyester Formation
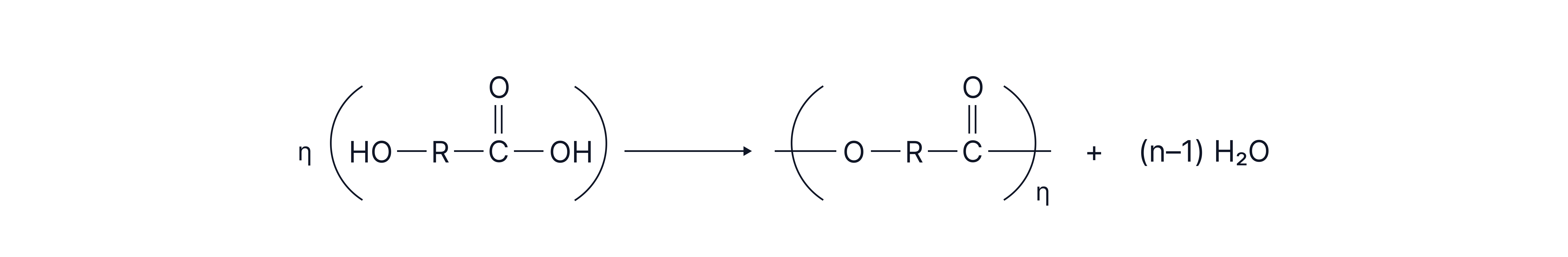
Esters are typically formed from carboxylic acids and alcohols
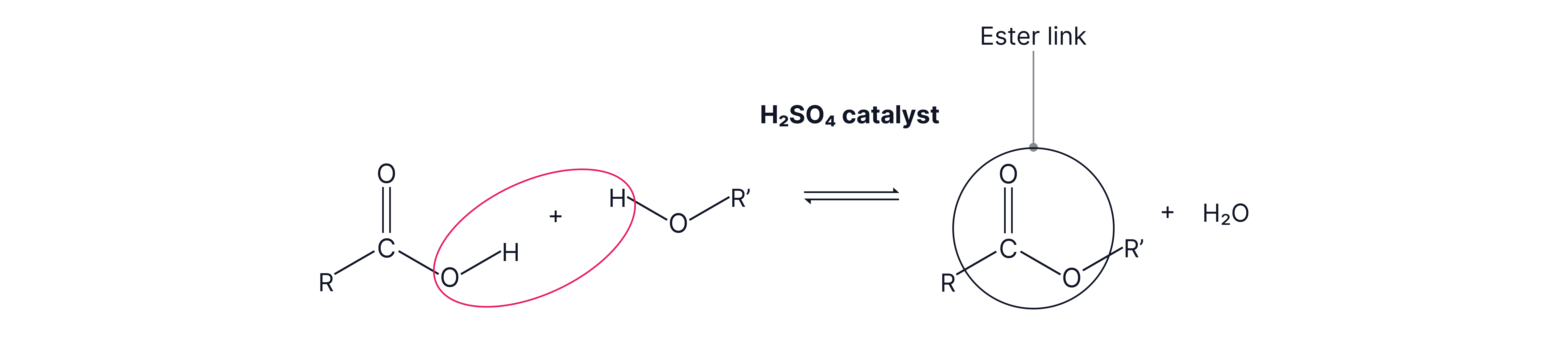
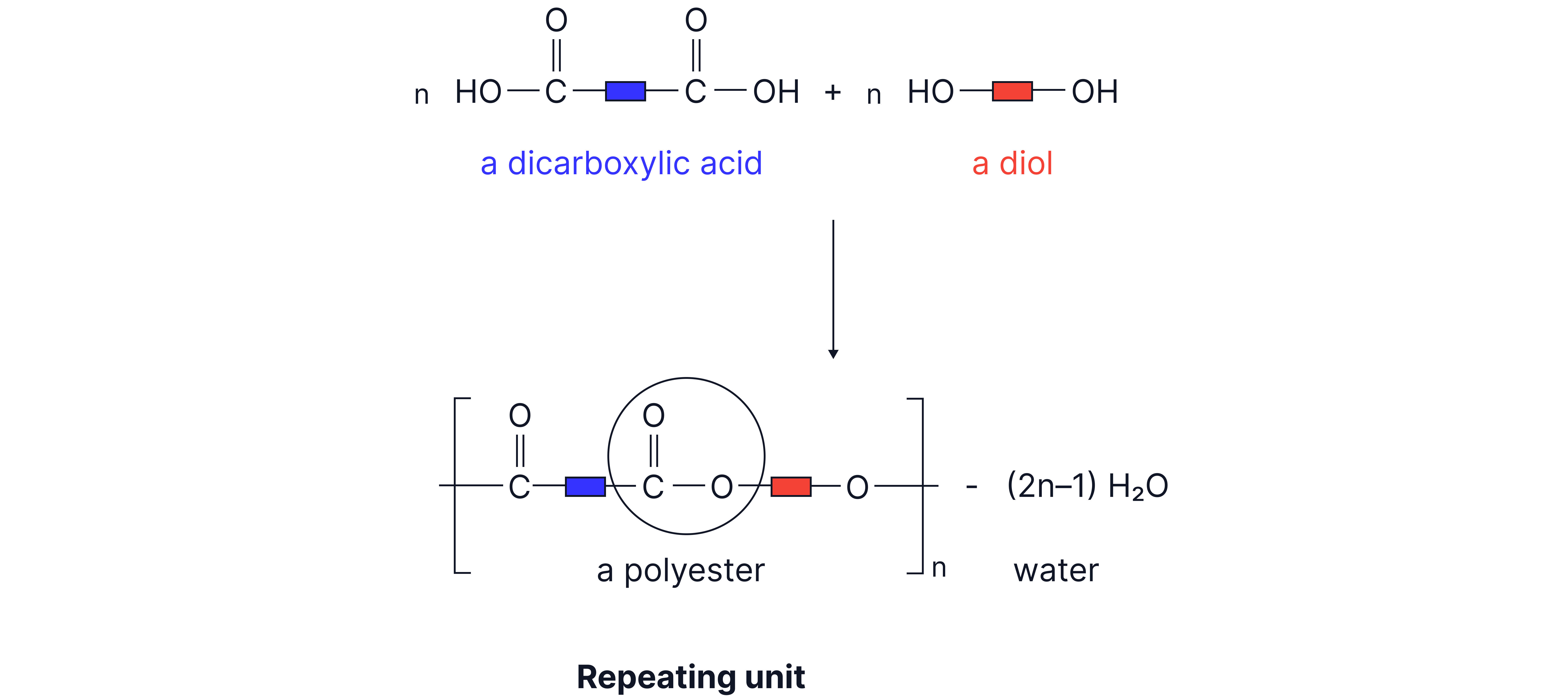
A polyester can be formed where the required functional groups are on both sides of the molecule forming a long chain.
Monomers:
Dicarboxylic Acid: Contains two carboxyl groups .
Diol: Contains two hydroxyl groups .
Example: Terylene (polyethylene terephthalate (PET)) used in textiles and plastic drinks bottles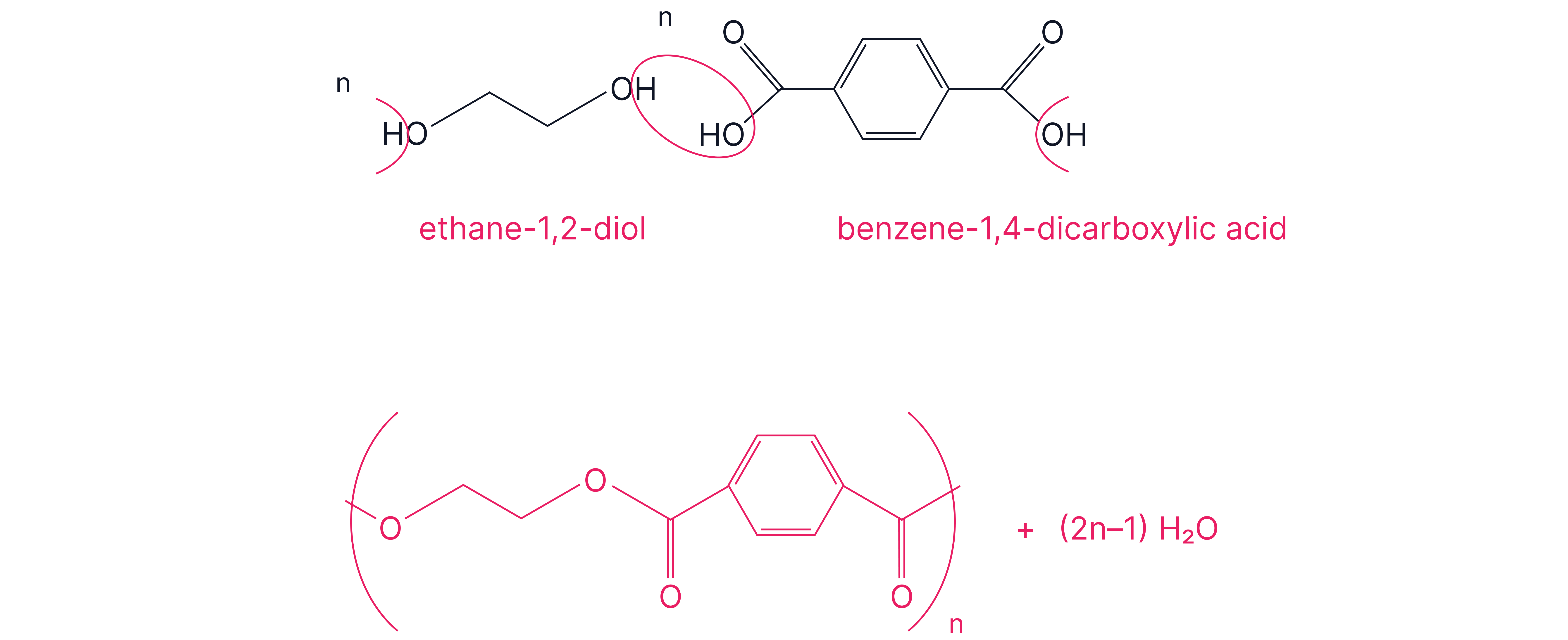
Monomer:
Hydroxycarboxylic acid: Contains one hydroxyl and one carboxyl group
The monomer contains both functional groups required for polyester formation and so only one monomer is needed.
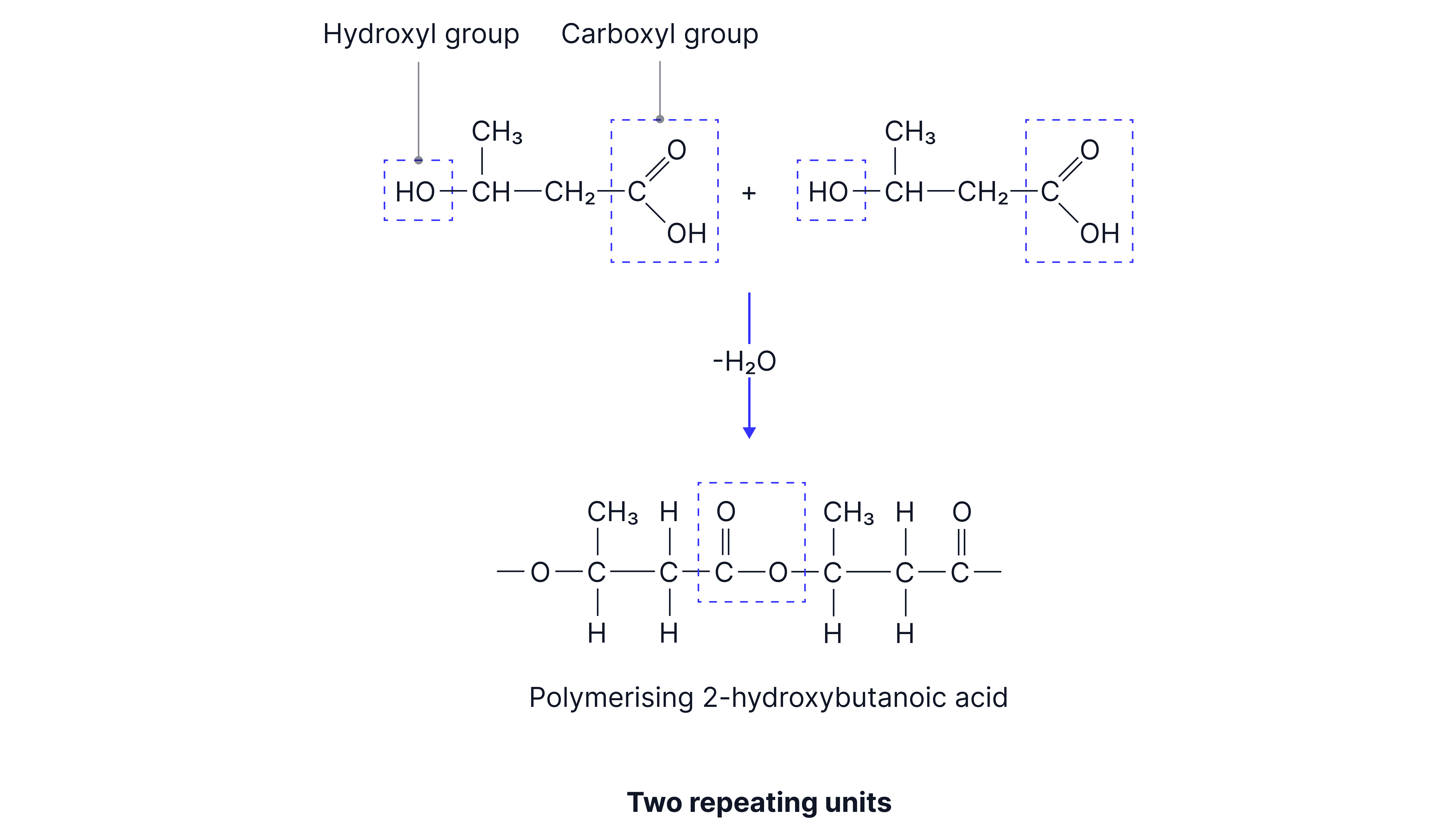
General equation:
Polyamide Formation
Amides can be formed from carboxylic acids (or derivatives such as acyl chlorides and acid anhydrides) and amines.
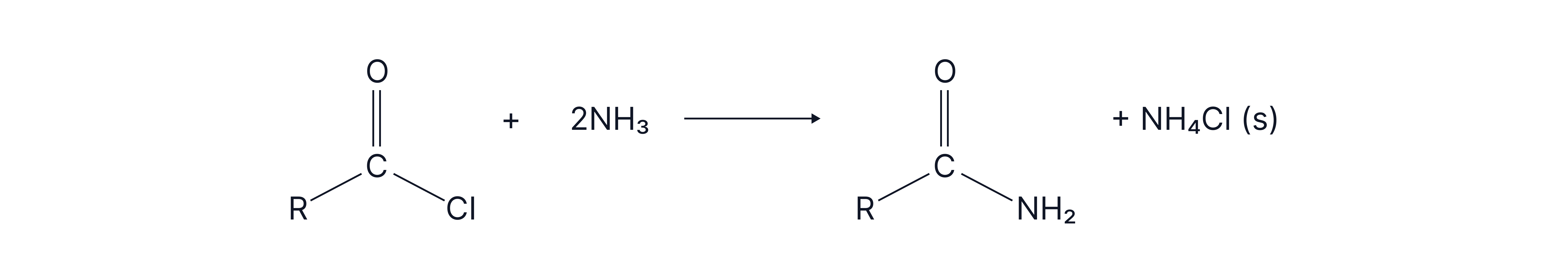
A polyamide can be formed where the required functional groups are on both sides of the molecule forming a long chain.
Monomers:
Diamine: Contains two amine groups .
Dicarboxylic Acid: Contains two carboxyl groups OR
Dioyldichlorides: Containing two acyl chloride groups
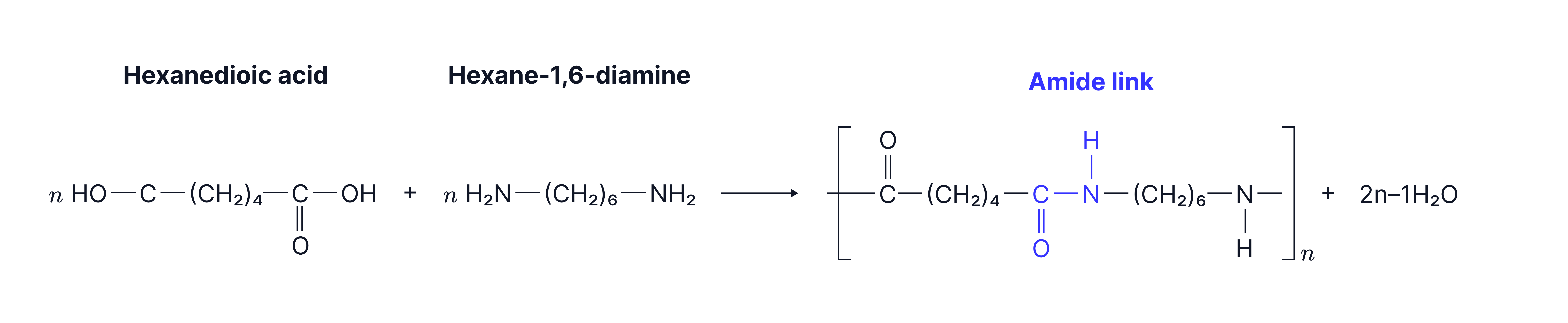
Example 1: Nylon 6,6 used in textiles
Example 2: Kevlar used in bulletproof vests
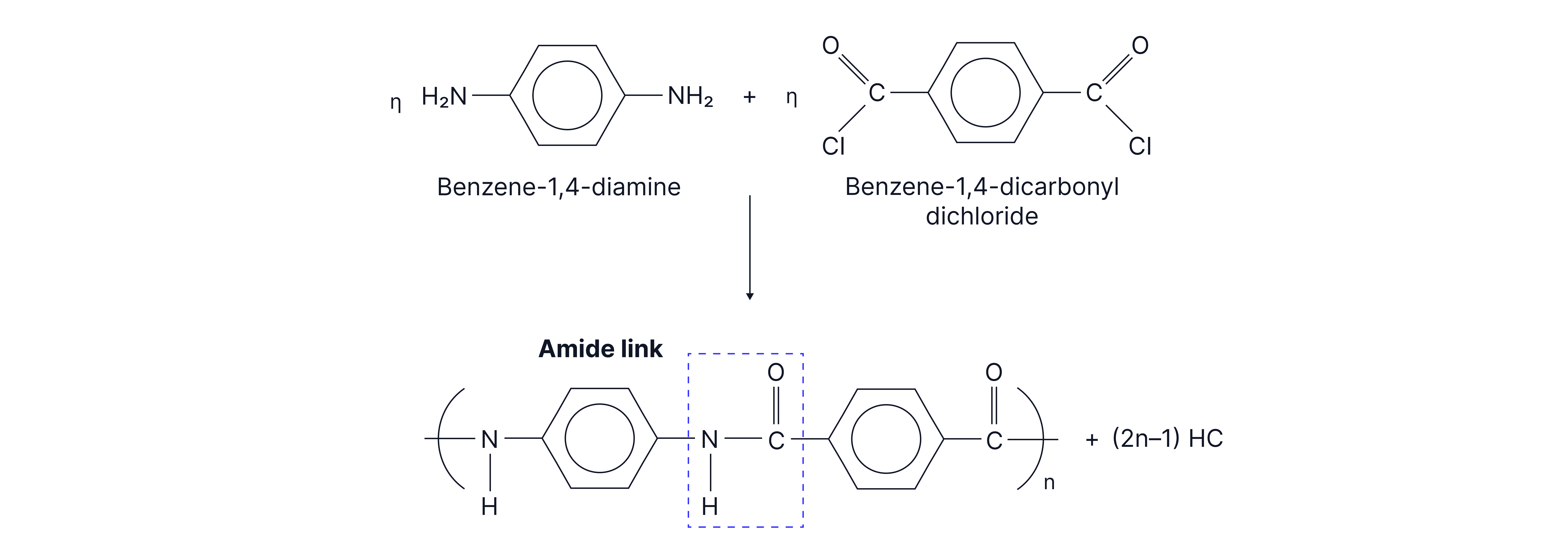
Hydrogen bonds can form between Kevlar polymer chains due to the lone pair on the O of the C=O and the electron deficient H of the N-H.
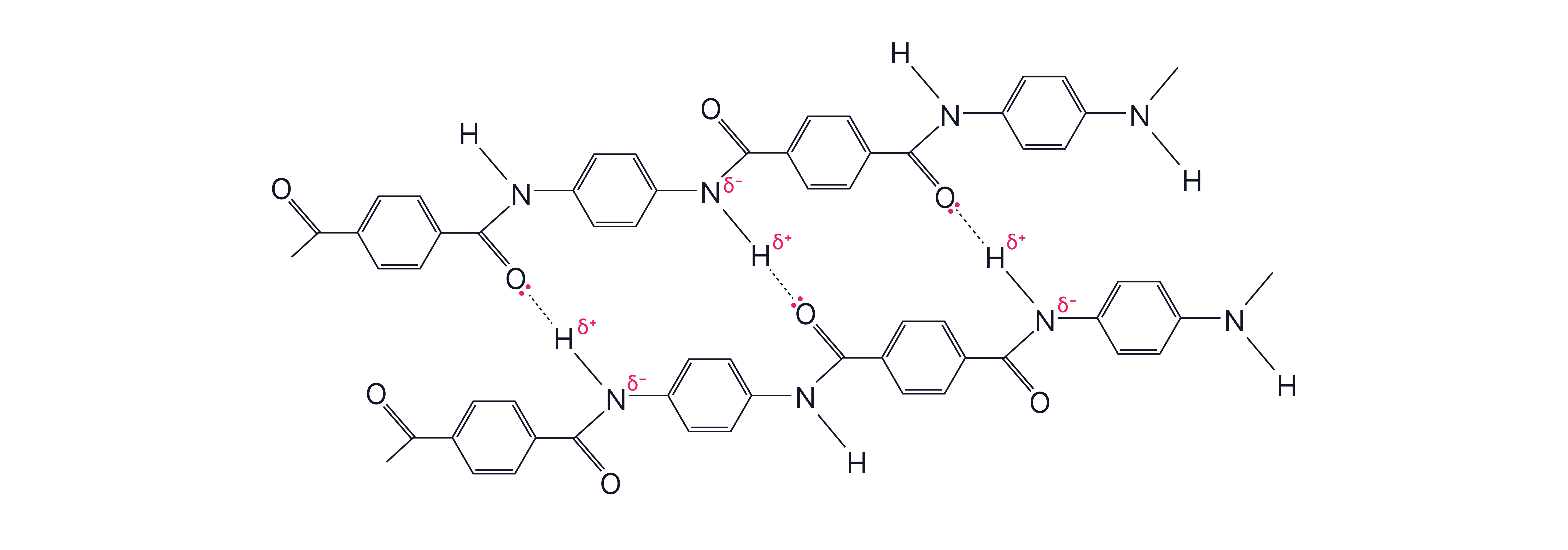
Protein Formation
Proteins are formed from amino acid monomers. Amino acids contain a carboxyl group (-COOH) and amino groups (-NH2) allowing the formation of amide (also named peptide) links in a polypeptide chain.
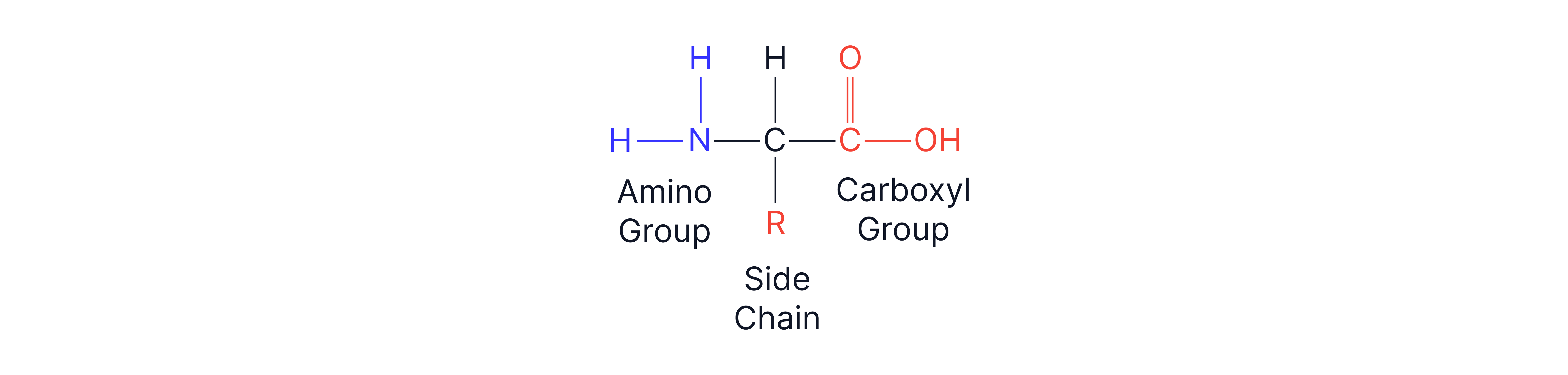
As both functional groups are on the same molecule only one monomer is required.
Two amino acids forming a dipeptide:
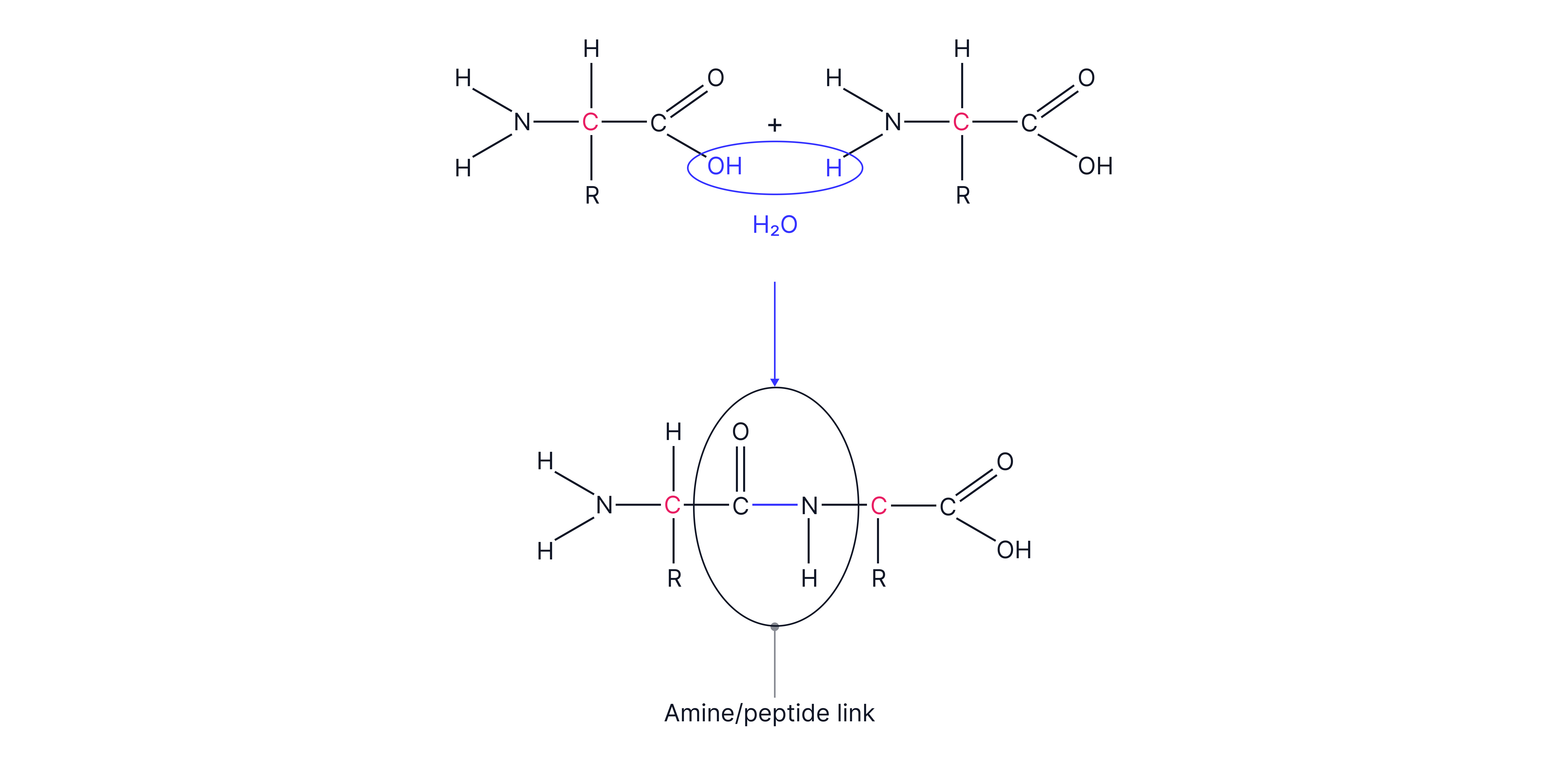
n amino acids forming a polypeptide chain
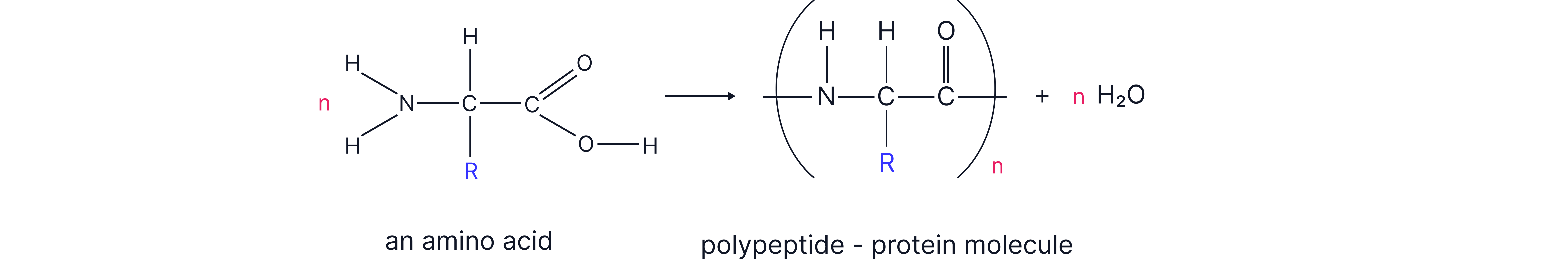
Addition Polymers vs Condensation Polymers
Addition polymers (polyalkenes) form by addition of alkenes.
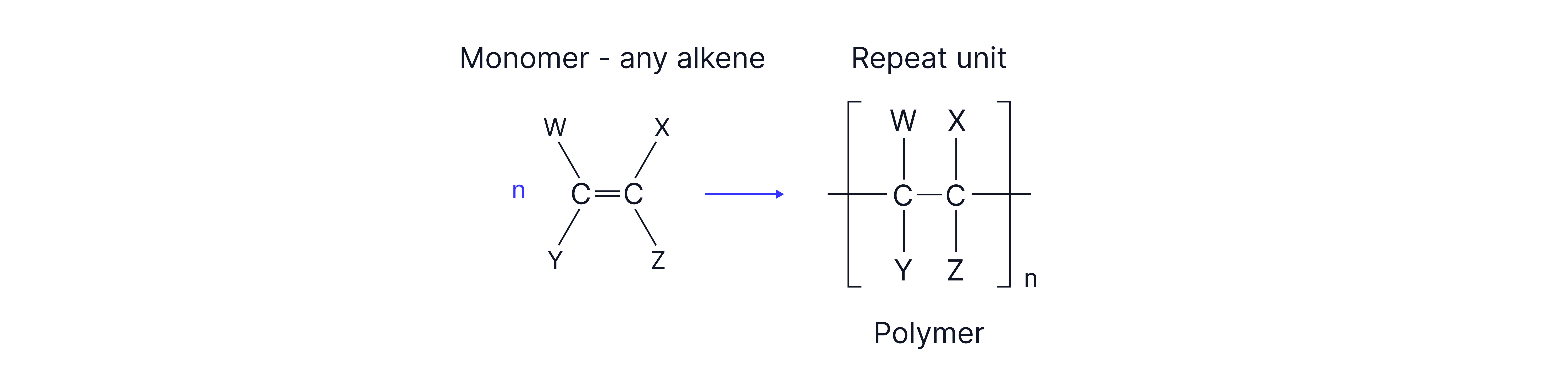
Backbone is C–C only which is non-polar and very unreactive.
C–C bonds are non-polar and there is no easy site for nucleophilic or electrophilic attack.
Polyalkenes, therefore, cannot be broken down naturally (are non-biodegradable).
Condensation polymers (polyesters, polyamides) form when monomers join and a small molecule is eliminated (e.g. ).
They contain polar ester or amide links which can be hydrolysed as the polar C=O allows nucleophilic attack
Therefore, they are typically biodegradable.
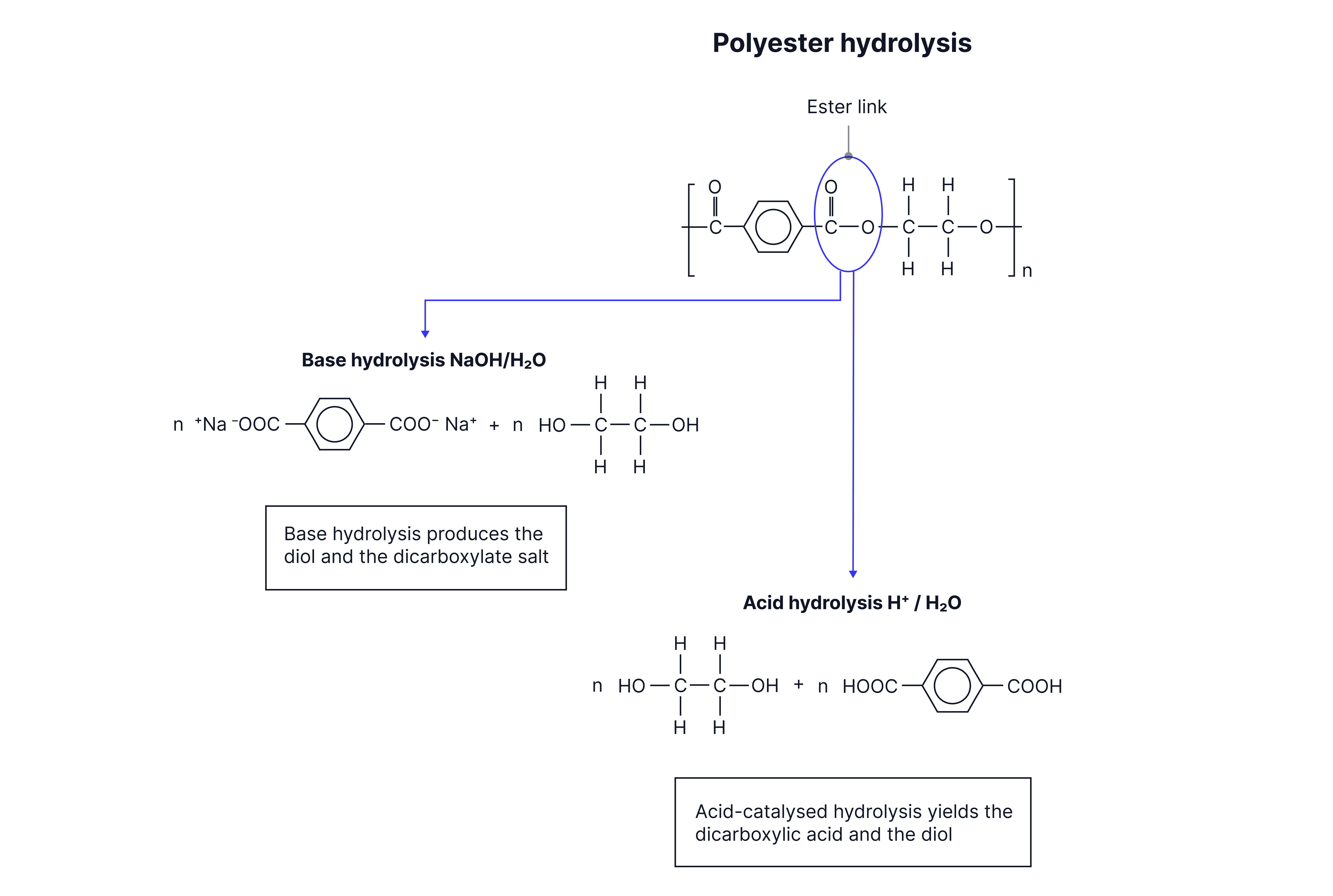
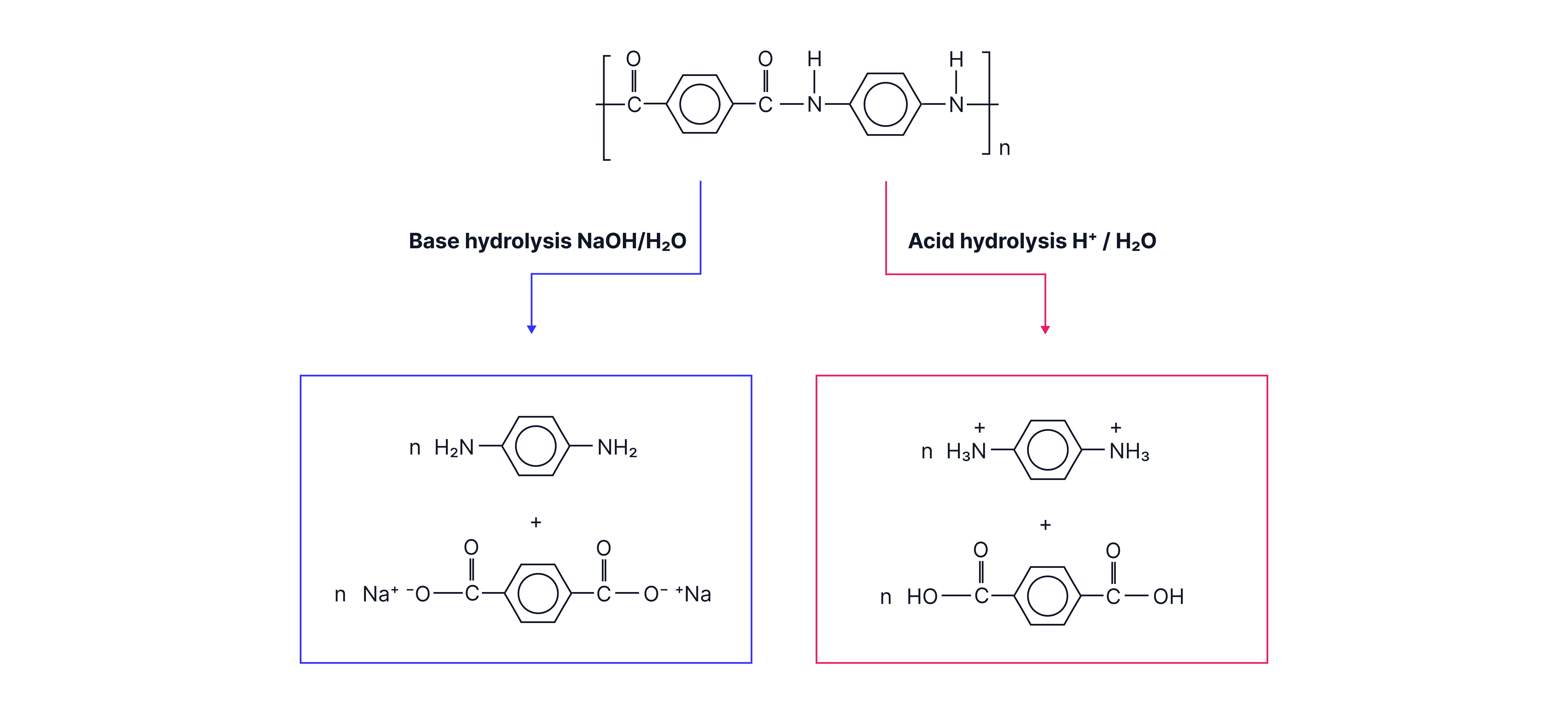
Hydrolysis of condensation polymers
Applying understanding of ester and amide hydrolysis, the products of acid and alkaline hydrolysis of polyesters and polyamides can be determined
Polyester hydrolysis
Polyamide Hydrolysis
Disposal of Polymers
Method | Advantages | Disadvantages |
|---|---|---|
Landfill | • Site is contained • Condensation polymers will biodegrade | • Degradation is very slow • Emissions of methane • Toxic compounds may leach into earth • Land availability is limited |
Recycling | • Reduces waste persisting in landfill • Conserves finite resources | • Energy and expense required for collection and processing • Limited number of remoulding cycles before quality loss |
Incineration (burning) | • Reduces waste persisting in landfill • Generates electricity from burning polymers | • Releases carbon dioxide (greenhouse gas) • Produces toxic gases into the atmosphere |
Summary of Applications of Condensation Polymers
Polyesters:
Poly(ethylene terephthalate) (PET): Used in the manufacture of plastic bottles, clothing fibres, and films.
Polyamides:
Nylon-6,6: Used in textiles, ropes, and carpets.
Kevlar®: Used in bulletproof vests and protective clothing due to its high strength