Group 2
Lajoy Tucker
Teacher

Contents
Group 2 elements are known as the alkaline earth metals.
They include: Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba).
They all have two electrons in their outer s-subshell (configuration ends in ns² ).
Element | Symbol | Electron Configuration |
|---|---|---|
Magnesium | Mg | 1s² 2s² 2p⁶ 3s² |
Calcium | Ca | 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² |
Strontium | Sr | 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² |
Barium | Ba | 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 5d¹⁰ 6s² |
They form ions by losing two electrons. They are oxidised themselves and therefore act as reducing agents.
Definitions:
First ionisation energy: The energy required to remove one mole of electrons from one mole of gaseous atoms.
Atomic radius: The average distance between the nucleus of an atom and its outermost electron shell.
Group 2 metals show clear trends down the group:
Increasing atomic radius.
Decreasing first ionisation energy.
Decreasing melting point (with some anomalies).
Increasing reactivity with water.
An increase in the solubility of hydroxides.
A decrease in the solubility of sulfates.
Explanation of Group 2 Trends
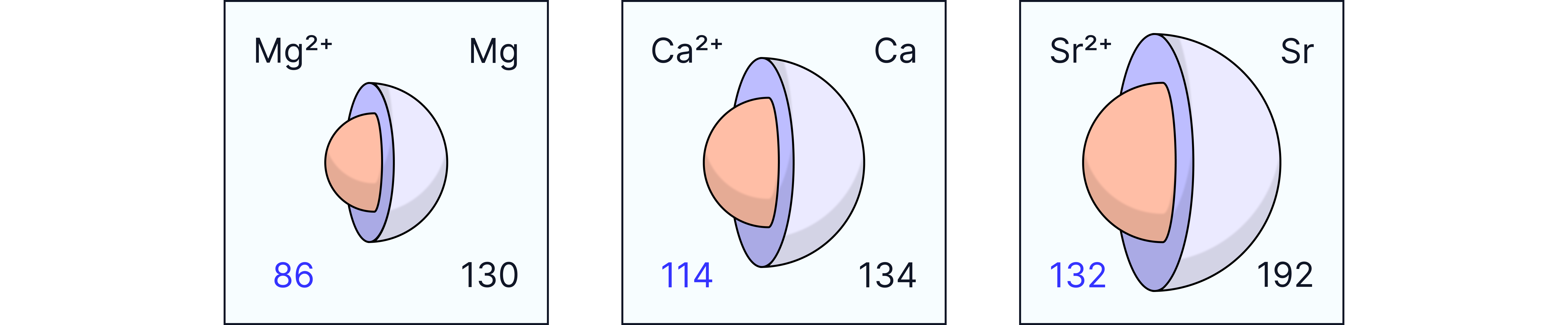
Atomic Radius
Atomic radius increases down the group:
Each successive element has an extra electron shell.
A greater distance between the nucleus and the outer electrons.
More shielding of nuclear charge.
First Ionisation Energy
Decreases down the group because:
Increased atomic radius.
Increased shielding.
Weaker electrostatic attraction between the nucleus and the outermost electrons.
Trends in Melting Points
Generally decreases down the group.
Metallic bonding weakens as ion size increases.
This results in a larger distance between the charged metal ion and delocalised electrons, and thus a weaker attractive force.
Reactions with Water
Group 2 metals react with water to form a metal hydroxide and hydrogen:
(M = Group 2 element)
Notes:
Magnesium reacts very slowly with water, but much faster with steam.
For Mg and steam:
Note – for the reaction between magnesium and steam, state symbols are typically expected.
The table below highlights the reactions of group 2 elements with water:
Element | Reaction | Equation | Observation |
|---|---|---|---|
Mg (cold water) | Slow | Mg + 2H₂O → Mg(OH)₂ + H₂ | Few bubbles |
Mg (steam) | Fast | Mg + H₂O (g) → MgO + H₂ | White solid, bright light |
Ca | Moderate | Ca + 2H₂O → Ca(OH)₂ + H₂ | Effervescence, white ppt |
Sr | Fast | Sr + 2H₂O → Sr(OH)₂ + H₂ | More vigorous |
Ba | Very fast | Ba + 2H₂O → Ba(OH)₂ + H₂ | Rapid bubbling |
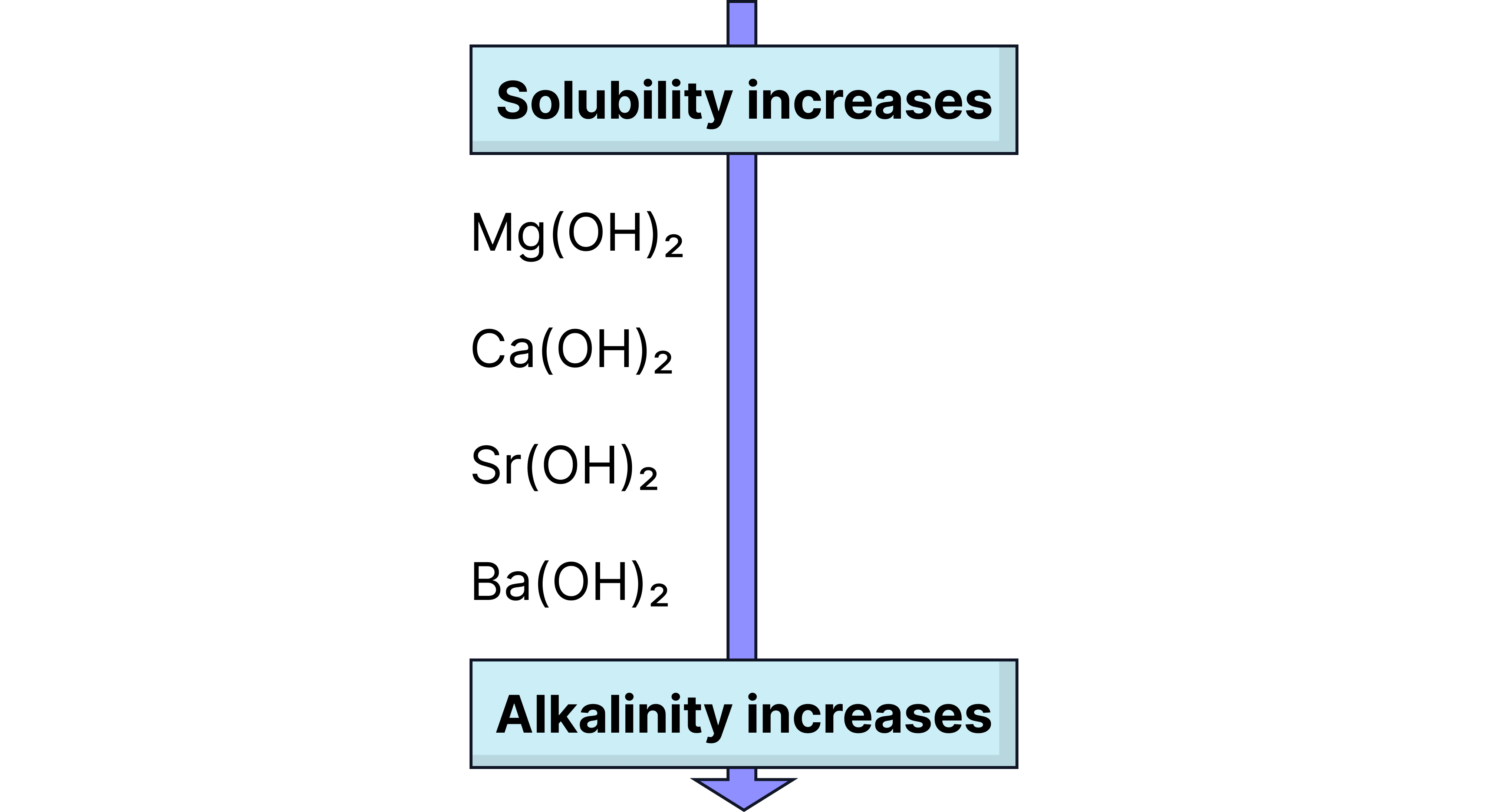
Solubility of Group 2 Hydroxides
Solubility increases down the group:
: sparingly soluble
: slightly soluble
and : increasingly soluble
Uses of Group 2 hydroxides
: Used as an antacid to neutralise stomach acid.
: Used in agriculture to neutralise acidic soils.
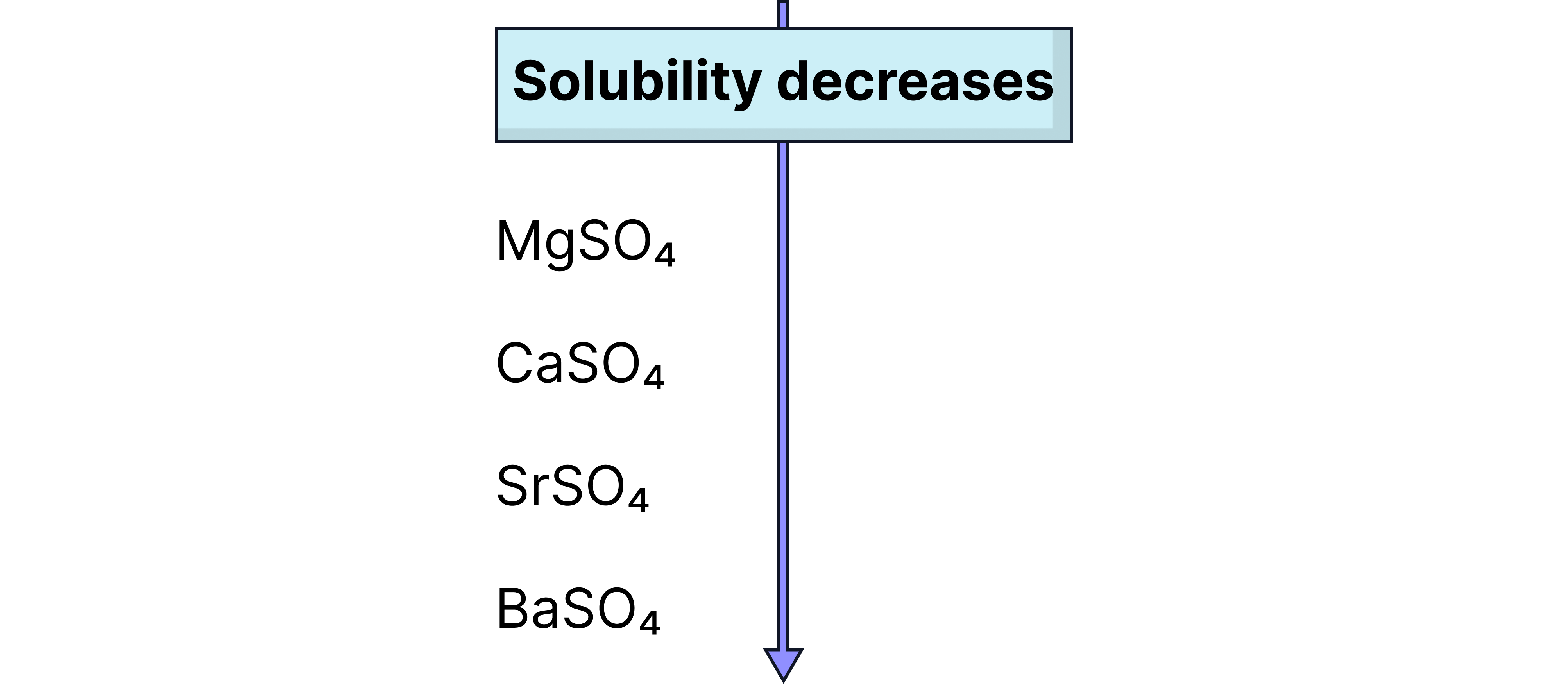
Solubility of Group 2 Sulfates
Decreases down the group:
: Soluble
: Slightly soluble
: Insoluble
Uses
Barium sulfate, being insoluble, makes it useful in medicine as X-ray ‘barium meals’. It lines the digestive tract and absorbs X-ray radiation allowing outlines of organs to be visualised. Although ions are toxic, as barium sulfate is insoluble, the ions are not absorbed into the blood
Barium chloride is used as a test for sulfate ions as it reacts to form barium sulfate, a white precipitate.
Uses and Reactions Involving Group 2 Metals
Titanium is extracted from TiCl₄ using magnesium:
Equation:
This is a redox reaction in which Mg reduces Ti⁴⁺ to Ti. The Mg is oxidised from 0 to +2.
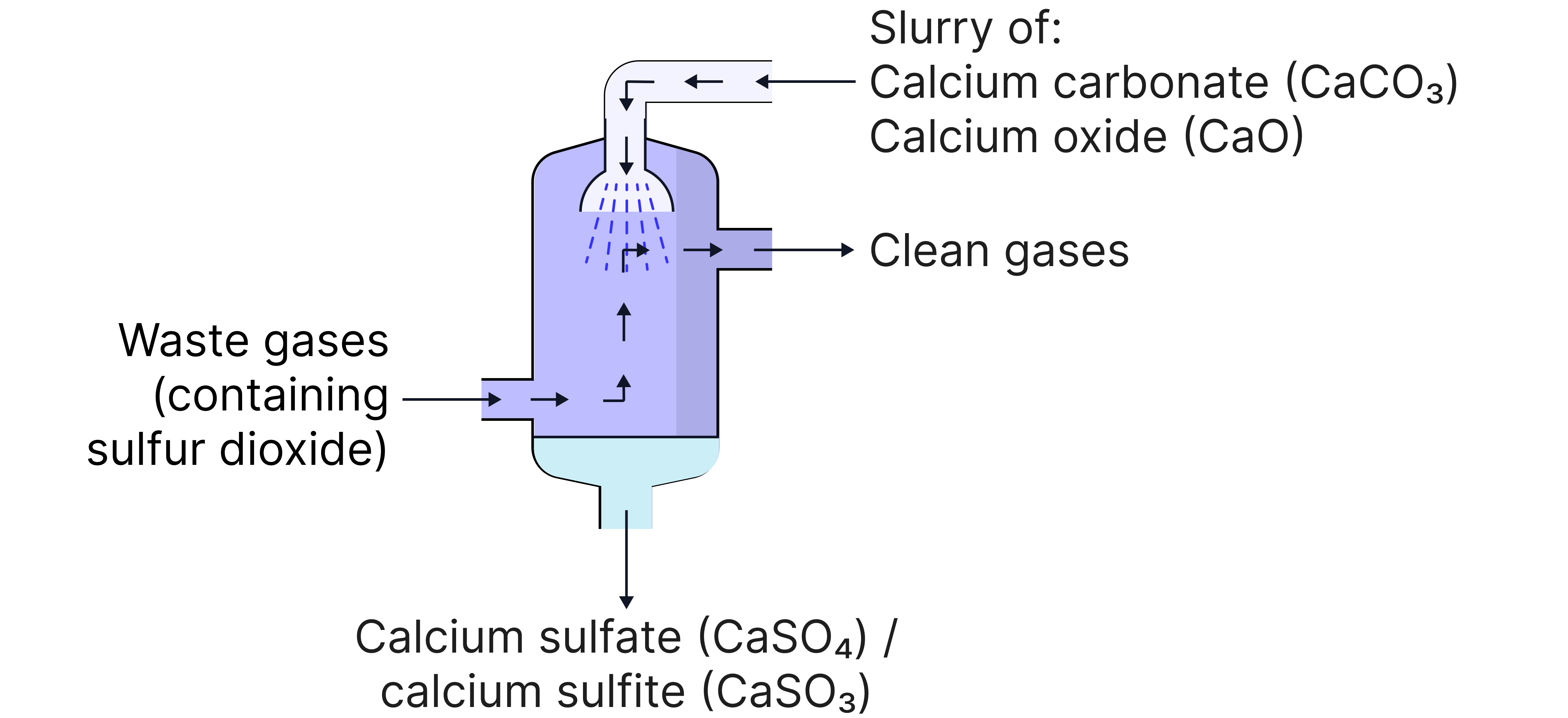
Removing SO₂ from Flue Gases:
SO₂ released when coal with sulfur impurities is burned in power stations.
If SO₂ is released into the atmosphere, it will dissolve in clouds/rainwater producing acid rain
CaO or CaCO₃ is used to remove the in the desulfurisation of flue gases in power stations
Example equations:
CaO + SO₂ → CaSO₃
CaCO₃ + SO₂ → CaSO₃ + CO₂
Worked Examples
Q: Write the ionic equation for the reaction between solutions of BaCl₂ with Na₂SO₄. Include state symbols
A:
Q: Explain why Mg(OH)₂ is used as an antacid rather than Ba(OH)₂.
A: Mg(OH)₂ is less soluble than Ba(OH)₂ so it releases fewer OH⁻ ions, making it gentle and safe for neutralising stomach acid.