Nucleophilic Addition to Carbonyls
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Nucleophilic Addition: A reaction where a nucleophile attacks an electrophilic carbon atom, breaking a π bond (typically in ) and forming a new bond.
Carbonyl Compounds: Molecules containing a polar bond, where the more electronegative oxygen atom leaves the carbon atom slightly electron deficient and leaving it susceptible to nucleophilic attack. e.g. aldehydes and ketones.

Key Reactions:
Reduction of carbonyls to alcohols using sodium borohydride
Formation of hydroxynitriles using hydrogen cyanide or potassium cyanide and dilute acid
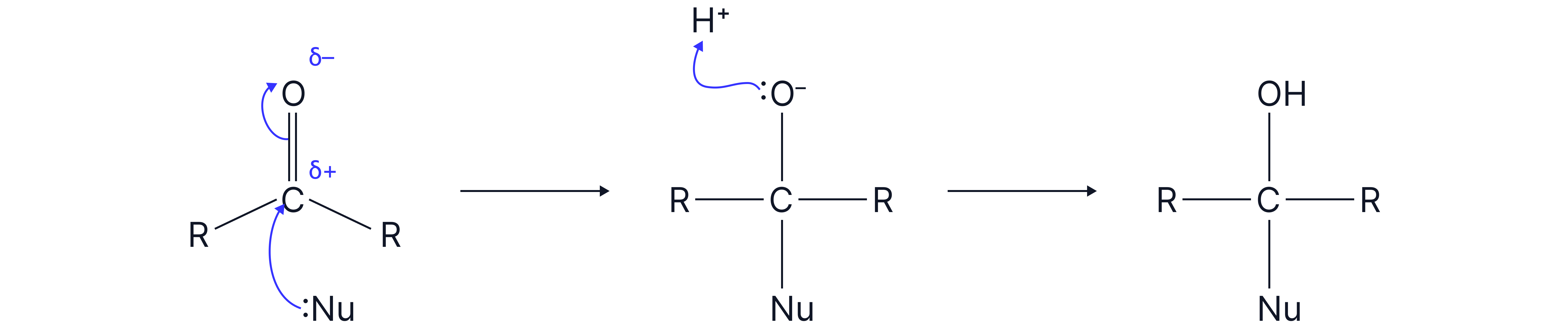
Nucleophilic Addition Explained
General Mechanism
The carbon in the bond is electrophilic , attracting nucleophiles.
A nucleophile (electron pair donor) attacks the electron deficient carbon atom resulting the the π bond breaking.
A lone pair on the now negatively charged O atom is protonated (accepts an ion)
Key Examples
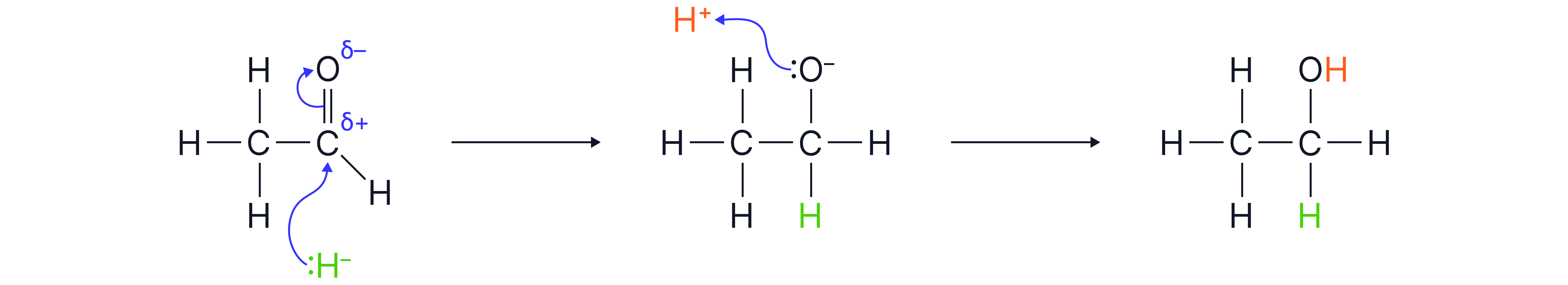
Reduction to Alcohols
Aldehydes are reduced to primary alcohols, and ketones are reduced to secondary alcohols.
Reagent: Sodium borohydride in aqueous or alcoholic solution.
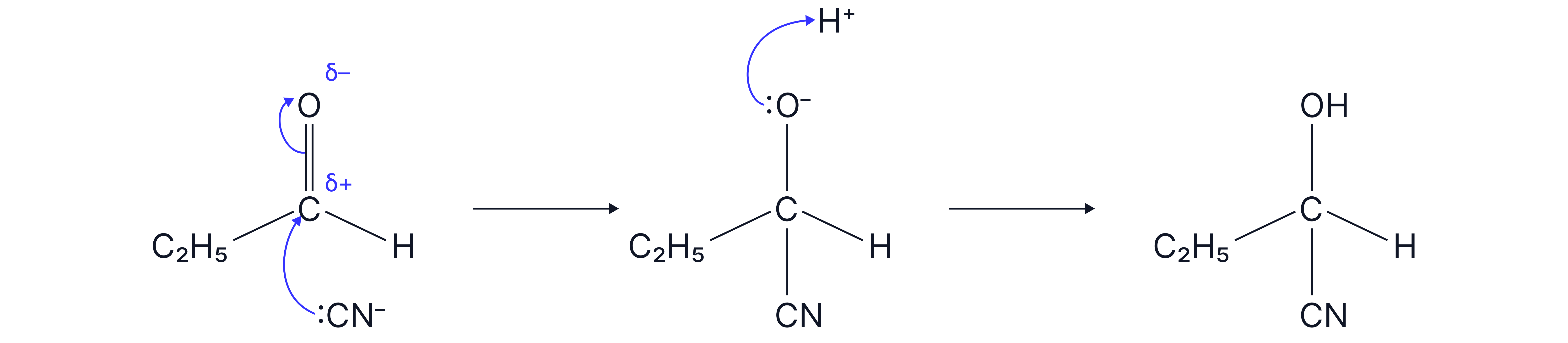
Mechanism
Hydride ion attacks δ⁺ carbon atom of the .
Formation of a tetrahedral intermediate with
Protonation by water or dilute acid to form the alcohol.
Example: Ethanol to ethanol.
Equation:
The reducing agent is not included in the equation and is instead represented as
General Equations:
For an aldehyde:
For a ketone:
and represents alkyl group.
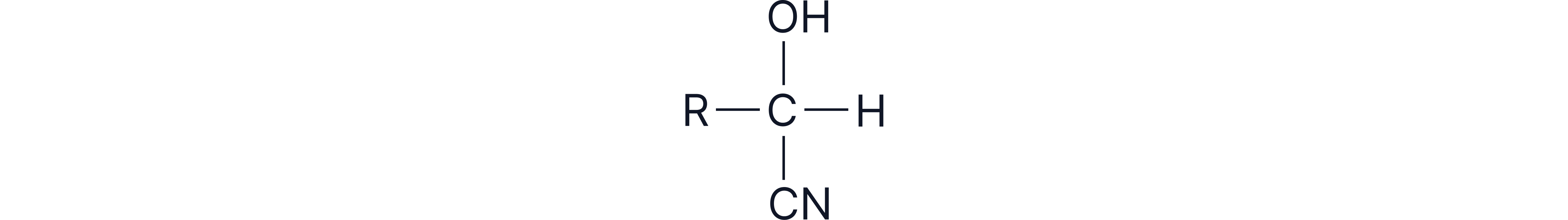
Formation of Hydroxynitriles
Hydroxynitrile general formula:
Reagent: Potassium cyanide followed by dilute acid hydrogen cyanide
The cyanide ion acts as the nucleophile due to a lone pair of electrons on the .

This reaction is often used to extend the length of the carbon chain which is useful for synthesis.
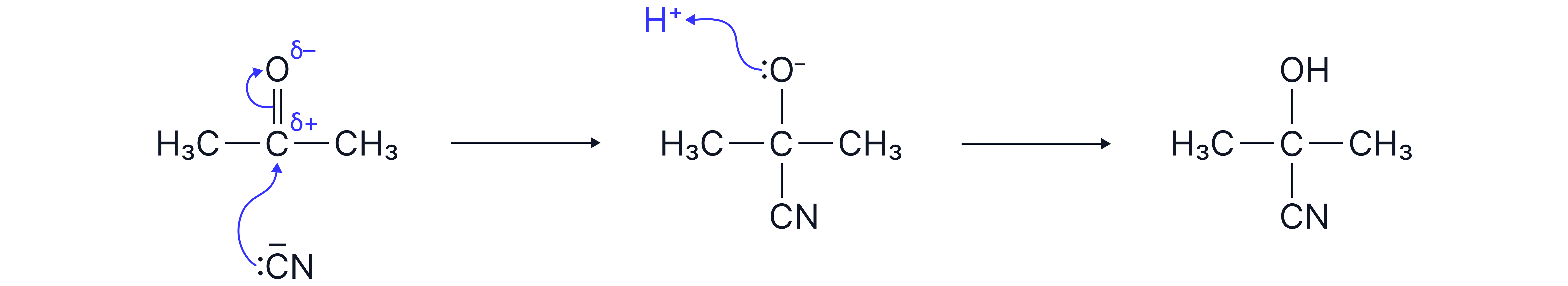
Mechanism
Cyanide ion attacks the carbon atom of the
Formation of tetrahedral intermediate with
Protonation by dilute acid to form the hydroxynitrile.
e.g Propanone to 2-hydroxy-2-methylpropanenitrile.
Equation Example:
For an aldehyde:
For a ketone:
Choice of Reagent
Potassium cyanide followed by acid is generally preferred over direct reaction with hydrogen cyanide because:
is highly toxic.
is a weak acid and so only partially dissociates into and ions. This results in a low concentration of the cyanide ions required for this reaction to take place.
Optical Isomerism
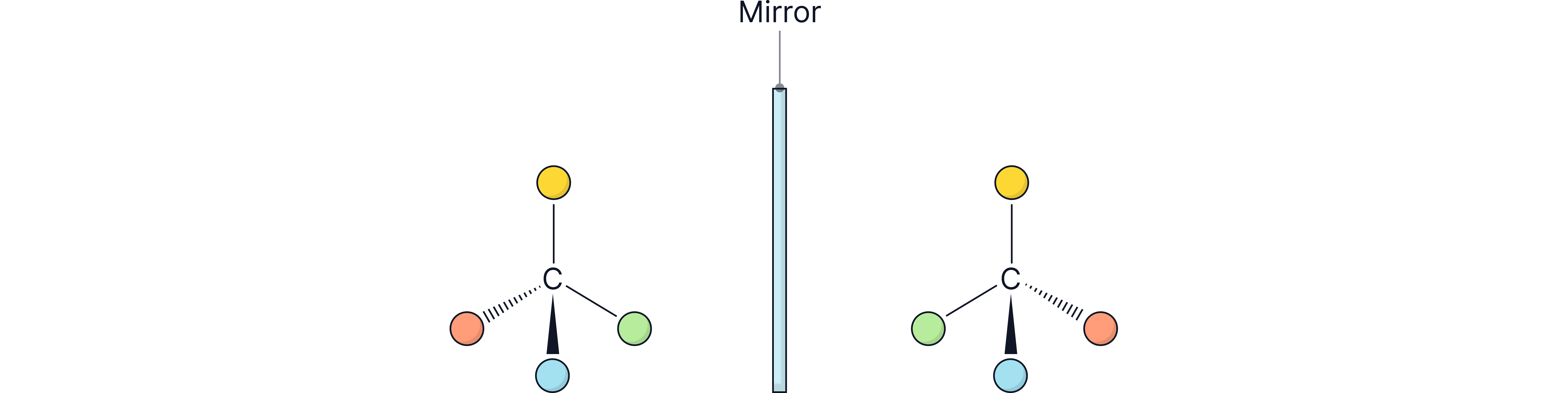
Optical isomerism arises when a compound has a carbon atom with four different groups attached to it (a chiral carbon centre).
Optical isomers (enantiomers) are non-superimposable mirror images (see `Optical Isomerism` note)
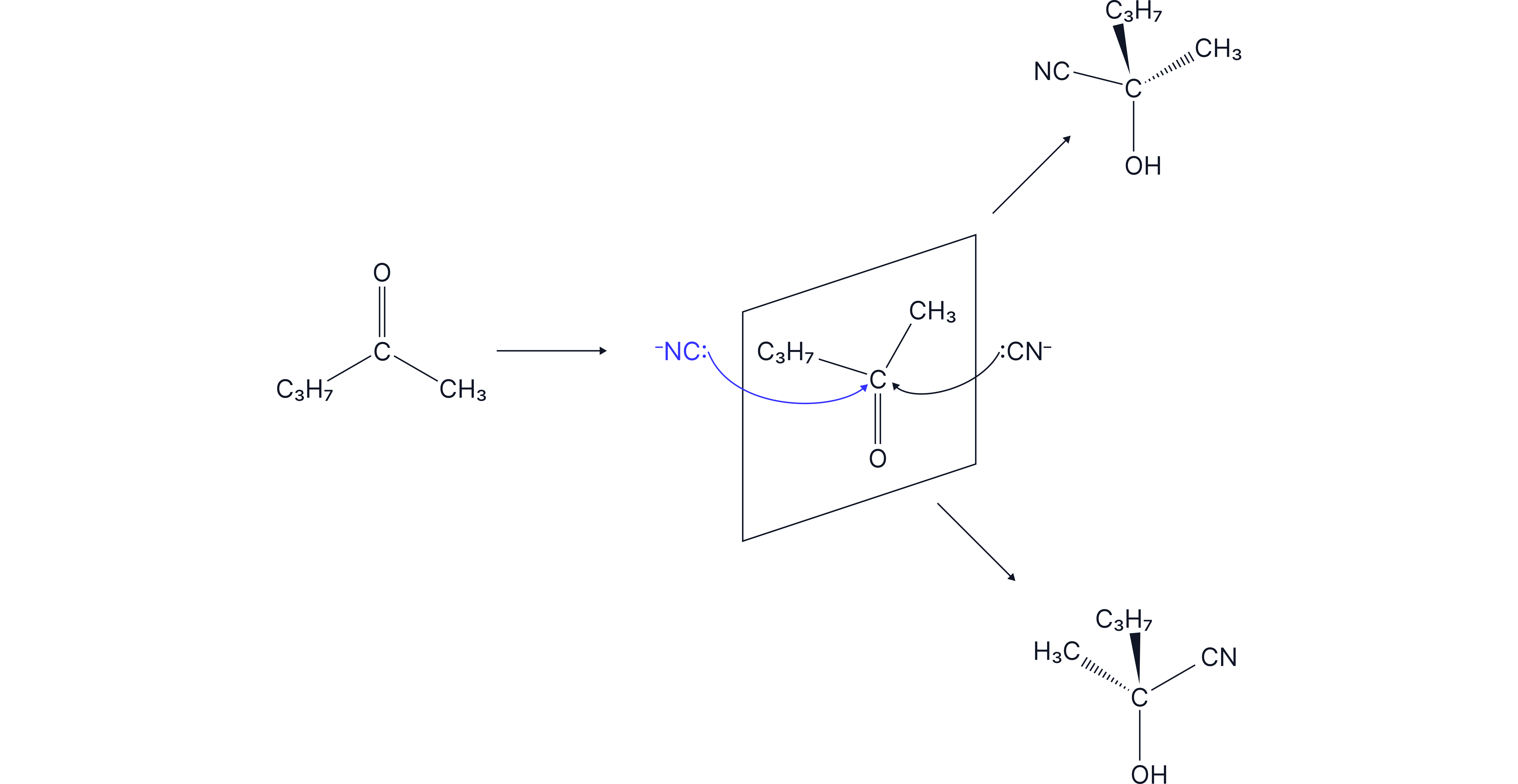
Nucleophilic addition reactions with carbonyl compounds can often lead to the formation of a racemic mixture where a 50/50 mixture of both optical isomers (enantiomers) are formed.
These mixtures do not have an effect on plane polarised light as the enantiomers rotate it in equal and opposite directions (cancelling the effects out).
Forming racemic mixtures
Racemic mixtures are formed as:
The carbonyl group is planar.
Nucleophilic attack can occur from either above or below the plane.
There is equal probability of attack from both sides which leads to the formation of a racemic mixture (50:50 mixture of enantiomers).
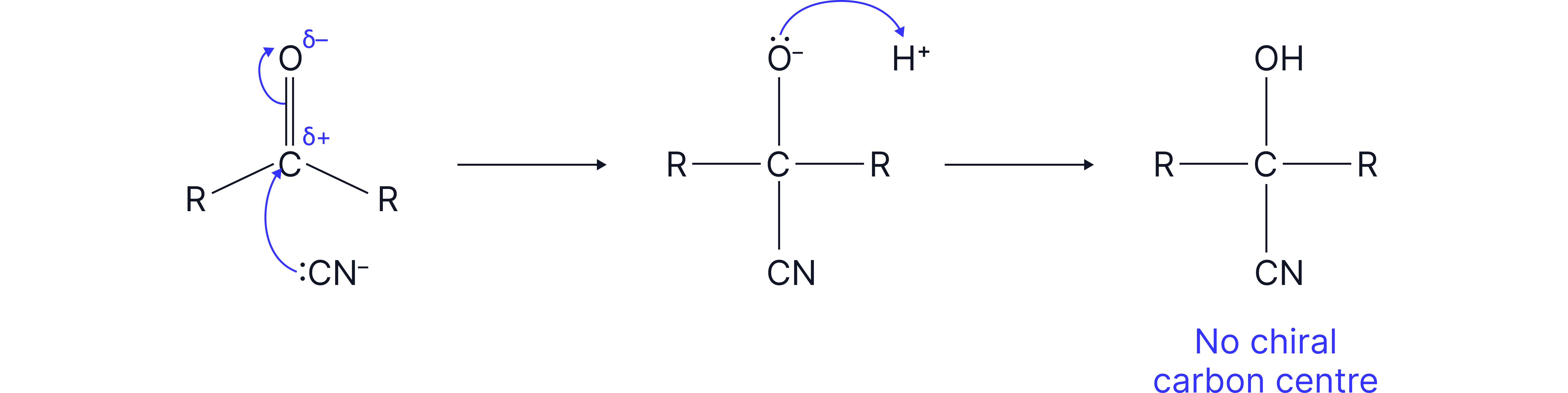
Note: Racemic mixtures are only formed when using an aldehyde or an unsymmetrical ketone. Symmetrical ketones would result in products where two of the groups attached to the carbon atom are the same and therefore would not have a chiral carbon centre.
Example: if both R groups are the same.
Practice Questions
Question 1:
Name and outline the mechanism for the reaction of propanal with .
Answer:
Nucleophilic Addition.
Question 2
Why does reaction of butanone with produce a racemic mixture?
Answer:
The carbonyl group is planar.
There is equal probability of attack by from above or below the plane.
Both enantiomers are formed in equal amounts.
Key Tips and Reminders
Always show curly arrows, charges, and lone pairs clearly in mechanisms.
Specify "attack above or below the plane" when explaining racemic mixture formation.
Racemic mixtures are only formed when the original carbonyl compound is unsymmetrical.