Optical Isomerism
Lajoy Tucker
Teacher

Contents
Optical Isomerism
Optical isomers are seen in carbon molecules with 4 different groups of atoms attached to the central carbon atom. When the central atom is carbon, the carbon is called an asymettric/chiral carbon, and the molecule is optically active.
Here are some examples of chiral carbon molecules.

Optical isomers exist as two isomers, known as enantiomers.
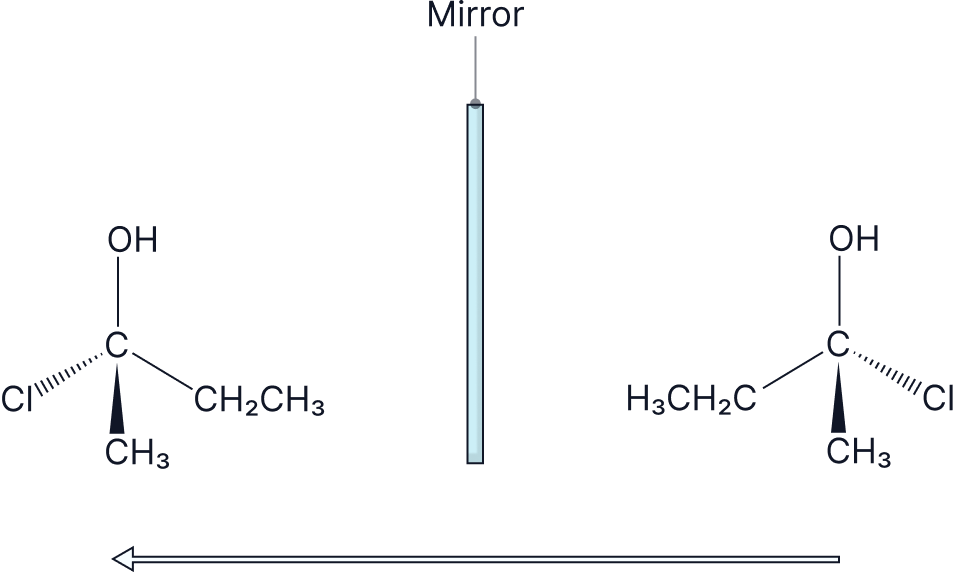
These are non-superimposable mirror images of one another, and they are distinguished by R/S, +/-, or D/L.
When drawing enantiomers, there are two methods that you can use:
1. Swap two groups
Swapping the orangeand red groups around.

2. Draw a mirror and directly draw the mirror image.
Draw the molecules as if there is a mirror in between them.
A mixture that contains equal amounts of each enantiomer is called a racemic mixture or a racemate and it is optically inactive.
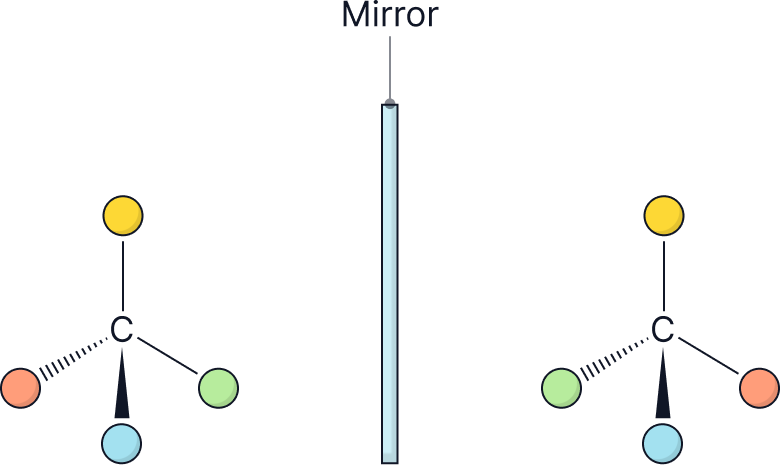
Enantiomers have the same physical and chemical properties. However, there are two exceptions to this statement.
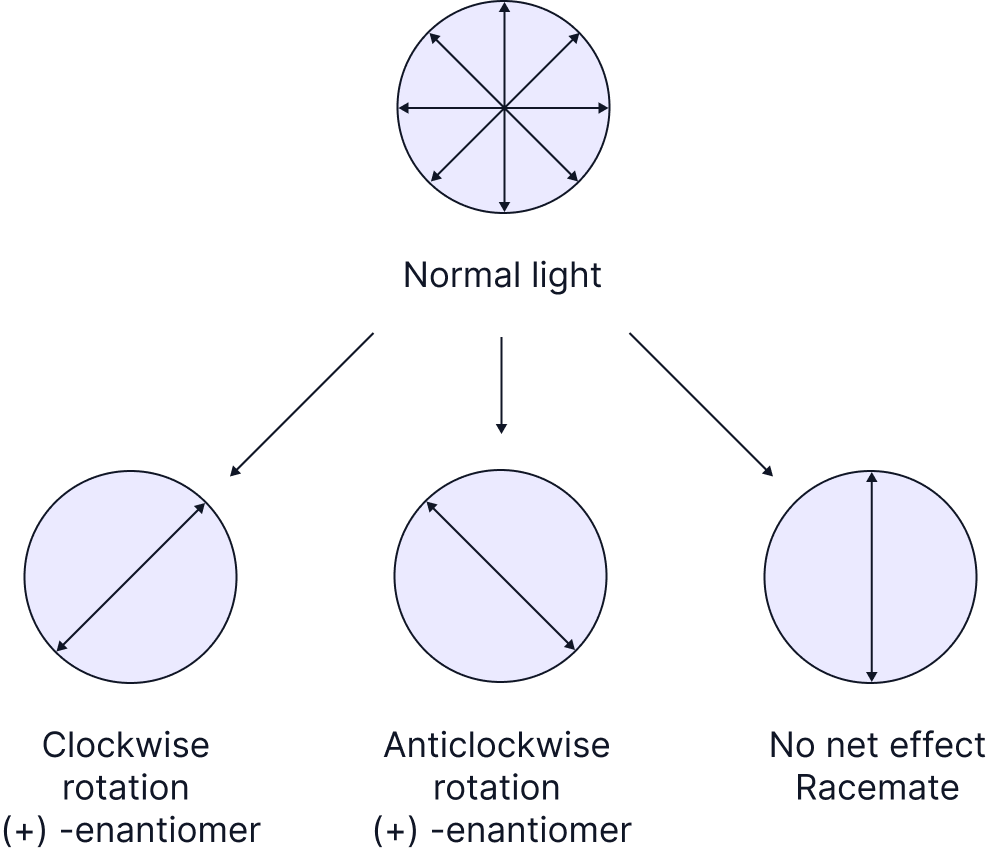
One exception is the effect that each enantiomer has on plane polarised light. One enantiomer will cause a clockwise rotation (+) of plane polarised light, and the other enantiomer will have an anticlockwise rotation (-) of plane polarised light. They will rotate in opposite directions but by the exact same amount.
A racemic mixture (or racemate) is a 1:1 mixture of the two enantiomers meaning it will have no overall effect on plane polarized light.
Another exception is the reaction that each enantiomer will have with other chiral molecules.
Question 1:
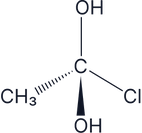
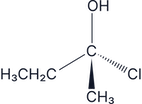
Which of the following molecules are chiral:
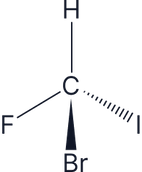
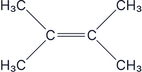
Answer:
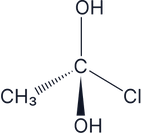
There are two –OH groups so this is an achiral molecule, therefore it does not have optical isomers
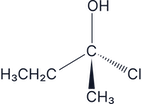
There are two –H groups so this is an achiral molecule, therefore it does not have optical isomers
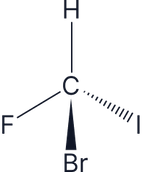
All four groups are different around the carbon centre, so this is a chiral molecule, therefore it has optical isomers
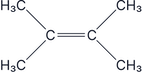
All carbons have the same group on them, so this is an achiral molecule, therefore it does not have optical isomers.
Question 2:
Draw the enantiomers of butan-2-ol and 1-chloro-3-methylpentane.
Answer:
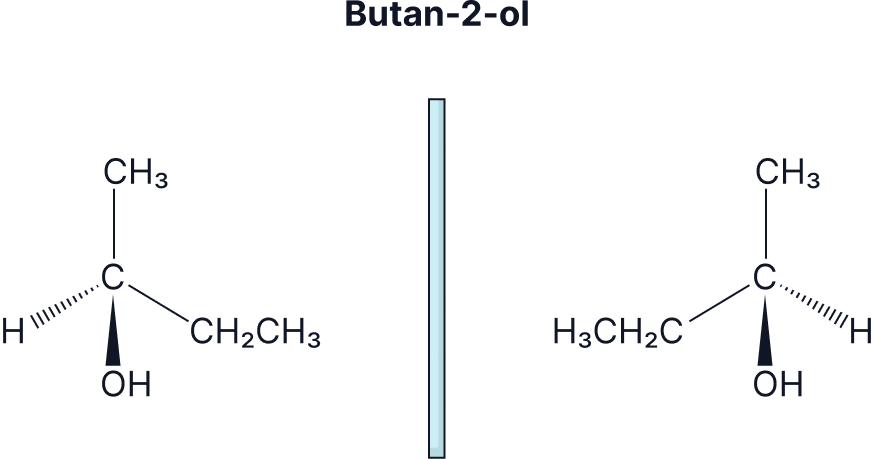
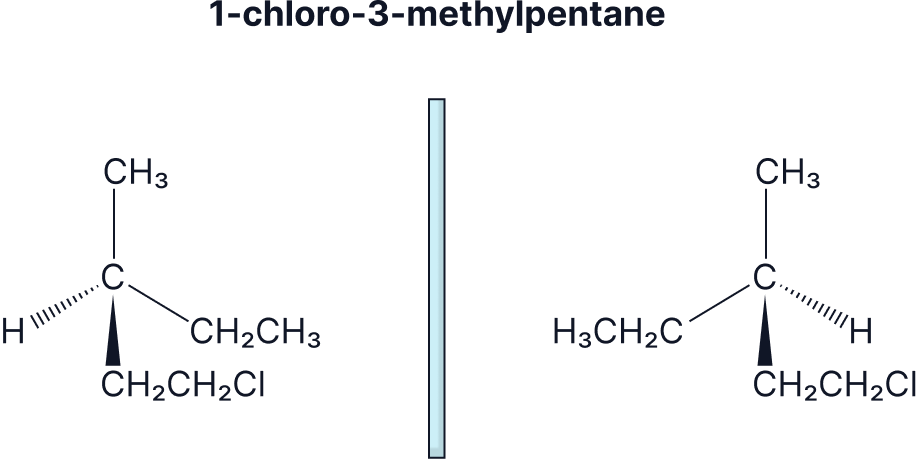
Stereoisomerism Practice Question Explainer Video
Producing racemic mixtures
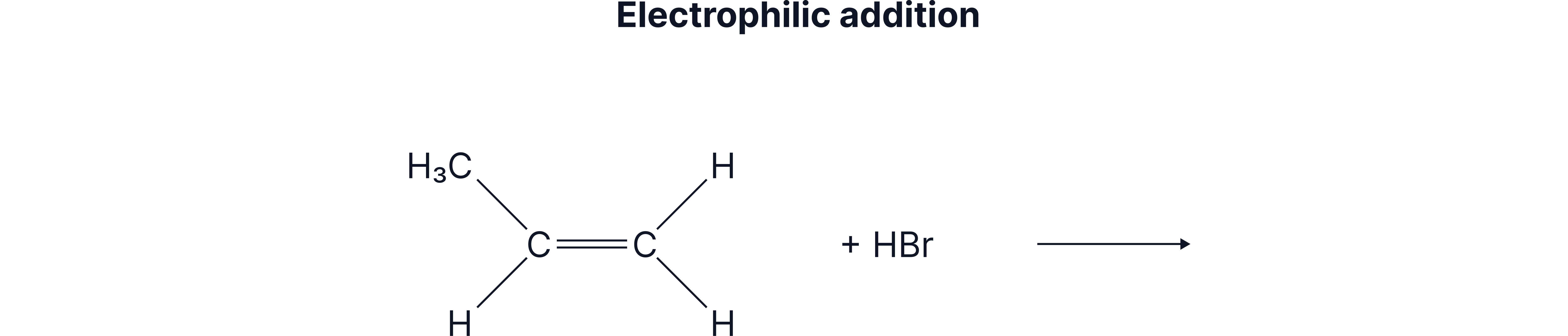
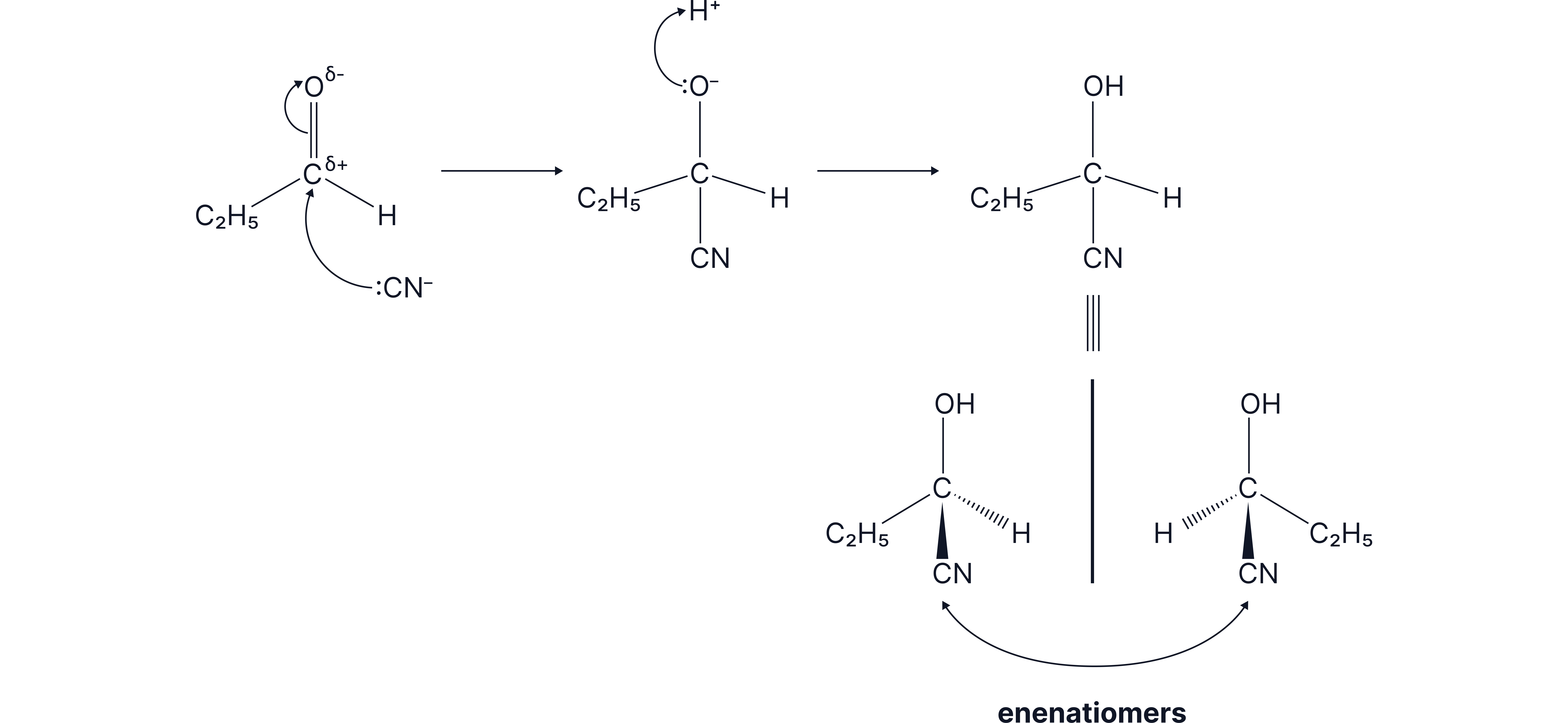
Racemic mixtures are formed when the mechanism involves a trigonal planar group such that the attack on this group happens from both sides and so there is an equal chance of either enantiomer forming. An example of this type of reaction is when HCN reacts with aldehydes/ketones. This is a Nucleophilic Addition reaction.
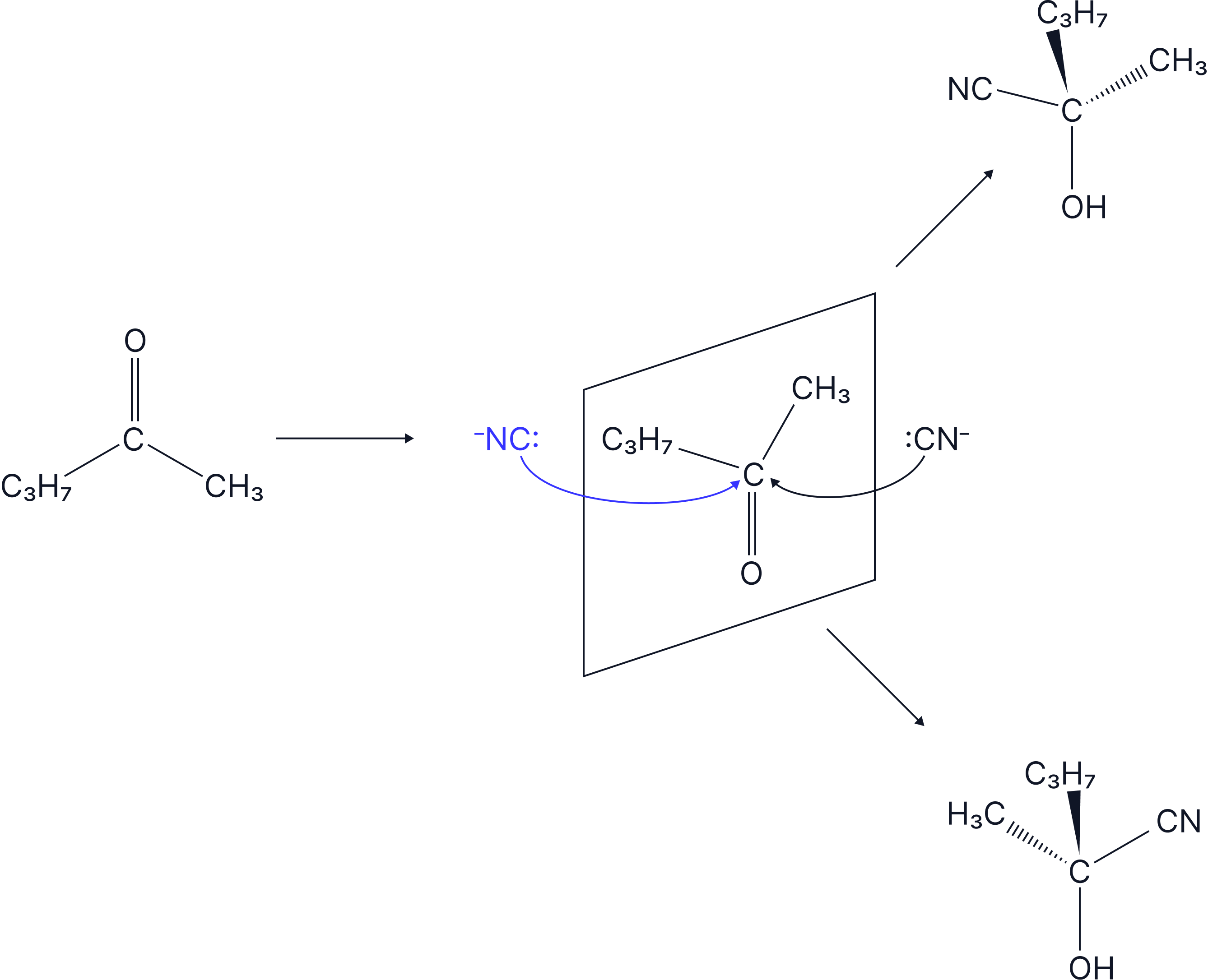
Here, there is an equal chance of attack of :CN- from the back and from the front of the planar carbonyl group.
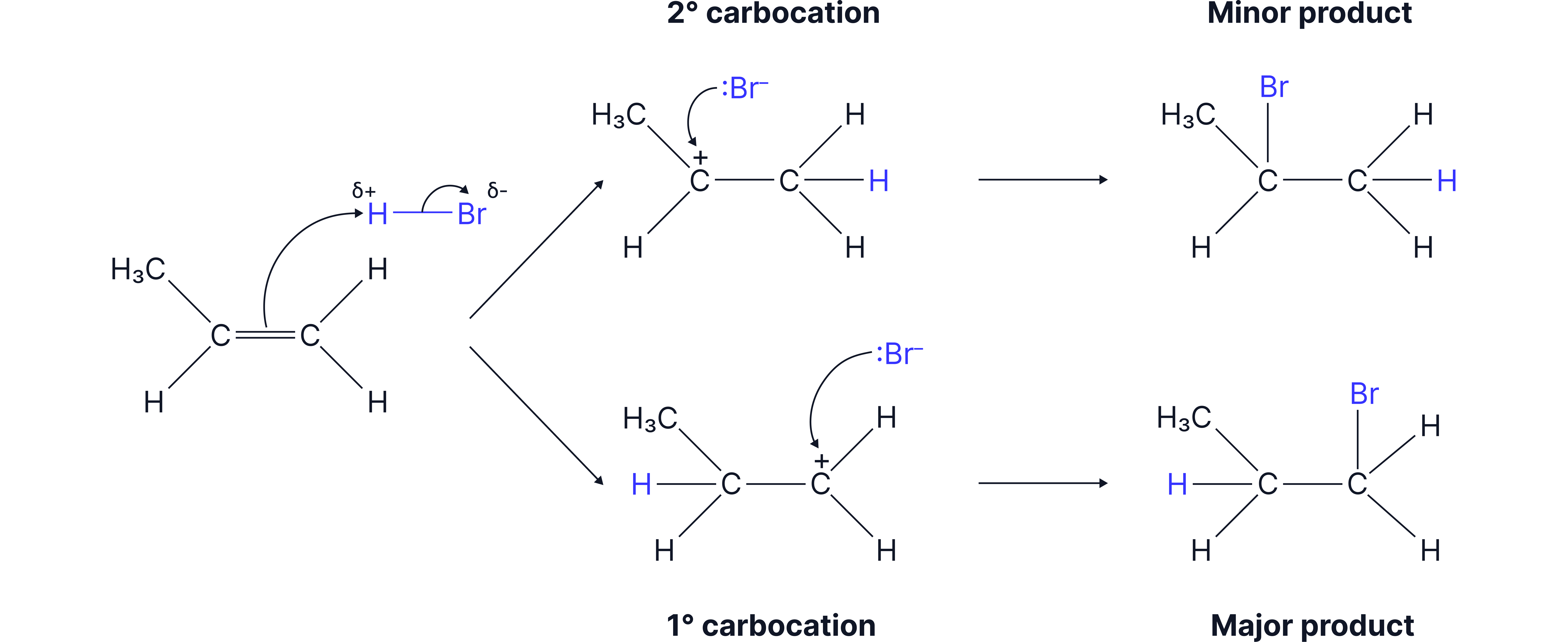
As a result, the products are formed in a 50:50 ratio so a racemate forms.
The full mechanism for a Nucleophilic Addition of HCN to an aldehyde/ketone is as follows
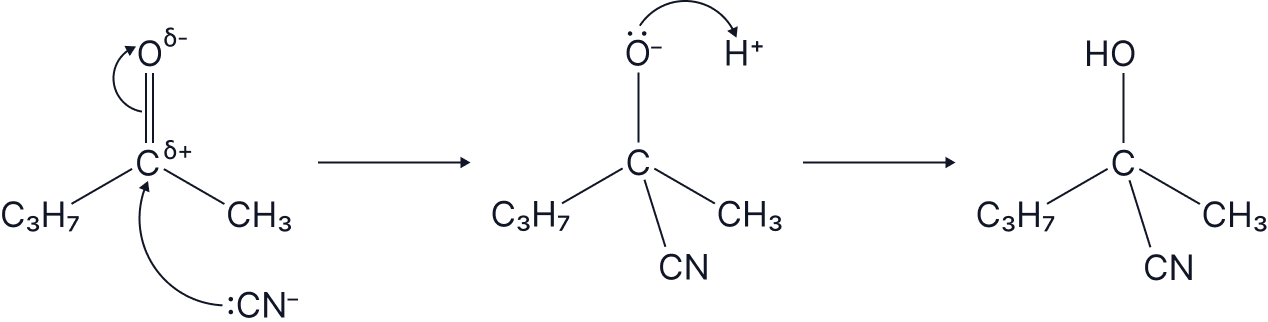
Don’t forget to draw the partial charges due to electronegativity differences.
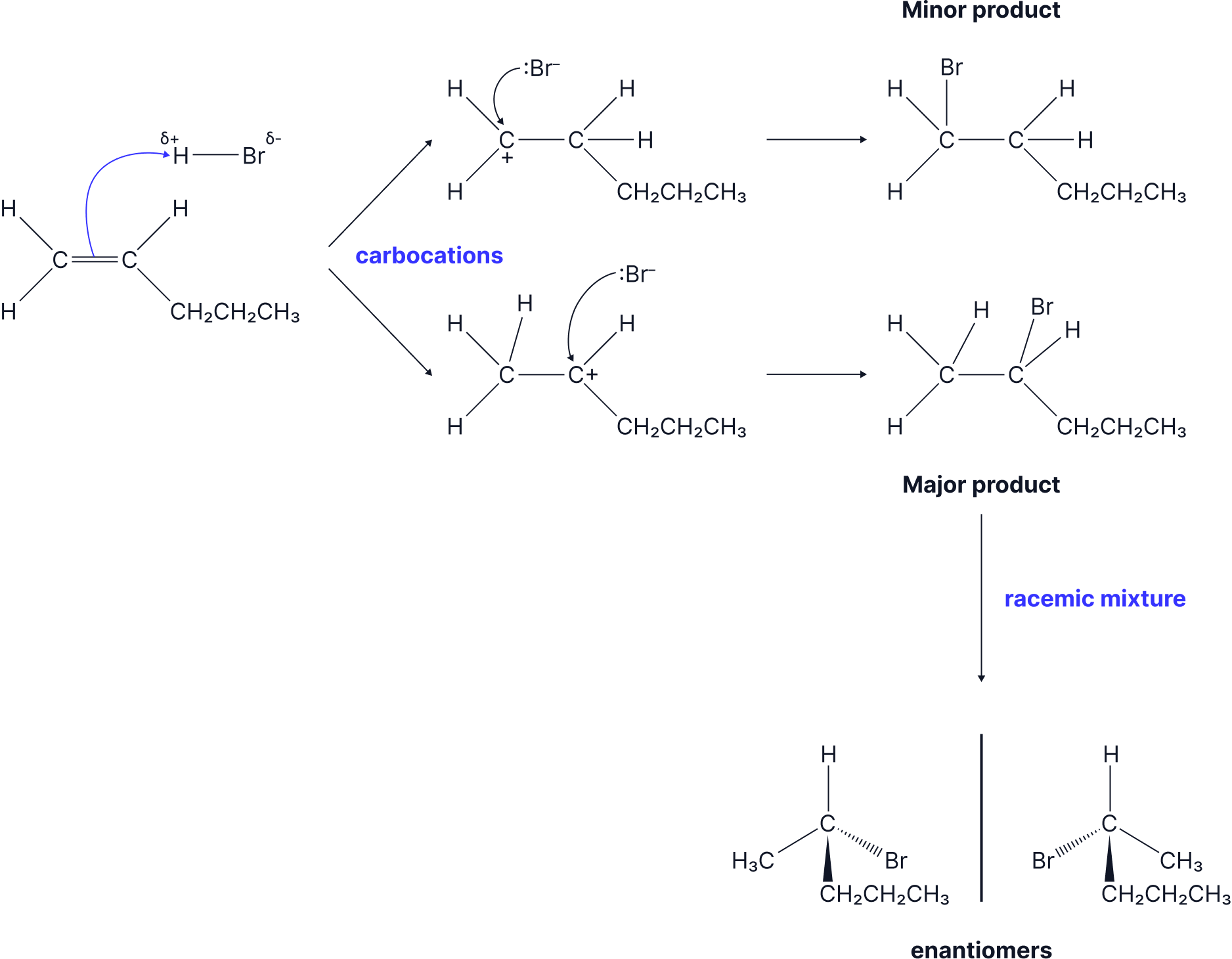
Another reaction which forms a racemic mixture is the Electrophilic Addition of HBr to an unsymmetrical alkene to form a halogenoalkane.

Practice Questions
Question 1:
Define a racemic mixture and state a property of it.
Answer:
Racemic mixture is a mixture which contains equal amounts of two enantiomers. It does not rotate plane polarised light since there are equal amounts of the two chiral molecules.
Question 2:
Draw the mechanism and indicate the products of the following reactions.
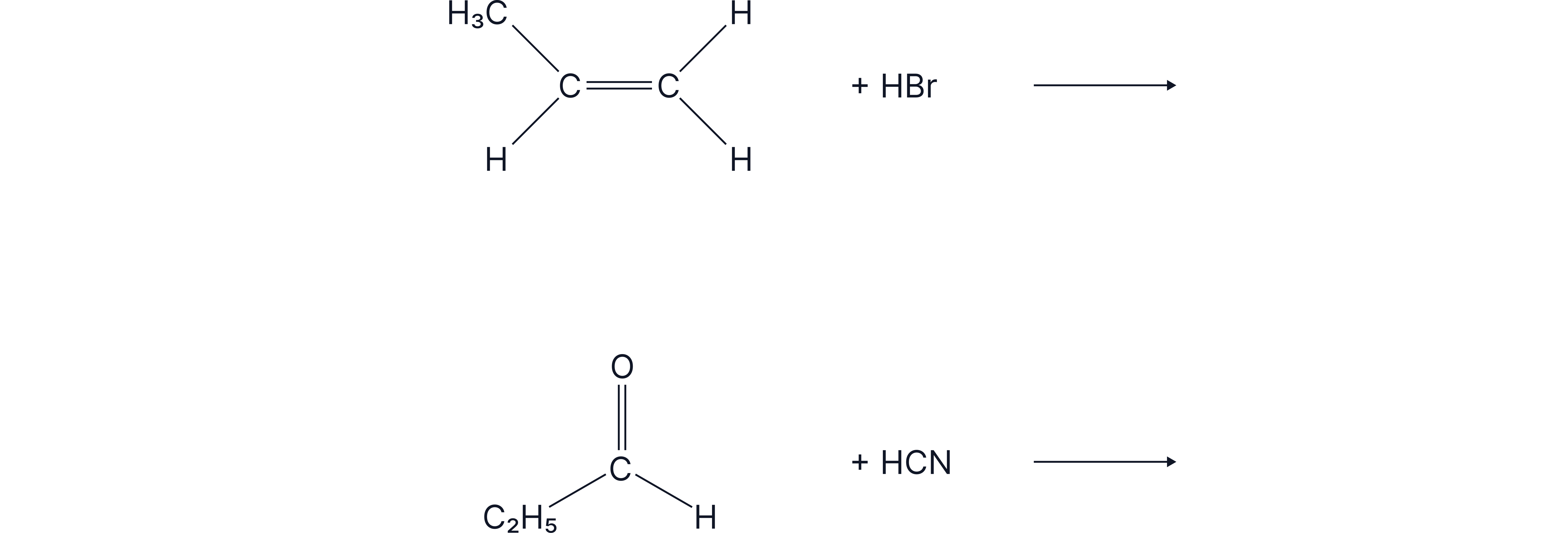
Answer: