Orders and the Rate Equation
Dr. Davinder Bhachu
Teacher

Contents
Introduction and Definitions
Rate of reaction can be defined as the change in concentration of a reactant or product in a given time period. The rate equation shows how the rate of reaction is dependent on the concentrations of reactants and catalysts.
Basic Principles
Rate Equation:
The general form of the rate equation is:
Rate =
where:
= rate constant
= concentrations of reactants A and B
m and n = orders of reaction with respect to A and B
Overall order of reaction =
The order of reaction refers to the power to which the concentration of a reactant (or catalyst) is raised to in the rate equation.
The rate constant (k) is a constant that links the rate of reaction to the concentrations of reactants (and catalysts) in the rate equation. It determines how fast a reaction proceeds under given conditions. k is dependent on temperature, as shown in the Arrhenius equation, .
Order of Reaction
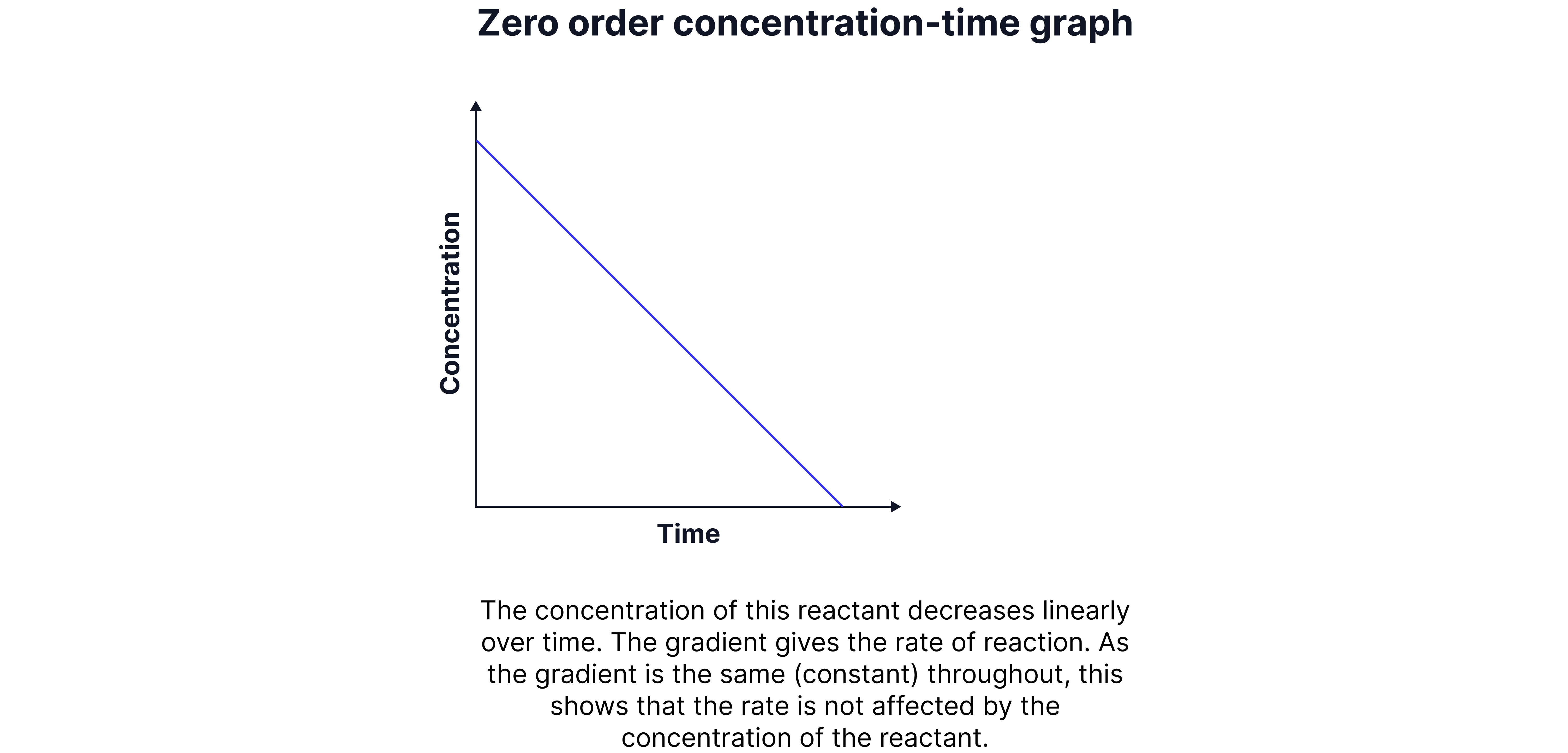
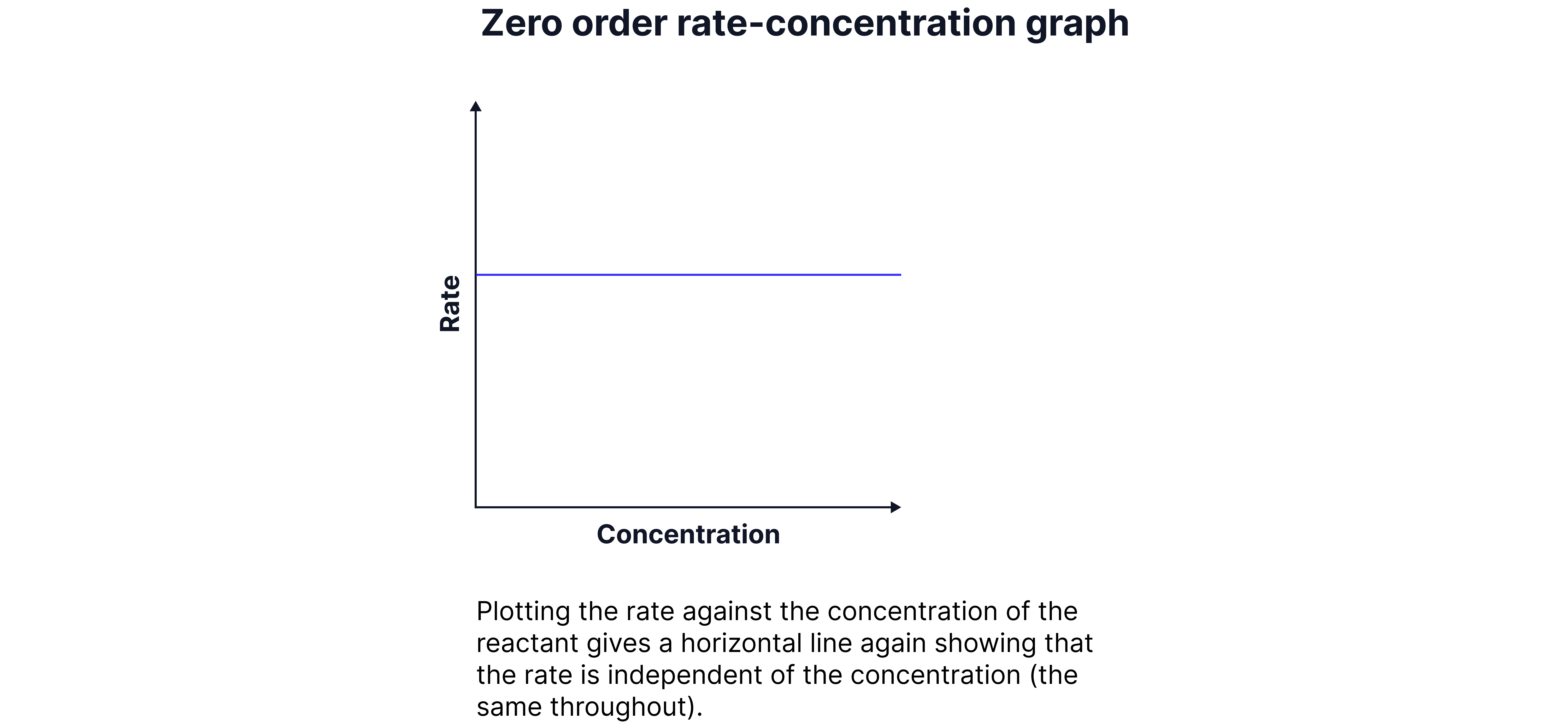
Zero Order
The rate is independent of the concentration of the reactant.
Order of Reactions Practice Question Walkthrough
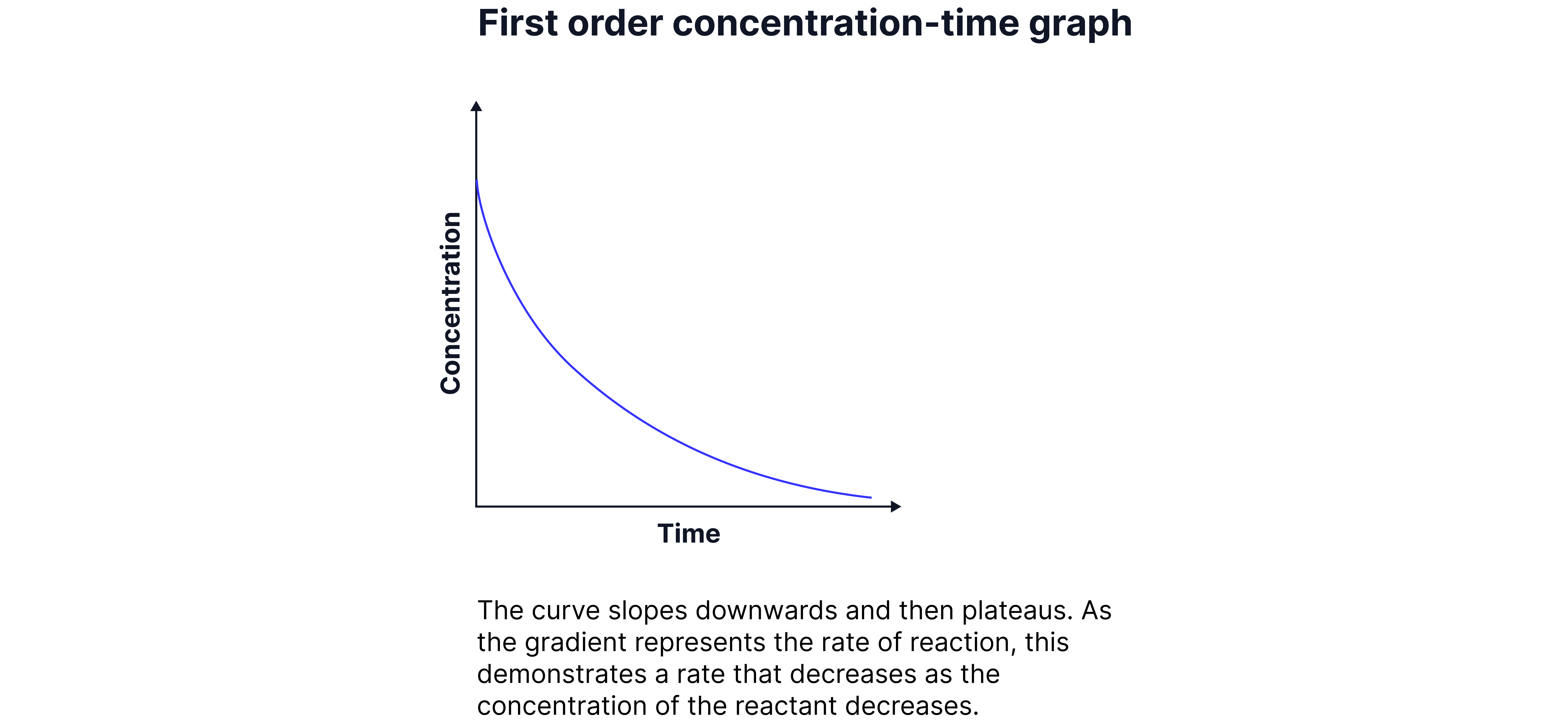
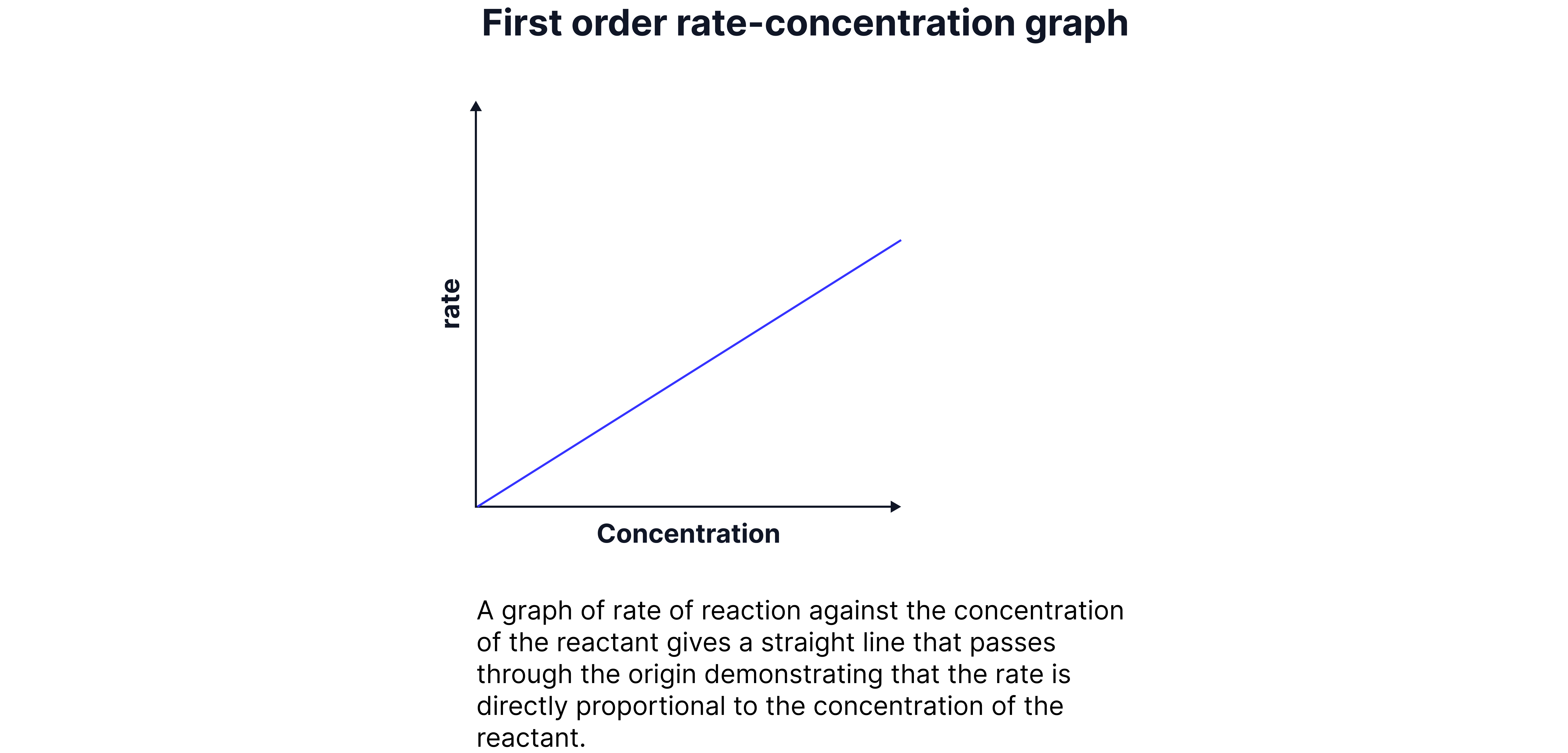
First Orders
The rate is directly proportional to the concentration of the reaction.
Rate =
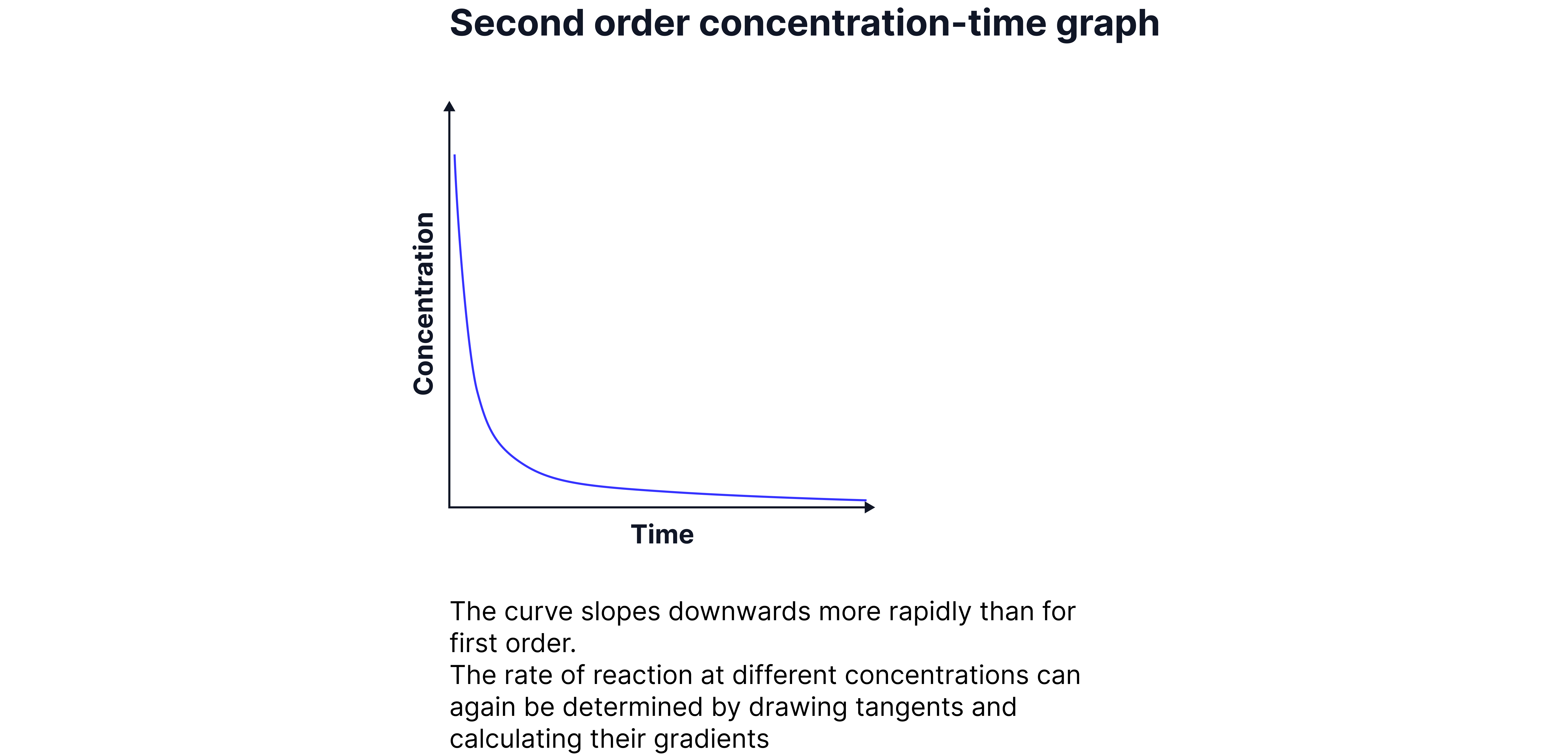
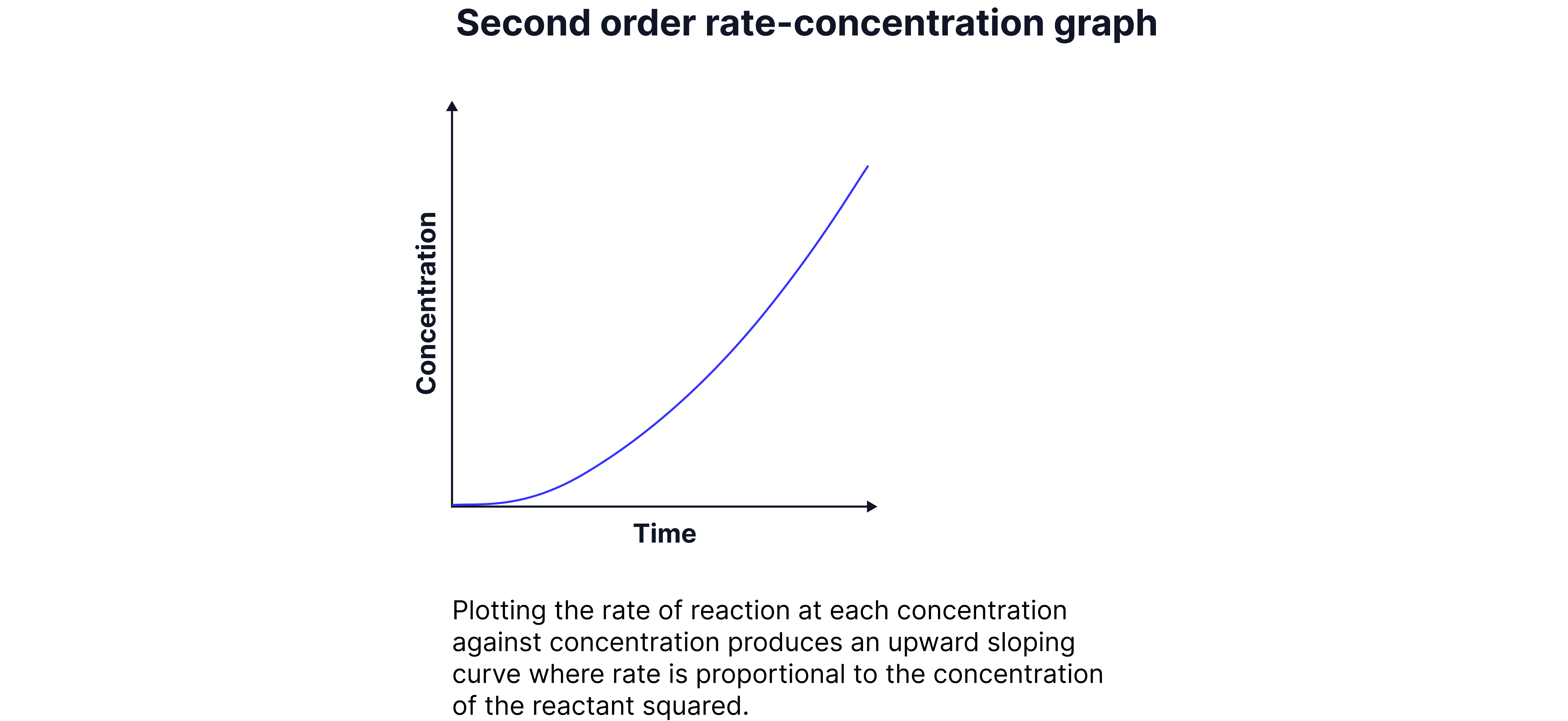
Second Orders
The rate is proportional to the square of the concentration of the reactant.
Rate =
Units of the Rate Constant,
The units of depend on the overall order of reaction.
They can be determined by rearranging the rate equation and substituting the appropriate units.
Worked Example
Determine the units for for the following rate equation:
Rate =
Rearrange
Substitute units
Simplify
(OR )
Determining the Order of Reaction
The order of a reaction can only be determined experimentally using:
1. Initial Rates Method: Measuring the rate of reaction at the very start of the reaction with different concentrations of reactants.
2. Continuous Monitoring Method: Following the concentration of reactants/products over time and plotting graphs. Tangents and gradients determine rate.
Orders can NOT be determined from the balanced symbol equation.
Determining orders from experimental data
For the reaction: the following data were collected:
Expt no. | [X] mol dm-3 | [Y] mol dm-3 | Initial rate mol dm-3 s-1 |
|---|---|---|---|
1 | 0.1 | 0.1 | 2 x 10-3 |
2 | 0.2 | 0.1 | 4 x 10-3 |
3 | 0.1 | 0.2 | 8 x 10-3 |
Expts. 1 and 2, when doubles and is constant, the rate doubles, therefore the reaction is first order with respect to .
Expts. 1 and 3, when doubles and is constant, the rate quadruples , therefore the reaction is second order with respect to .
Rate equation: rate =
Units of k: (OR )
Practice Question
Question 1
State the overall order of reaction based on the rate equation below.
Rate =
Answer:
Third order (First order with respect the concentration of , second order with respect to the concentration of )
Question 2
Determine the rate equation from the following data:
[A] mol dm-3 | [B] mol dm-3 | Rate mol dm-3 s-1 |
0.1 | 0.1 | 1.2 x 10-4 |
0.2 | 0.1 | 2.4 x 10-4 |
0.2 | 0.2 | 4.8 x 10-4 |
Answer:
Rate =
Rate of each doubles as concentration doubles. Therefore, rate and concentration are directly proportional.
Reminders:
Orders are zero, first, and second (not zero, one, and 2)
Orders can NOT be deduced directly from the balanced symbol equation, experimental data is needed.
As orders related to the concentrations of species, square brackets are required.