Organic Synthesis
Lajoy Tucker
Teacher

Introduction to Organic Synthesis
What is Organic synthesis?
The goal is to convert raw materials into desired target compounds, typically through chemical reactions
Key Considerations in Industrial Organic Synthesis
Safety and efficiency are crucial factors when selecting a synthetic route for an organic compound in the industry to ensure it is sustainable and profitable.
Use of non-hazardous starting materials
Chemists aim to choose starting materials that are less hazardous wherever possible.
Examples:
Acid anhydrides are preferred over acyl chlorides because they are less reactive and safer to handle.
Potassium cyanide (KCN) followed by dilute acid is used instead of hydrogen cyanide (HCN), which is highly toxic.
Minimising Solvent Use
Avoiding the use of solvents is beneficial because many are flammable and require distillation for removal, which increases energy consumption.
Reducing the Number of Reaction Steps
Shorter synthetic routes are more cost-effective because:
Fewer steps mean fewer reagents and pieces of equipment are needed.
Each additional step increases the risk of product loss, reducing overall yield.
Fewer steps also contributes to higher atom economy, meaning more of the reactant atoms are incorporated into the desired product.
Checking understanding:
Suggest why chemists might try to design production methods with a high atom economy?
Answer:
A high atom economy minimises waste.
This makes the process more sustainable are more profitable.
Reaction Pathways
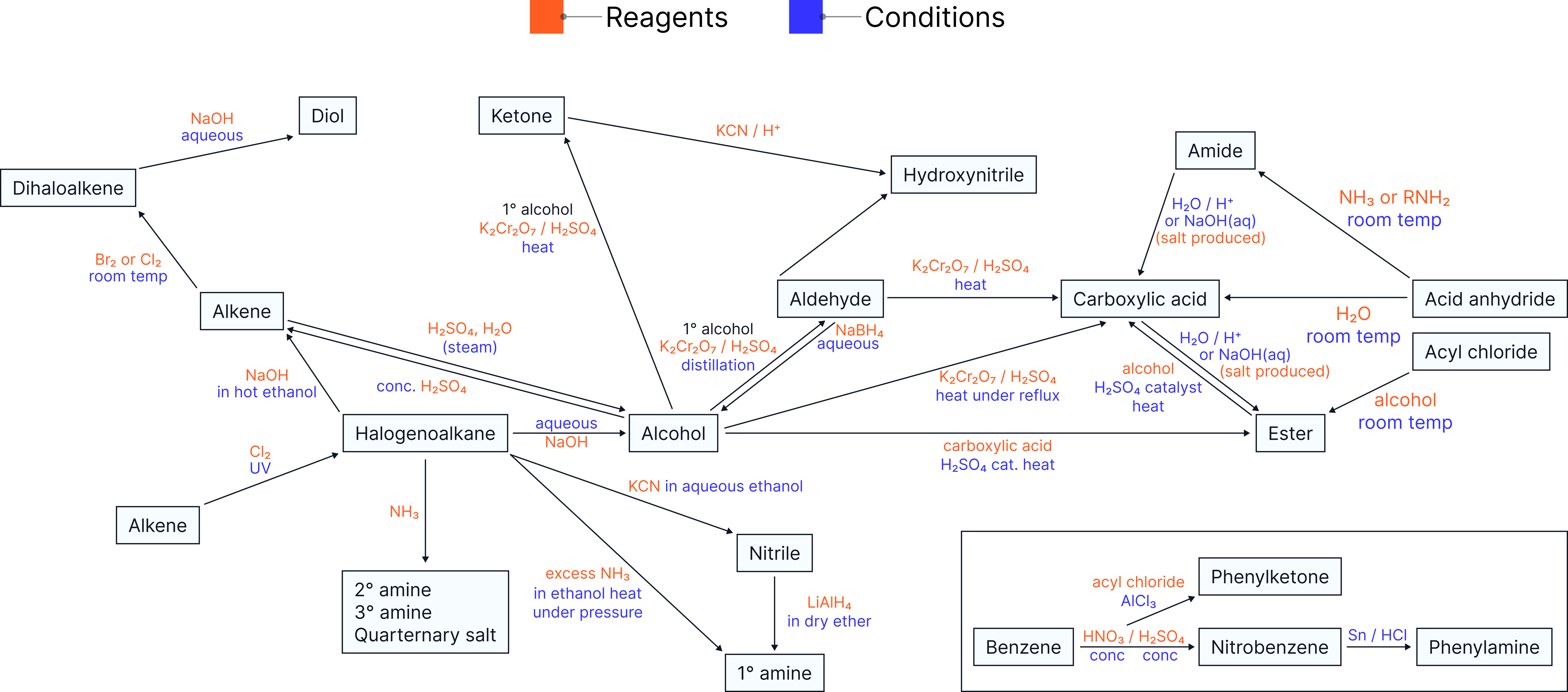
You need to know the reaction pathways from across the specification as well as the reagents and conditions required to form the desired organic product.
Reflux vs Distillation
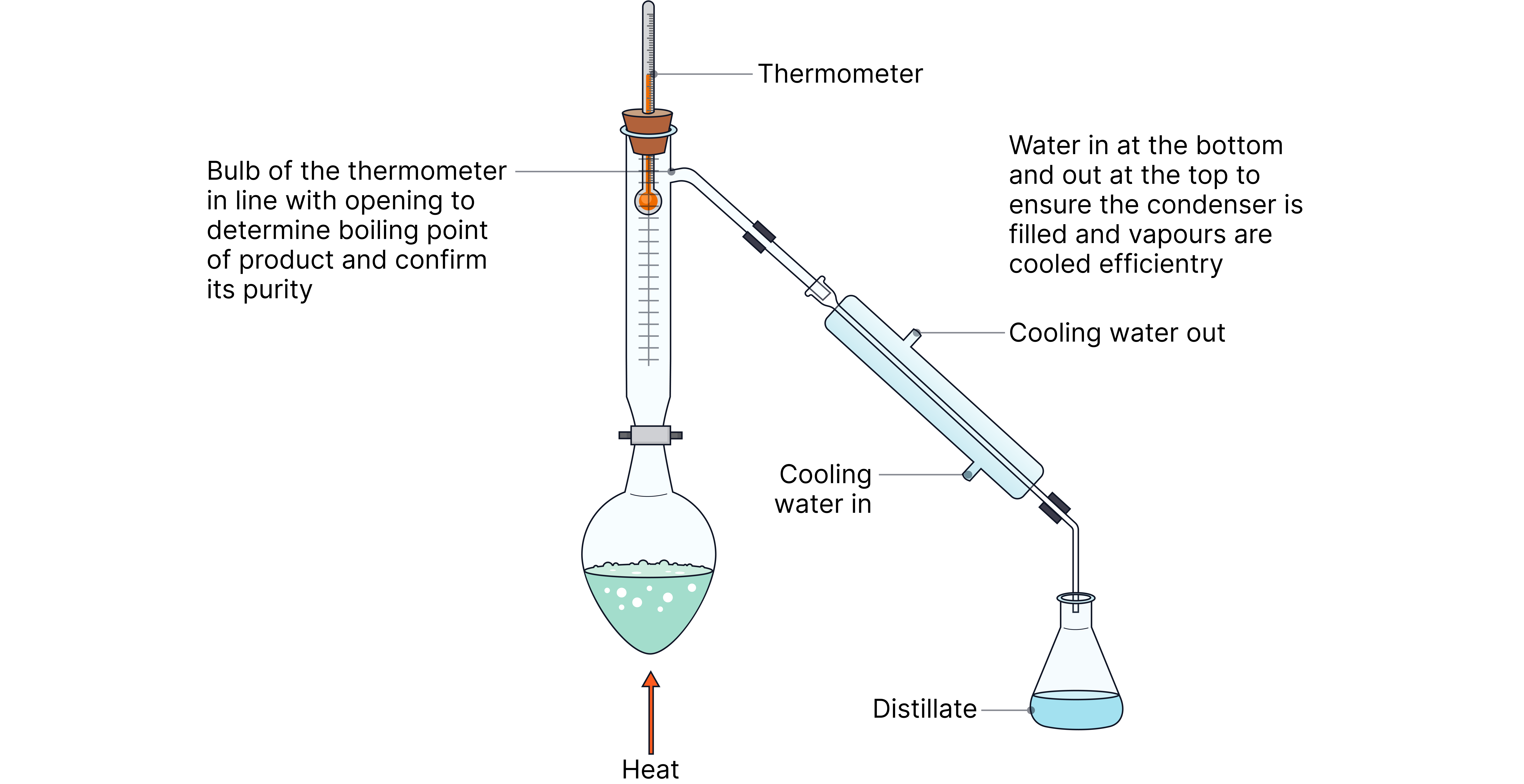
Distillation
Distillation can be used to separate and therefore collect your desired product as it forms. If your desired product has a lower boiling point than your reagents, it will vaporise first, condense and collect in a receiving flask e.g. partially oxidising a primary alcohol to an aldehyde. The aldehyde has a lower boiling than the alcohol meaning it can be separated and collected preventing further oxidation to a carboxylic acid.
The thermometer should read the exact boiling point of your product which allows you to determine its purity.
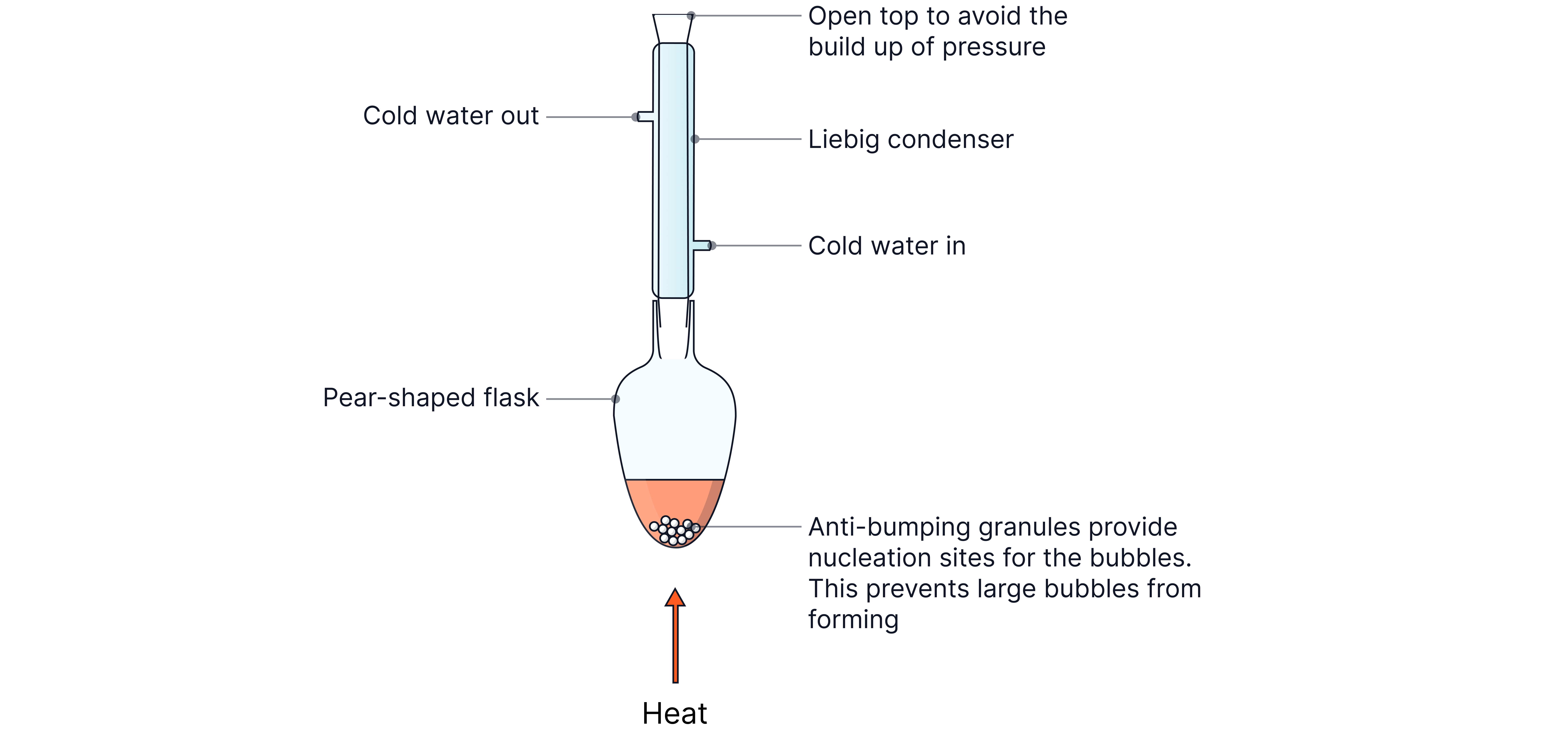
Reflux
Reflux allows reaction mixtures to be heated without risking the loss of volatile species.
The condenser cools and condenses any vapours allowing them to fall back into the reaction flask and therefore the reaction can go to completion. E.g. oxidising a primary alcohol to a carboxylic acid.
Summary
Functional group | Type of reaction | Mechanism(s) | Product(s) |
Alkane | Free radical substitution | Halogenoalkane | |
Alkene | Electrophilic addition | Halogenoalkane Alcohol | |
Halogenoalkane | Nucleophilic substitution Elimination (basic) | Alcohol Amine Nitrile Alkene | |
Alcohol | Dehydration Oxidation | Elimination (acid catalysed) | Alkene Aldehyde Ketone Carboxylic acid |
Aldehyde | Oxidation | Carboxylic acid | |
Aldehyde/ketone | Reduction Addition | Nucleophilic addition | Alcohol Hydroxynitrile |
Carboxylic acid | Condensation | Esters | |
Ester | Hydrolysis | Carboxylic acid (if acid hydrolysis) Carboxylate salt (if alkaline hydrolysis) Alcohol | |
Amine | Acylation Further substitution | Nucleophilic addition –elimination Nucleophilic substitution | Amide 2o/3o amine quarternary ammonium salt |
Nitrile | Reduction | Amine | |
Amide | Hydrolysis | Carboxylic acid (if acid hydrolysis) Carboxylate salt (if alkaline hydrolysis) Amine (if alkaline hydrolysis) Ammonium salt(if acid hydrolysis) | |
Acyl chloride/acid anhydride | Acylation | Nucleophilic addition –elimination | Carboxylic acid Ester Amide |
Benzene | Nitration Friedel-Crafts Acylation | Electrophilic substitution | Nitrobenzene Phenylketone |
Designing synthetic pathways
Key Tips:
At A-Level, most functional groups that you meet only have one associated mechanism. Use that the narrow down your options for suitable reagents.
For multi-step syntheses it is often easier to work backwards from your product (retrosynthesis).
Look out for where the carbon chain has been extended e.g producing a but- from a prop- this suggest the use of CN- to extend the carbon chain.
It is important to consider whether the chosen synthetic pathway produces only one product or a mixture of products.
For example, when evaluating the synthesis of a primary amine, reduction of a nitrile may be chosen over substitution of a halogenoalkane as the nucleophilic substitution reaction can lead to further substitution and therefore a mixture of unwanted products.
Worked Example
Propose a three step pathway to synthesise propanamine acid from ethene. Include reagents and conditions.
Answer:
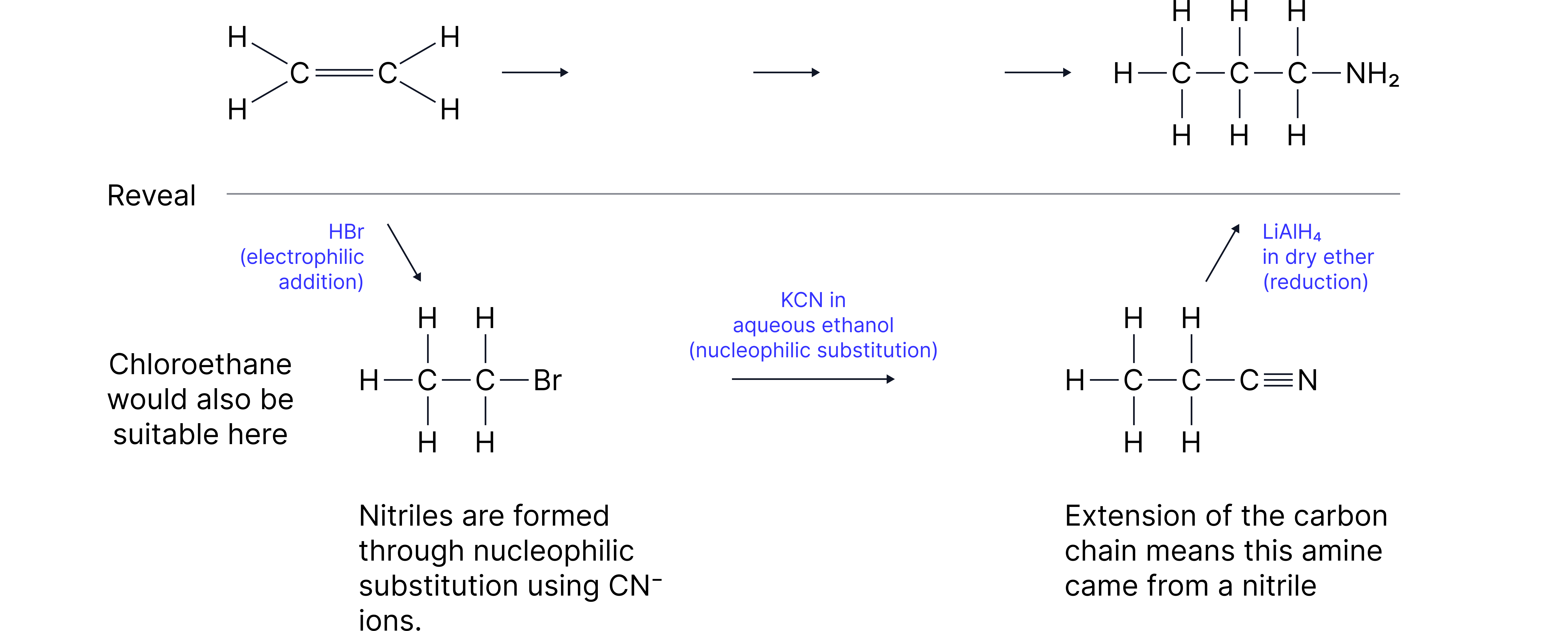
Practice Question:
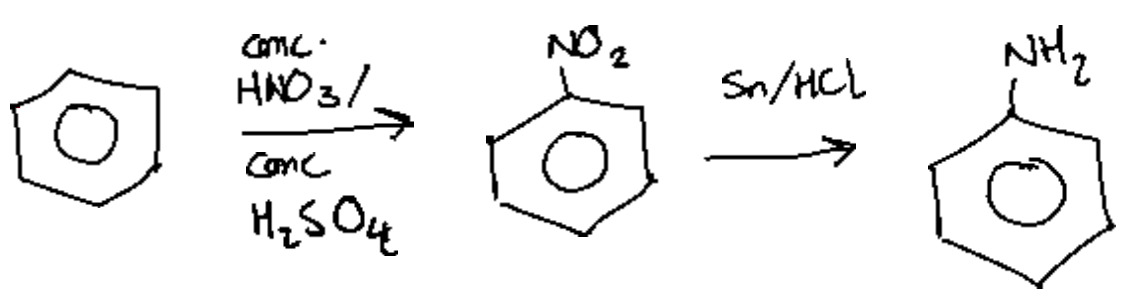
Which sequence of reactions is required to synthesise phenylamine from benzene?
A. Nitration, followed by reduction with
B. Bromination, followed by substitution with
C. Nitration, followed by substitution with
D. Bromination followed by substitution with
Answer:
A.
Benzene undergoes electrophilic addition reactions including nitration using concentrated nitric and sulfuric acid.