RP12 - Thin Layer Chromatography
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Chromatography: A technique for separating and identifying components in a mixture.
Thin Layer Chromatography (TLC): A method using a stationary phase (a thin layer of silica/alumina on a plate) and a mobile phase (solvent) to separate substances by their polarity.
Retention factor : The ratio of the distance moved by a compound to the distance moved by the solvent front.
= Distance moved by substance / Distance moved by solvent front.
Thin Layer Chromatography (TLC) Explained
Basic Principles
Stationary phase: Solid surface (usually silica gel or alumina) coated on a TLC plate.
Mobile phase: Solvent which travels up the plate by capillary action.
Components in a mixture separate depending on the balance between solubility in the mobile phase and retention by the stationary phase.
The principles of chromatography are covered in detail in the ‘Chromatography’ revision note.
Practical Method
Aim: To separate and identify the components in an organic mixture using thin layer chromatography (TLC).
Apparatus & Reagents:
Silica TLC plate
Glass jar with lid
Pencil
Ruler
Capillary tubes
Solvent
Mixture of amino acids or organic dyes
UV lamp
Procedure:
Step | Reason |
1. Draw a baseline in pencil 1cm from the bottom of the TLC plate. | Pencil is insoluble in the solvent and will not interfere with the sample being analysed |
2. Mark spots on this line equally spaced — one for each sample. | No overlap – spots have enough space to separate clearly as they travel up the plate. Fair comparison – each sample starts from the same baseline, making Rf values accurate and comparable. |
3. Use capillary tubes to apply a small drop of each solution to the marked spots. | Reliable identification – prevents mixing of spots that could make results unclear. |
4. Allow each spot to dry before reapplying if needed. | |
5. Add the solvent to a jar to a depth of about 0.5 cm. | The solvent must be placed below the baseline so that the spots do not dissolve directly into the solvent and run down— instead, they travel upwards with the mobile phase, allowing proper separation. |
6. Place the TLC plate in the tank without submerging the spots. | |
7. Cover with a lid | Ensure saturation with solvent vapour. Prevents potentially toxic, volatile solvent from escaping the chromatography tank |
8. Allow the solvent to rise until it is just below the top of the plate. | This allows the solvent front to be marked so the Rf value can be calculated |
9. Remove the plate and immediately mark the solvent front with a pencil | Once the solvent dries, the solvent front cannot be marked. It must therefore be marked immediately so that the Rf value can ne later calculated |
10. Wait for the plate to fully dry. | |
11. Use a UV lamp to mark the spot positions | The spots are invisible and so a locating agent such as ninhydrin or ultraviolet light is used to visualise the spots |
12. Measure the distance moved by each spot and the solvent front then calculate Rf value. |
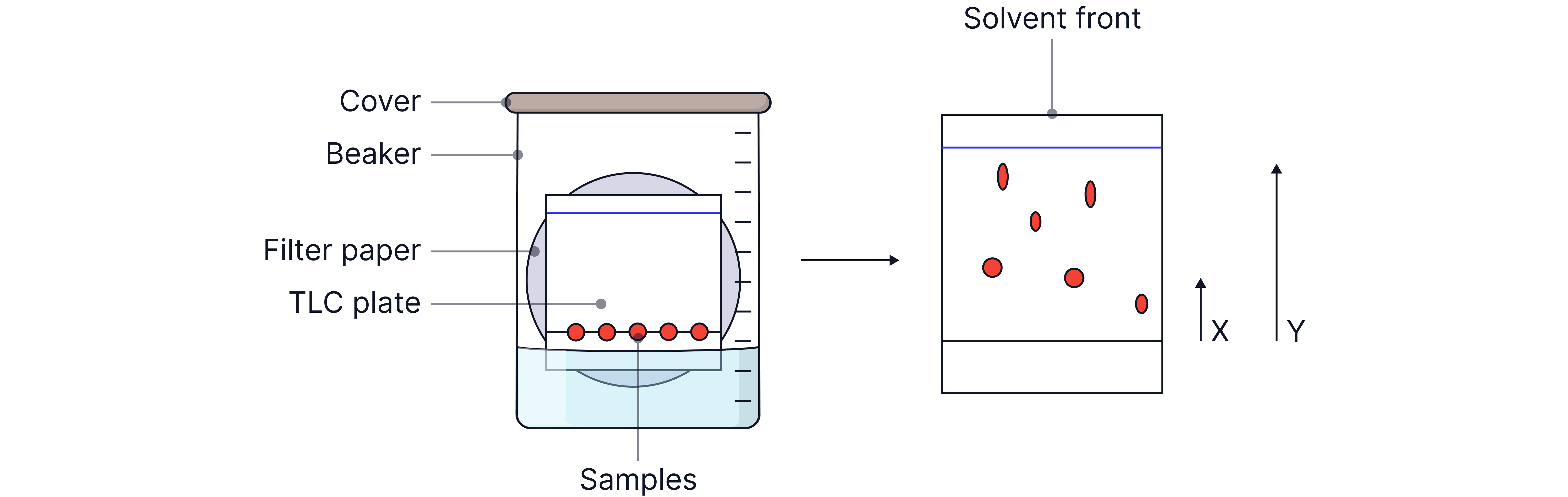
Analysis and Identification
x = distance travelled by sample
y = distance travelled by solvent
The amino acids can be identified by comparing the Rf values to known values in reference books.
Practice Questions
Question 1
Why must the baseline and solvent front be marked in pencil, not pen?
Answer
Mark Scheme:
Ink from a pen would dissolve in the solvent and interfere with the results.
Question 2
A compound has an Rf value of . The solvent front moved . How far did the compound move?
Answer
Distance Solvent front
Key Tips and Reminders
Always use a pencil — pen inks dissolve in solvent.
Ensure solvent level is below the baseline.
TLC plates must be kept covered to prevent solvent evaporation.
Visualisation is essential for colourless compounds (use UV lamps).
Rf values are unitless and always < 1.