Standard Electrode Potentials
Lajoy Tucker
Teacher

Contents
The Potential of an Electrode
1. When a metal contacts a solution of its ions, an equilibrium establishes:
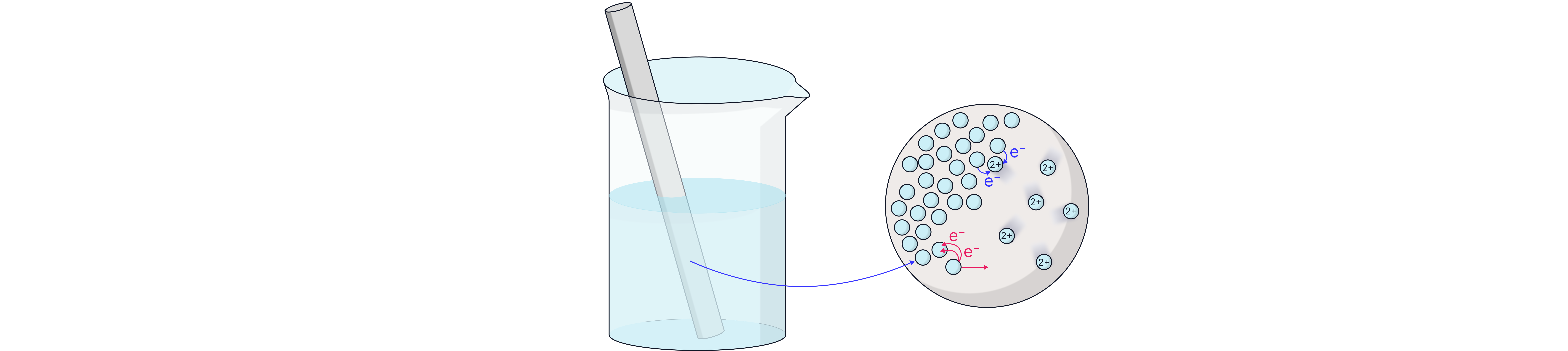
This equilibrium position determines the electrode's potential:
Equilibrium left: Electron buildup on metal → negative potential
Equilibrium right: Electron depletion from metal → positive potential
Three Types of Electrodes:
1. Metal electrodes: - metal in contact with its ions
2. Gas electrodes: - requires inert platinum electrode
3. Redox electrodes: - two oxidation states of same element
You cannot measure the potential of a single electrode - just like you cannot measure a single person's height without a reference point. We can only measure potential differences between two electrodes.
Representing Cells and Calculating Cell EMF Explainer Video
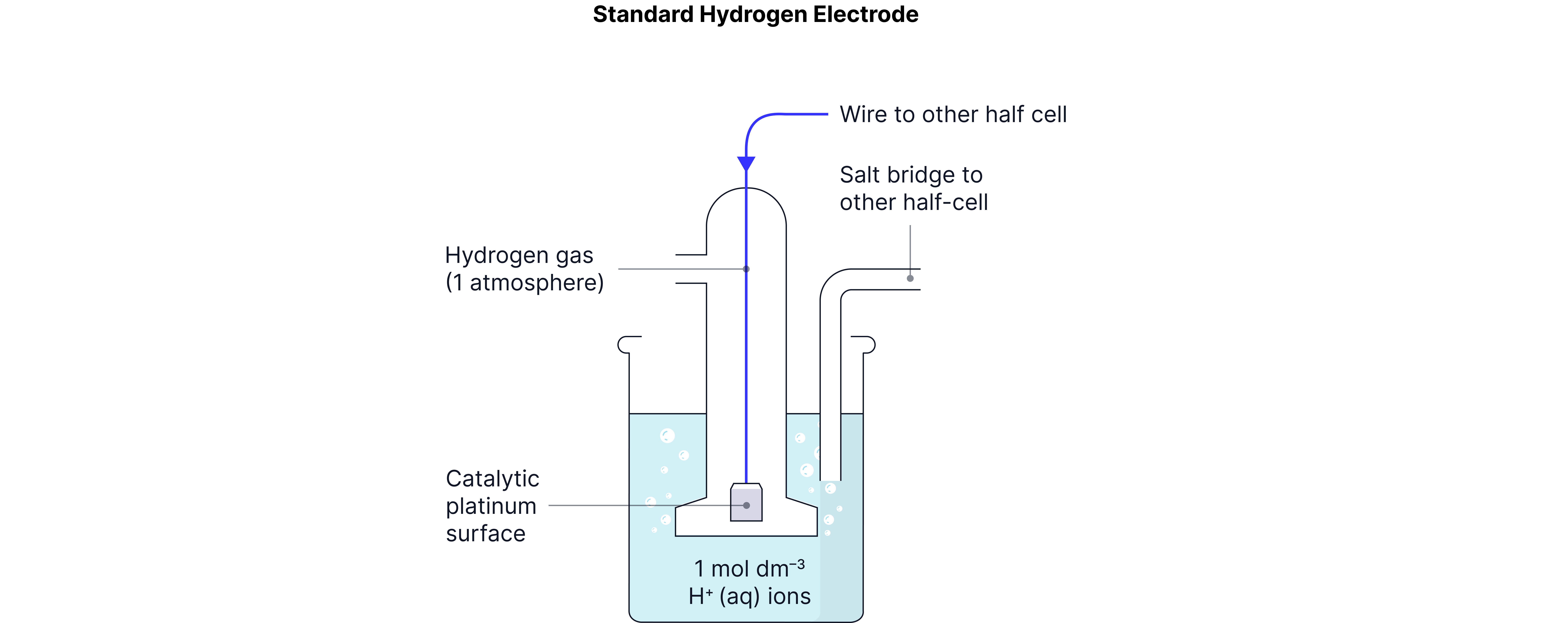
The Standard Hydrogen Electrode (SHE) - Our Reference Point
The Standard Hydrogen Electrode is assigned by convention and serves as our "reference" for all other measurements.
Components of SHE:
Platinum electrode (coated with platinum black for surface area)
Hydrogen gas at 100 kPa pressure
(usually 1M HCl)
Temperature:
Half-equation:
Cell notation:
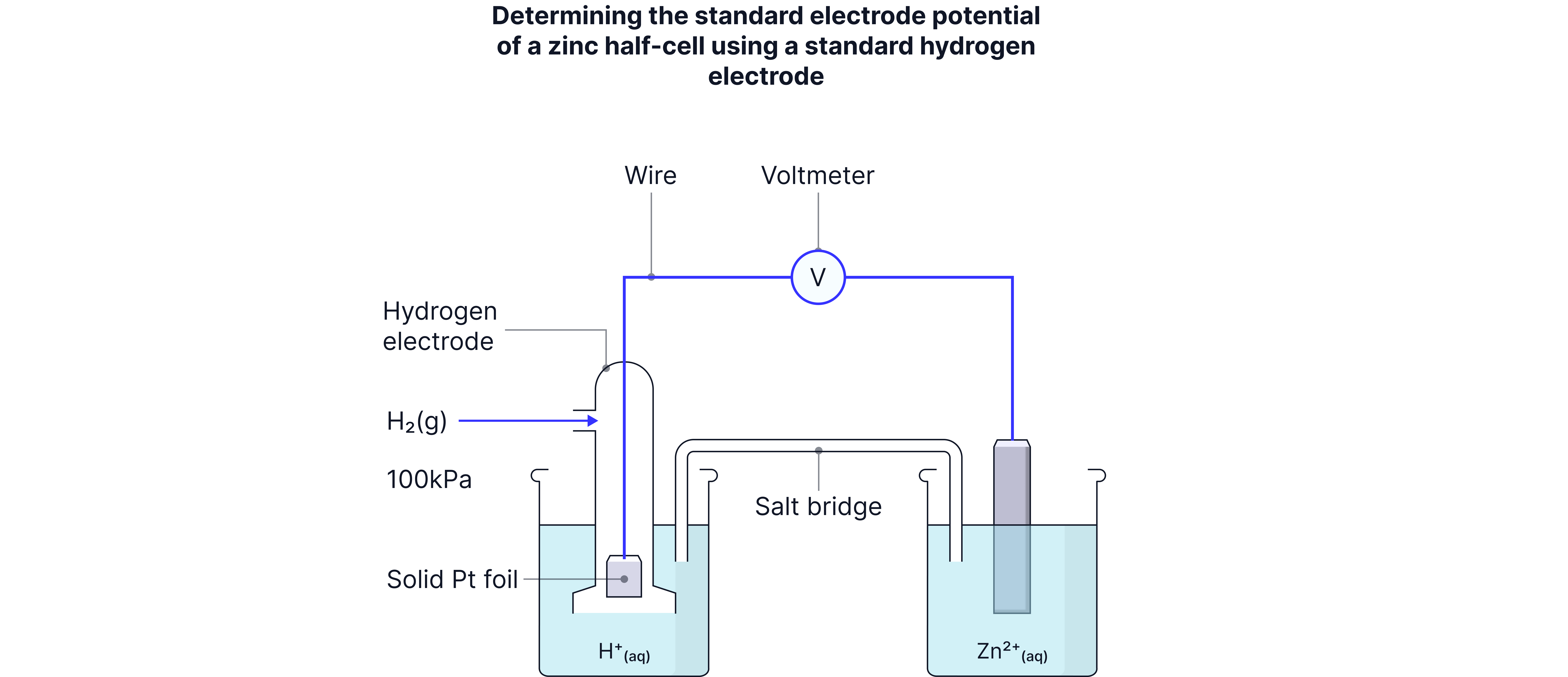
Measuring Against the SHE
The Formula:
When measuring unknown electrode potentials:
SHE always on the left
Test electrode on the right
Secondary Standards - Practical Alternatives
Since SHE is difficult to use (involves flammable gas), secondary standards are often used:
Silver/silver chloride:
Calomel electrode:
These are calibrated against SHE but much more convenient for routine use.
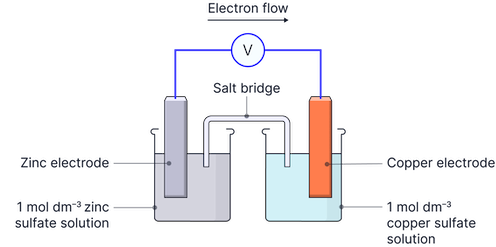
Setting Up Electrochemical Cells
Every electrochemical cell consists of:
Two half-cells: Separate compartments where oxidation and reduction occur
Electrodes: Conductive materials (usually metals) that allow electron transfer
Electrolyte solutions: Ionic solutions that facilitate ion movement
Salt bridge: Connection allowing ion flow between half-cells
External circuit: Wire connecting the electrodes for electron flow
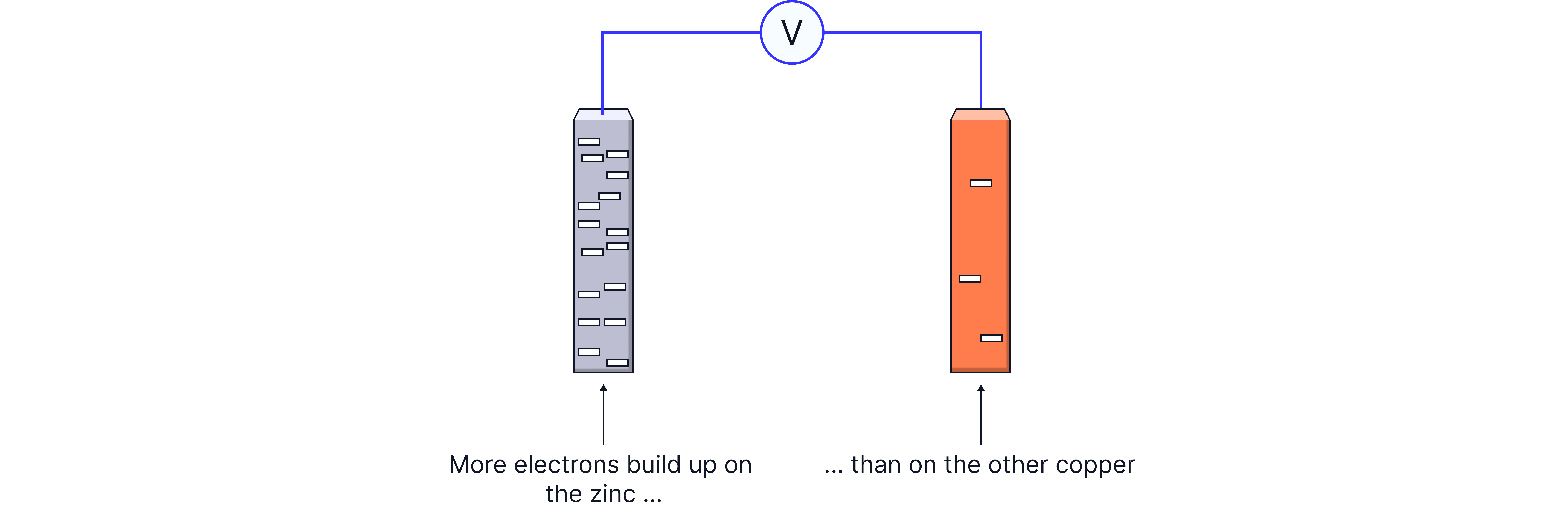
Why Do We Get a Voltage?
When two different metals are placed in solutions of their own ions, they have different tendencies to lose electrons (oxidize). The metal with the greater tendency to oxidize will:
Release more electrons into the external circuit
Create a negative charge buildup
Become the negative electrode (anode)
The other metal will:
Accept electrons from the external circuit
Become the positive electrode (cathode)
Allow reduction to occur
This difference in electron-losing tendency creates a potential difference (voltage) between the electrodes.
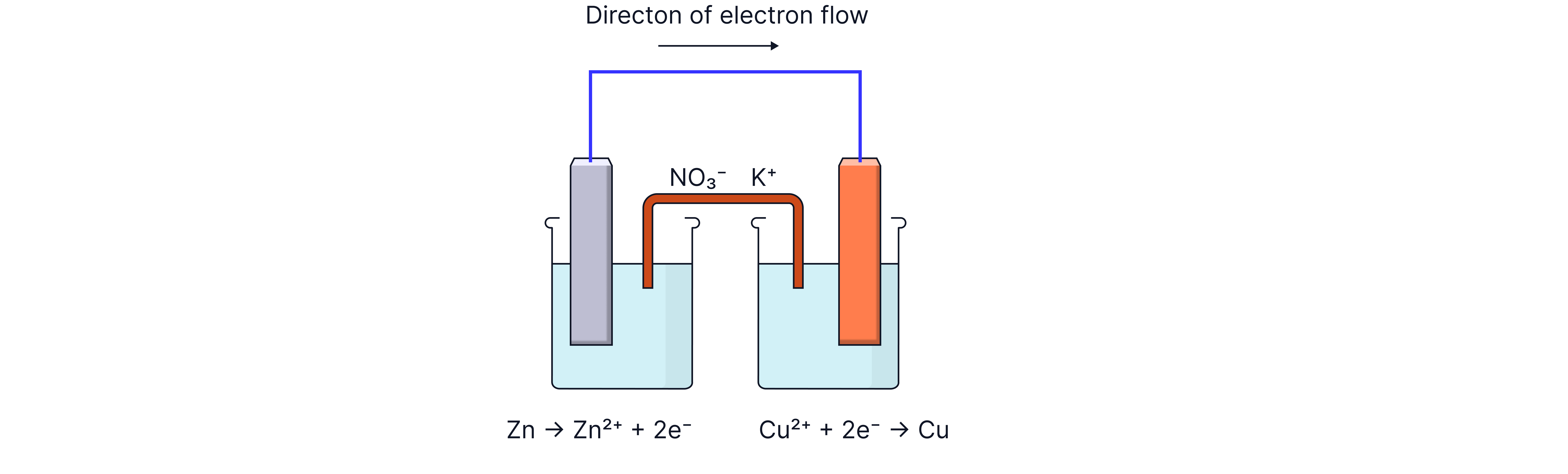
The Salt Bridge - A Crucial Component
What is a Salt Bridge?
A salt bridge is typically a piece of filter paper or porous material soaked in an unreactive salt solution (commonly potassium nitrate, ).
Why Do We Need It?
Without a salt bridge, the cell would stop working almost immediately because:
1. As electrons flow through the external circuit, positive ions accumulate in one half-cell in this case, the left-hand half-cell
2. Negative ions accumulate in the other half-cell i.e. the right-hand half-cell
3. This charge buildup would prevent further electron flow
The salt bridge solves this by:
Allowing anions (negative ions) to flow toward the anode (negative electrode) where oxidation occurs
Allowing cations (positive ions) to flow toward the cathode (positive electrode) where reduction occurs
Maintaining electrical neutrality in both half-cells
Completing the circuit without allowing the solutions to mix
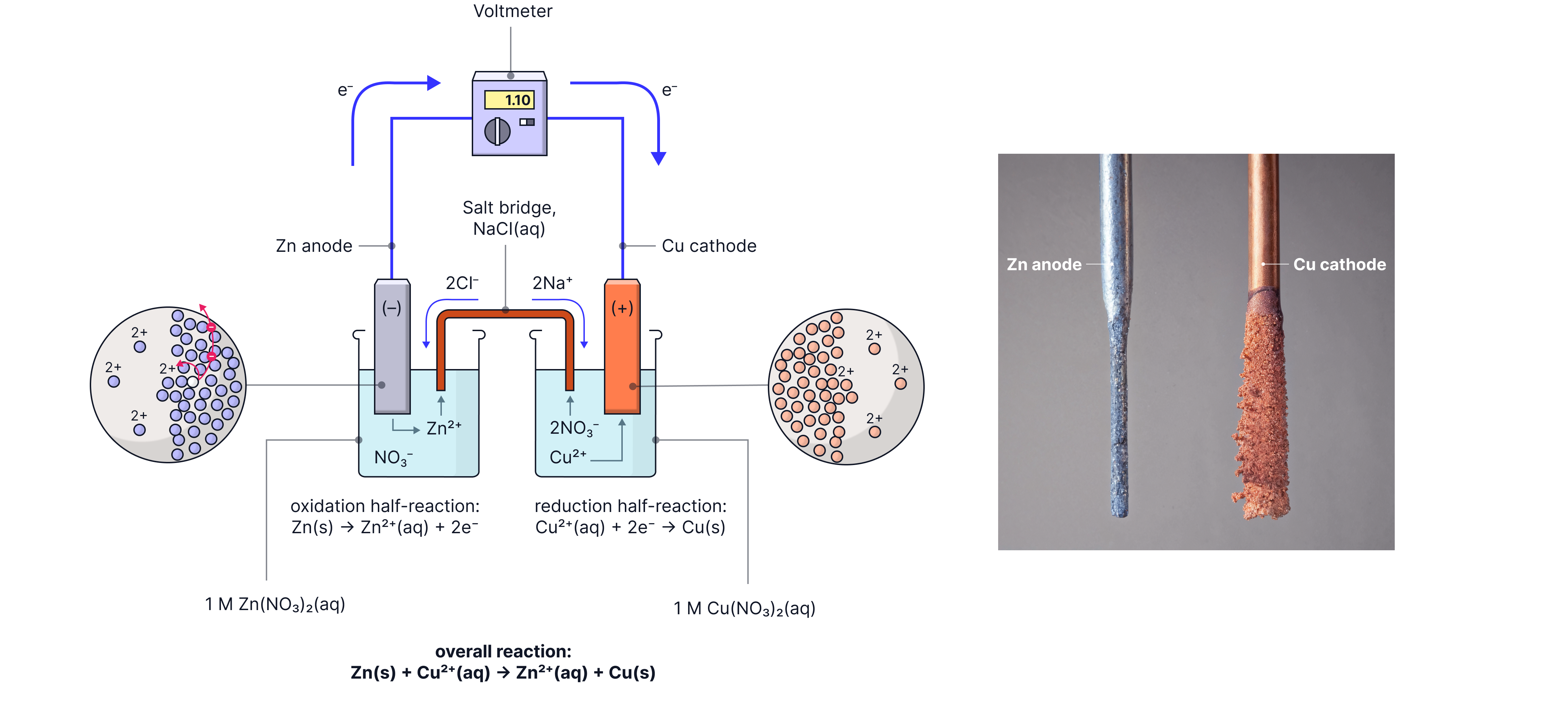
Why this happens:
At the anode: Metal atoms lose electrons and become positive ions, creating excess positive charge
Anions from the salt bridge flow in to balance this positive charge
At the cathode: Positive ions gain electrons and are reduced, creating excess negative charge
Cations from the salt bridge flow in to balance this negative charge
Why Not Just Use a Wire?
Wires conduct electricity through movement of electrons only
Solutions conduct electricity through movement of ions (both cations and anions)
The salt bridge provides ionic conduction to complete the circuit
Without ionic movement, charge buildup in the solutions would quickly stop the cell from working
Think of it this way: electrons flow through the external wire, but ions must flow through the salt bridge to maintain electrical neutrality in both half-cells. You need BOTH types of charge movement for the cell to function.
Salt Selection Criteria
The salt must be:
Chemically inert - doesn't react with electrodes or solutions
Highly soluble - ensures good ionic conductivity
Unreactive ions - won't form complexes or precipitates
Examples of incompatible combinations:
with copper systems: ions form complexes with ions
with manganate systems: ions can be oxidized by (manganate is a strong oxidizing agent)
with silver systems: precipitate would form
This is why is preferred - both and are relatively unreactive ions that:
is very difficult to reduce (highly negative )
is stable to oxidation under normal conditions
Neither form complexes or precipitates with most electrode system
Standard Conditions Required:
Temperature: 298K
Concentration: 1.0 mol dm⁻³ for all ions
Pressure: 100 kPa for gases
Minimal current flow (high-resistance voltmeter)
Why Standard Conditions?
The position of electrode equilibria change depending on the conditions. This is why we use standard condition. If we deviate from standard conditions we can predict the effect on the cell voltage by applying Le Chatelier's principle:
For
Increasing []: Shifts right → more positive potential
Decreasing []: Shifts left → more negative potential
The forward reaction is endothermic. If the temperature is increased, then the equilibrium moves in the endothermic direction, i.e. moves forward and the potential becomes more positive.
If the temperature is decreased, then the equilibrium responds by moving in the exothermic direction, i.e. the reverse reaction and the potential becomes more negative.
High-Resistance Voltmeters - A Crucial Detail
Why High Resistance?
A high-resistance voltmeter is essential because:
1. Minimizes current flow: Tiny current (nanoamps) means negligible reaction rates
2. Maintains near-equilibrium: Reactions occur extremely slowly
3. Accurate measurement: Voltage reading remains stable
4. Prevents cell discharge: Reactants aren't consumed during measurement
5. When measuring: Negligible current → reactions virtually stopped → stable voltage
6. When in use: Significant current → reactions proceed → voltage drops over time and eventually drops to 0V as the reactants are consumed
Determining Cell Voltage
Universal Formula:
Step-by-Step Method:
1. Identify which half-reaction has higher E° (this is reduced)
2. Apply formula:
3. Positive result confirms reaction feasibility
Example 1
Calculate the standard cell voltage for a cell containing zinc and copper electrodes.
Step 1: Identify which has the higher (more positive)
(more positive)
(more negative)
Step 2: More positive electrode will be reduced (cathode)
Step 3: More negative electrode will be oxidized (anode)
Step 4: Apply formula
Overall reaction:
Worked Examples and Solutions
Question 1
A cell is constructed using magnesium and silver electrodes. Calculate the cell voltage.
Solution:
More positive:
More negative:
Overall reaction:
Question 2
Calculate the EMF of a cell made from lead and nickel electrodes.
Solution:
More positive:
More negative:
Overall reaction:
Question 3
Calculate the cell voltage when Fe³⁺/Fe²⁺ and Cu²⁺/Cu half-cells are connected.
Fe³⁺(aq) + e⁻ → Fe²⁺(aq) E° = +0.77V
Cu²⁺(aq) + 2e⁻ → Cu(s) E° = +0.34V
Solution:
More positive:
More negative:
Balanced overall reaction:
Reduction:
Oxidation:
Overall:
Question 4
A cell contains I₂/I⁻ and Br₂/Br⁻ half-cells. Calculate the standard EMF.
Solution:
More positive:
More negative:
Overall reaction:
Question 5
Calculate the cell voltage for a hydrogen half-cell connected to a zinc half-cell.
Solution:
More positive:
More negative:
Overall reaction:
Question 6
Calculate the EMF of a cell containing permanganate and chlorine systems under acidic conditions.
Solution:
More positive:
More negative:
Balanced overall reaction:
Reduction:
Oxidation:
Overall: