Trends Across Period 3
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
What are the period 3 elements?
The elements from sodium () to argon () are in the third row of the periodic table.
We refer to the elements of this row as period 3 elements.
Periodic trends:
Periodic trends are predictable patterns in the properties of elements across a period or down a group.
In A-Level chemistry, we discuss trends in atomic radius, first ionisation energy and melting point.
Basic principles to consider when discussing trends across period 3:
Atomic structure - explains the trends in size and ionisation energy
Structure and bonding – determine the melting points of elements:
The elements across period 3 have varying types of bonding
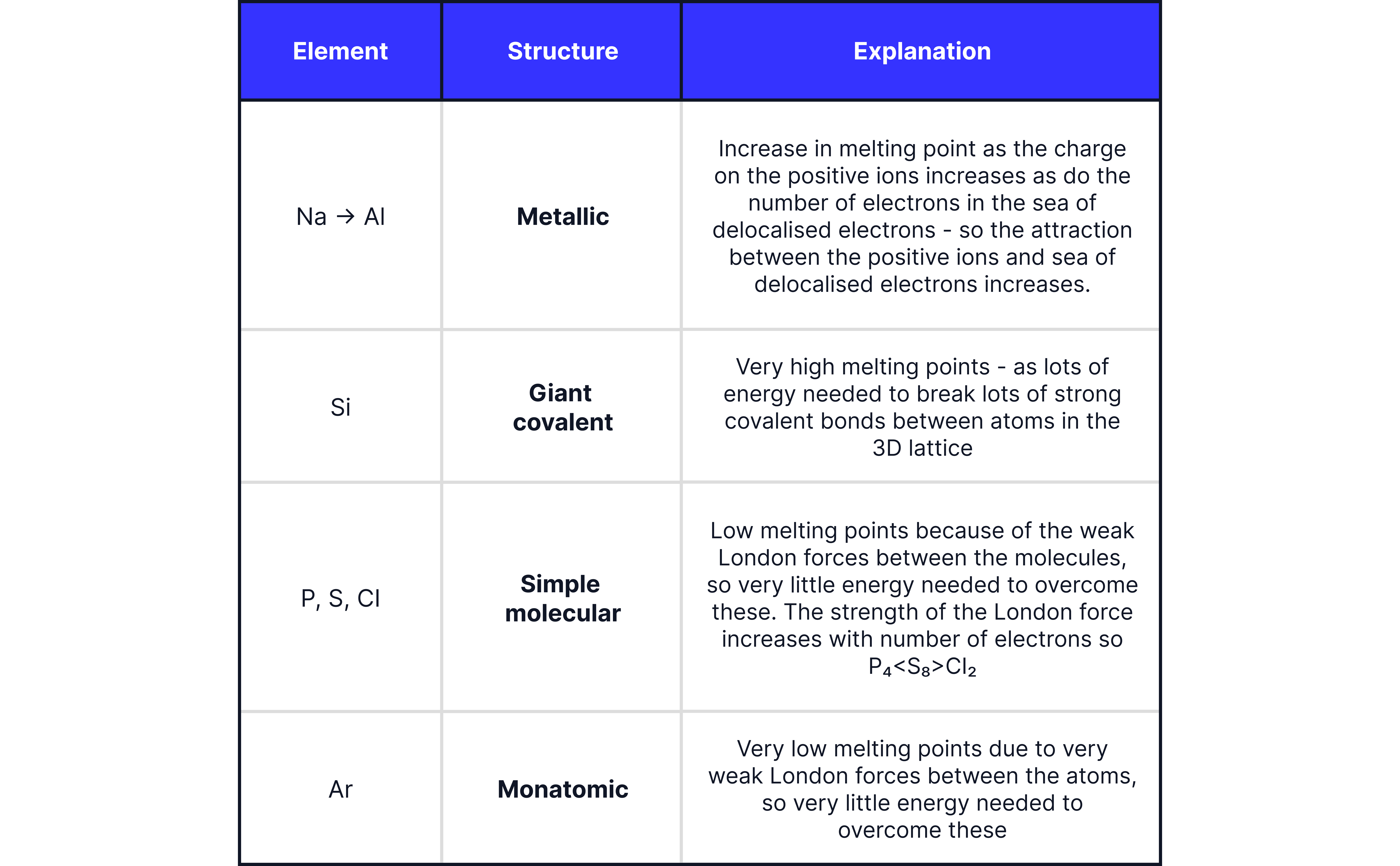
to : Metallic bonding.
: Giant Covalent structure.
, , : Simple molecular substances.
: Monatomic gas.
Explaining Trends Across Period 3
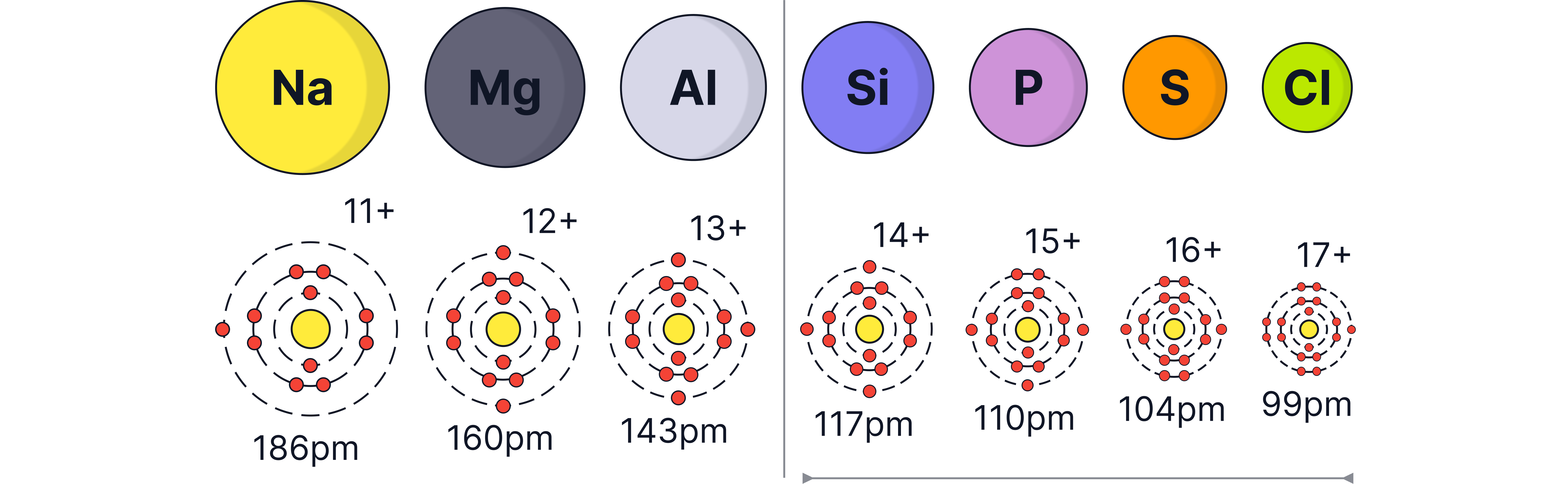
Atomic Radius
Trend:
The atomic radius decreases across Period 3 ()
Explanation:
Across period 3, there is an increasing nuclear charge as there is an increase in protons
Electrons are added to the same energy level and so no significant increase in shielding.
There is a greater electrostatic attraction between the nucleus and the outer electrons.
Therefore, the atomic radius decreases as you move across period 3 from left to right
First Ionisation Energy
First ionisation energy is the energy required to remove one mole of electrons from one mole of gaseous atoms
Trend:
Overall, there is a general increase in first ionisation energy for the same reason as atomic radius above leading to more energy being required to remove the outermost electron.
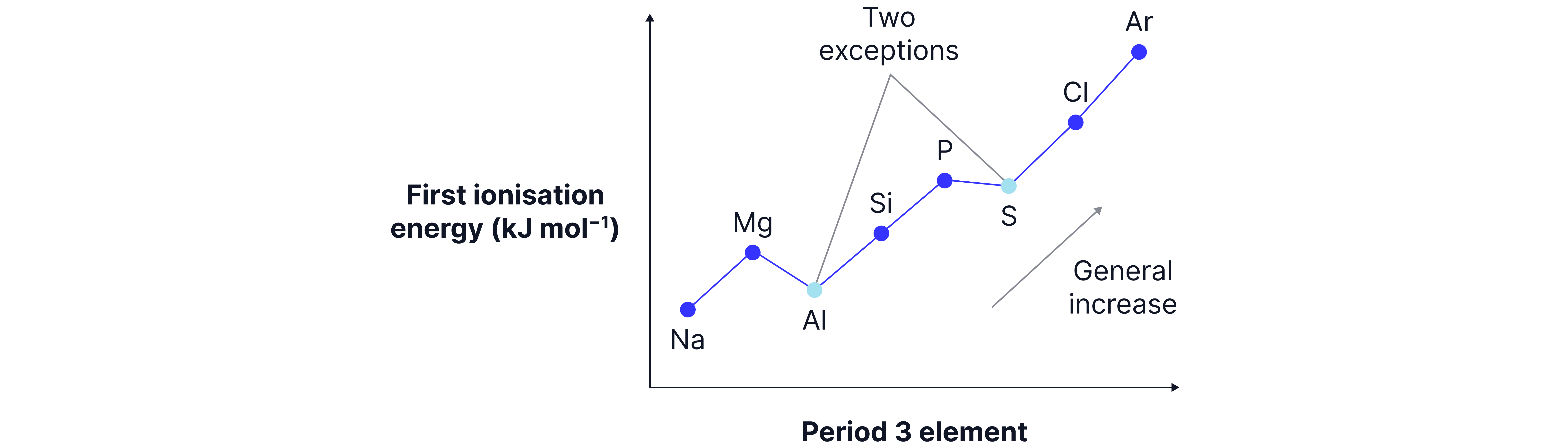
There are two anomalies.
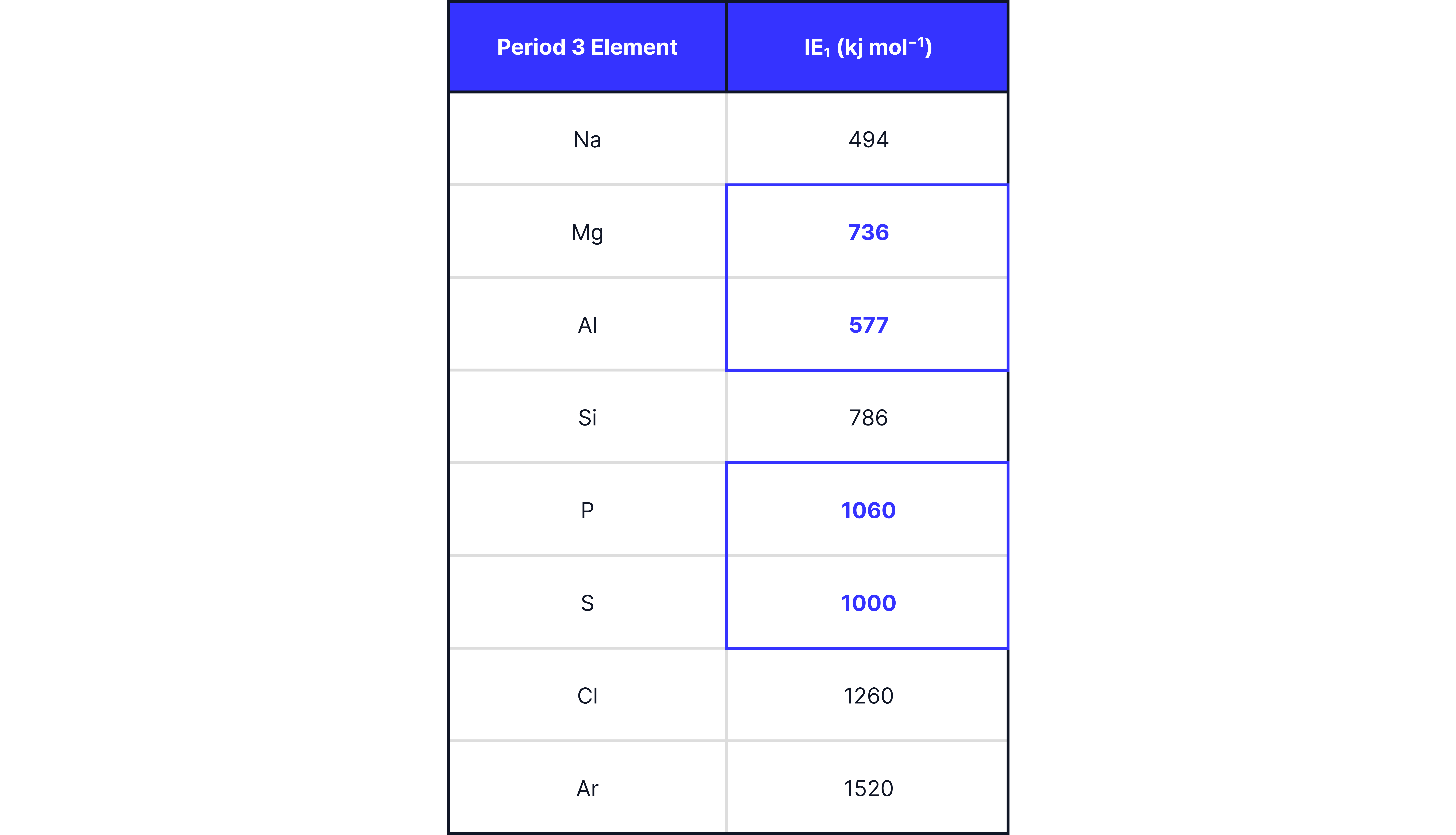
Anomalies:
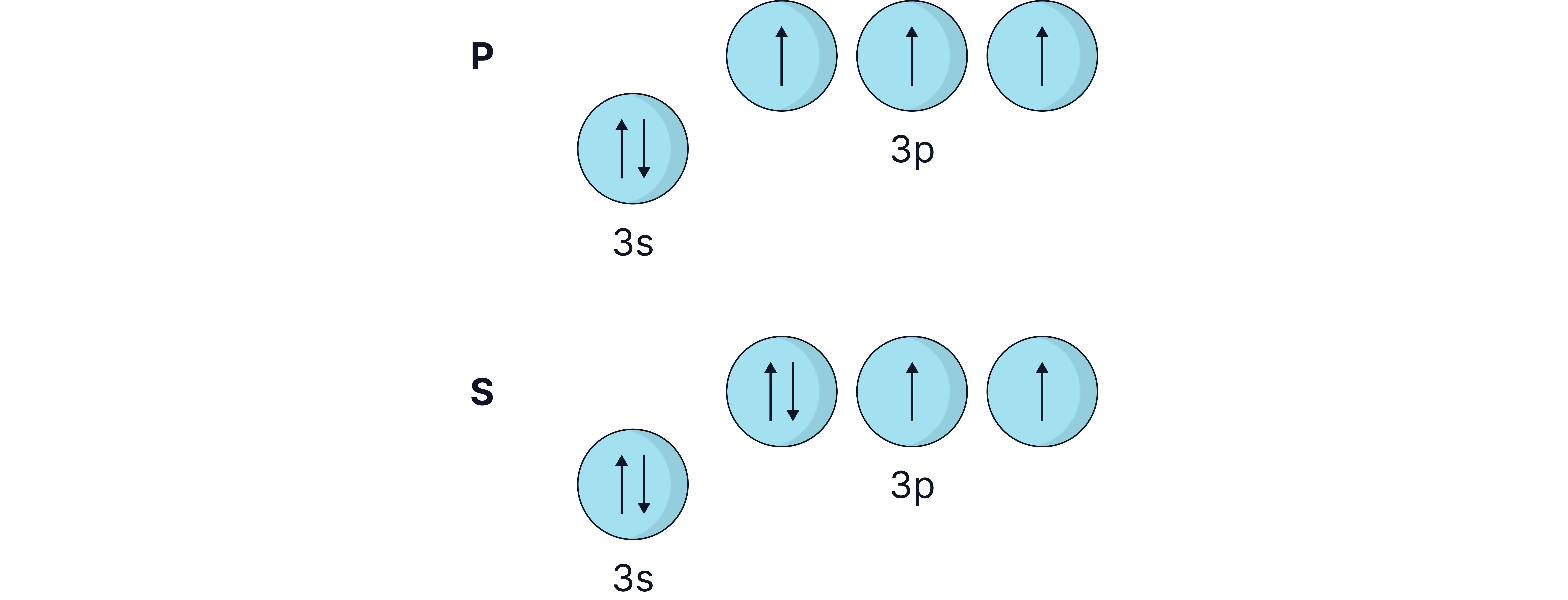
Drop from to :
’s outer electron is in a 3p subshell, which is higher in energy than ’s 3s.
This means less energy is required to remove Al’s electron than ’s
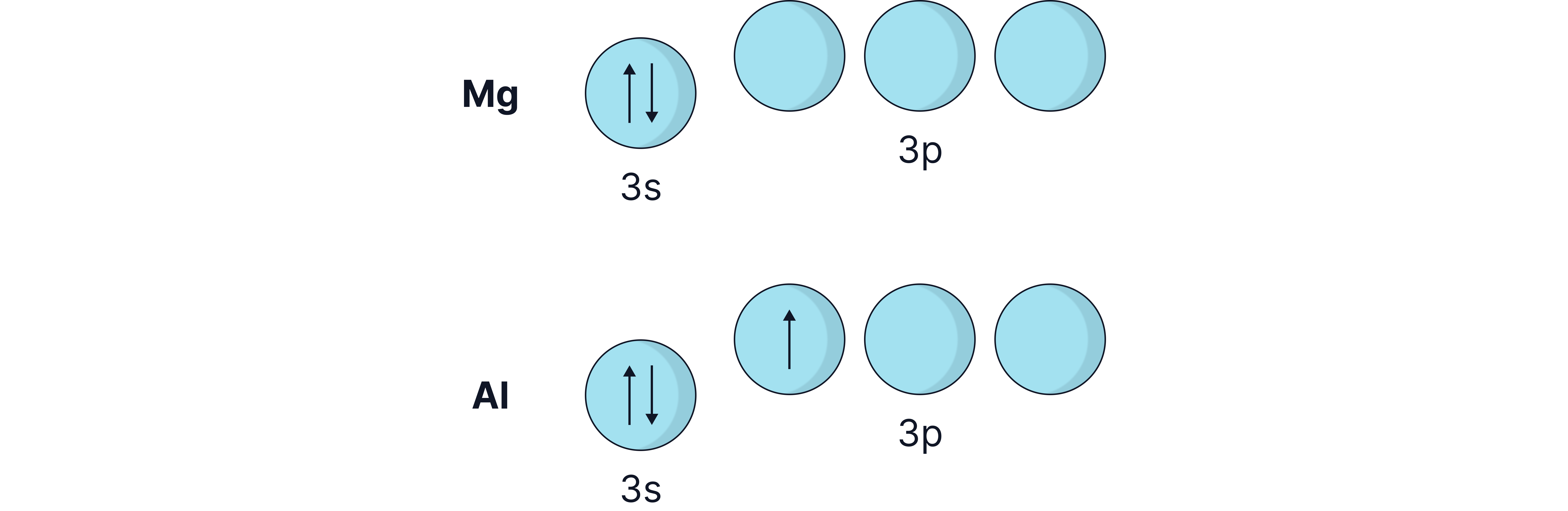
Drop from P to S:
S has paired electrons in a 3p orbital, causing electron-electron repulsion, making it easier to remove.
Melting Points
Melting points depend on the type of structure and bonding present
The table below explains the melting point of the elements in terms of their structure and bonding
Summary - Graphs highlighting trends across period 3
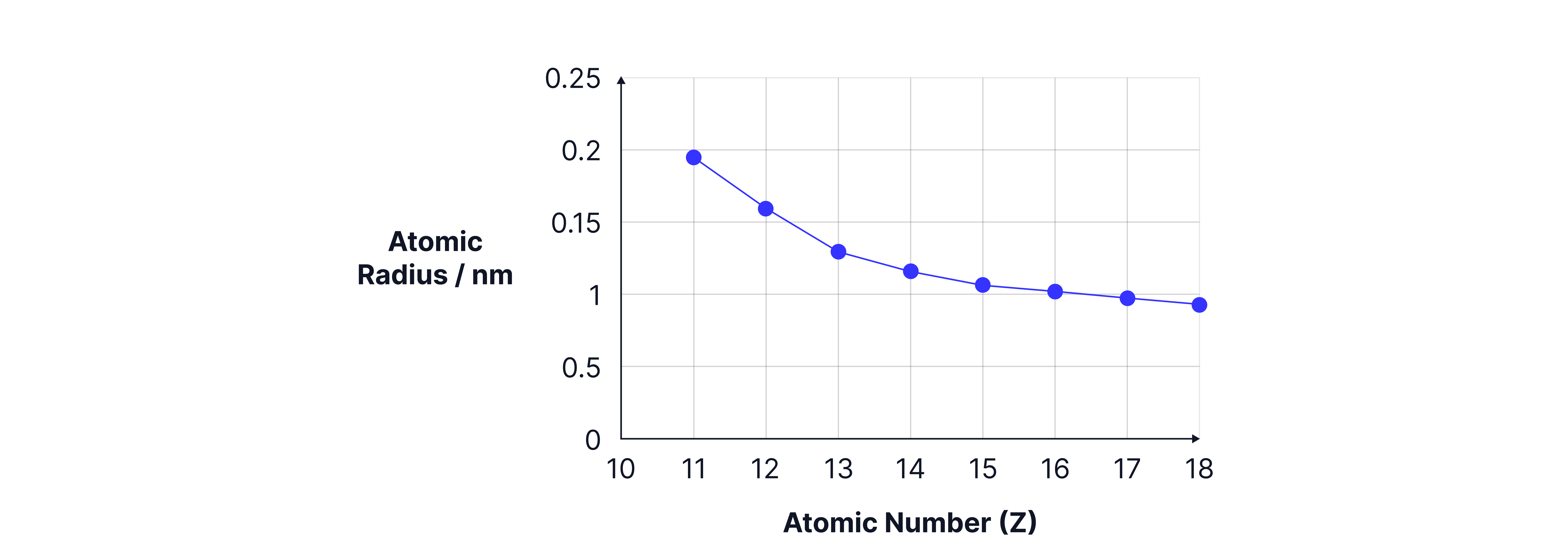
Atomic Radii Graph
The graph shows a decrease in the atomic radius of elements across period 3
Ionisation Energy Graph
The graph shows a general increase with drops at and
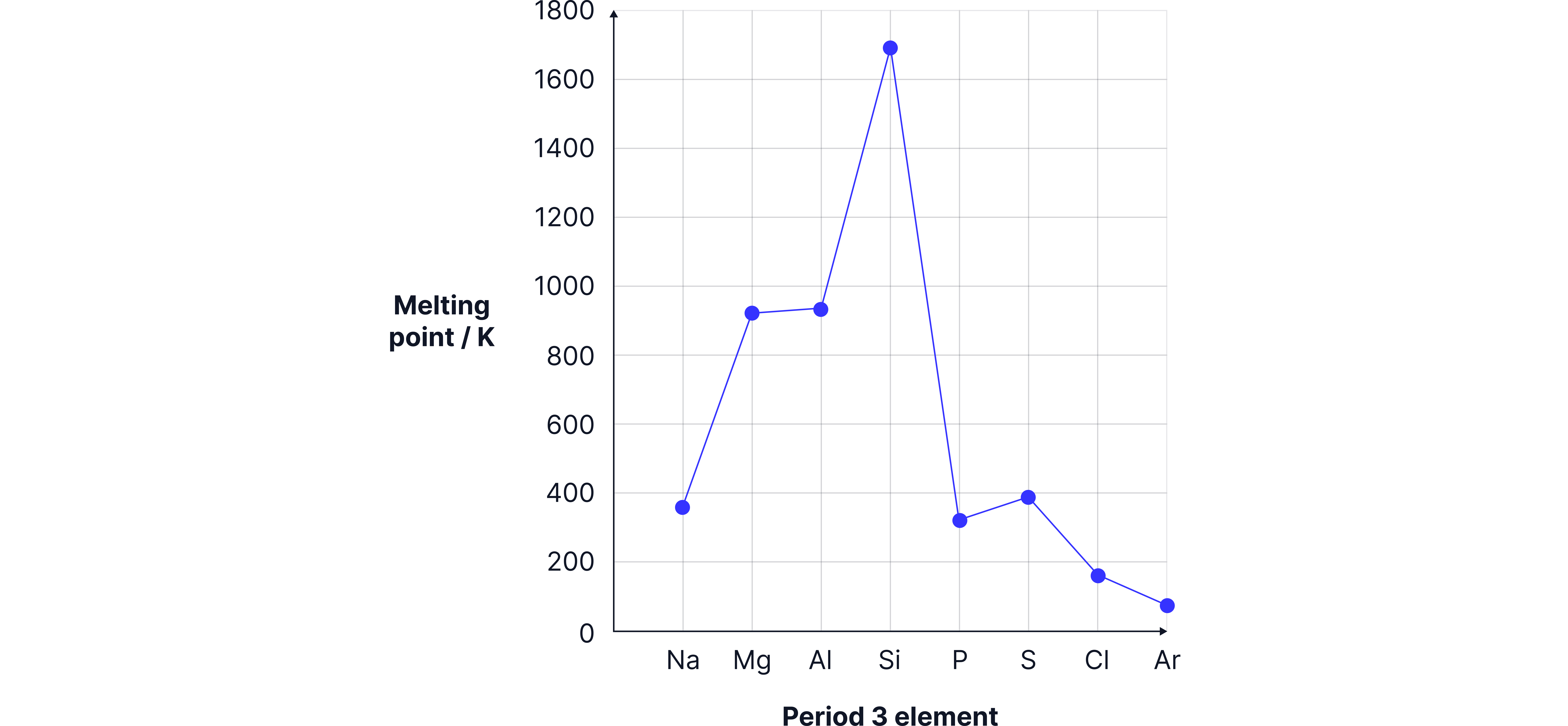
Melting Point Graph
Melting point rises from to , then drops sharply and gradually decreases from to
Key Tips and Reminders
For melting point questions, always relate to structure and the strength of the bonds/forces holding that structure together.
Trends repeat across periods. If the question asks about period 2, your explanations will be the same!