Weak Acid and Ka
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Weak acids only partially dissociate in water.
Equilibrium is established between undissociated acid molecules and ions.
For a weak acid, HA, dissociating as:
OR
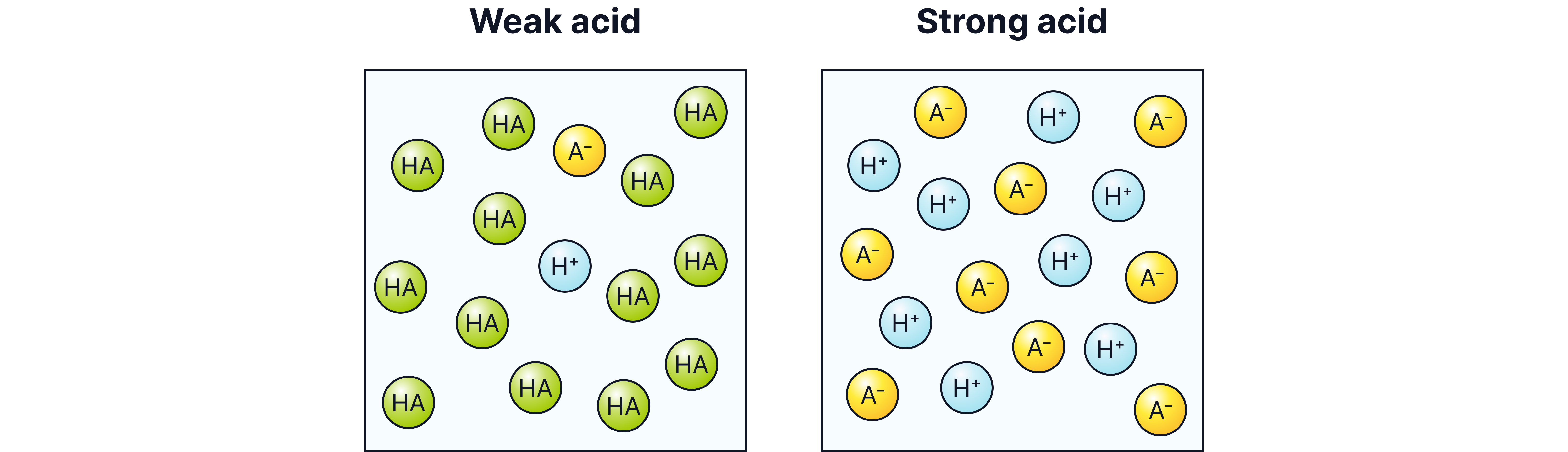
Unlike strong acids, weak acids do not release all their H⁺ ions in solution.
Ka — Acid Dissociation Constant
Ka is the acid dissociation constant, a measure of the extent of dissociation of a weak acid.
The Ka expression is:
Where:
= concentration of hydrogen ions
= concentration of conjugate base
= concentration of undissociated acid
Units of Ka are typically
The more dissociation a weak acid undergoes, the greater the concentrations of and and therefore the greater the value of Ka.
Example: Ethanoic Acid
For ethanoic acid :
= concentration of hydrogen ions
= concentration of conjugate base
= concentration of undissociated acid
pKa — Logarithmic Form of Ka
To compare Ka values more easily, we often use pKa:
To convert back:
Lower pKa = stronger acid (more dissociation)
Higher pKa = weaker acid (less dissociation)
Calculating the pH of a Weak Acid
A few assumptions must be made before using the Ka expression to calculate and therefore pH.
1. Dissociation of the weak acid HA is negligible. This means that and the given acid concentration can be used in calculations.
2. Dissociation of water is negligible. This means that all the ions in the solution come from the acid and not from the partial dissociation of water allowing the simplification ,
This lead to the simplified expression:
This is used to calculate Ka or when dealing with weak monoprotic acids.
Worked Example
A weak acid has and . Calculate its pH.
Answer
Use K
Worked Examples
Writing the Ka Expression
Write the expression for Ka for nitrous acid,
Answer
Calculating Ka from Concentration and pH
A solution of a weak acid has a pH of . Calculate the value of Ka.
Answer
Step 1: Calculate
Assume and
Converting Ka to pKa
If , Calculate pKa.
Answer
Summary Table
Concept | Formula |
Ka expression | |
Simplified Ka | |
pKa from Ka | |
Ka from pKa |
Practice Questions
1. Write the Ka expression for the dissociation of methanoic acid,
Answer:
2. A solution of a weak acid has a pH of 3.40. Calculate Ka.
Step 1:
3. If , calculate pKa.
4. A weak acid has and a concentration of . Calculate and pH.