Cathode rays
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
What are cathode rays?
Cathode rays are a stream/beam that travels away from the cathode (negative electrode) towards the anode (positive electrode) in an evacuated (or low-pressure) tube when a high voltage is applied.
Early experiments showed these “rays” behave like particles and are the same regardless of the gas used. They were later identified as electrons (negatively charged particles).
Evidence that cathode rays travel in straight lines (Maltese cross)
Set-up: A Maltese cross is placed in the path of the cathode rays inside the tube, with a fluorescent/glowing screen at the far end.
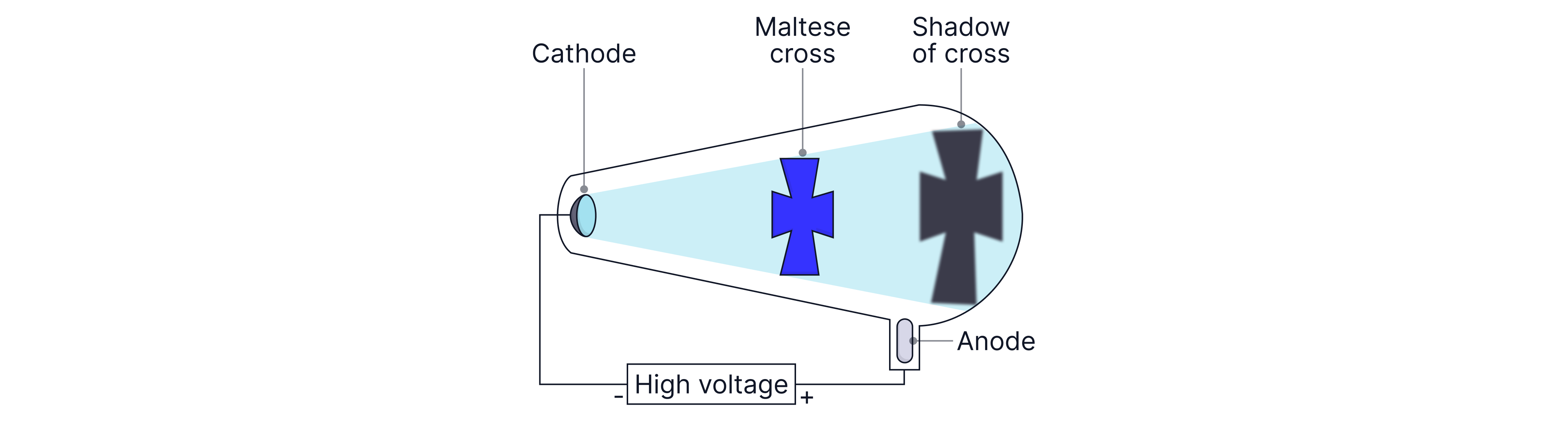
Observation: A sharp shadow of the cross appears on the screen.
Conclusion: Cathode rays must be travelling in straight lines from the cathode to the screen.
Evidence that cathode rays are particles with momentum (paddle wheel)
Set-up: A very light paddle wheel is placed so that the cathode ray beam strikes the paddles.
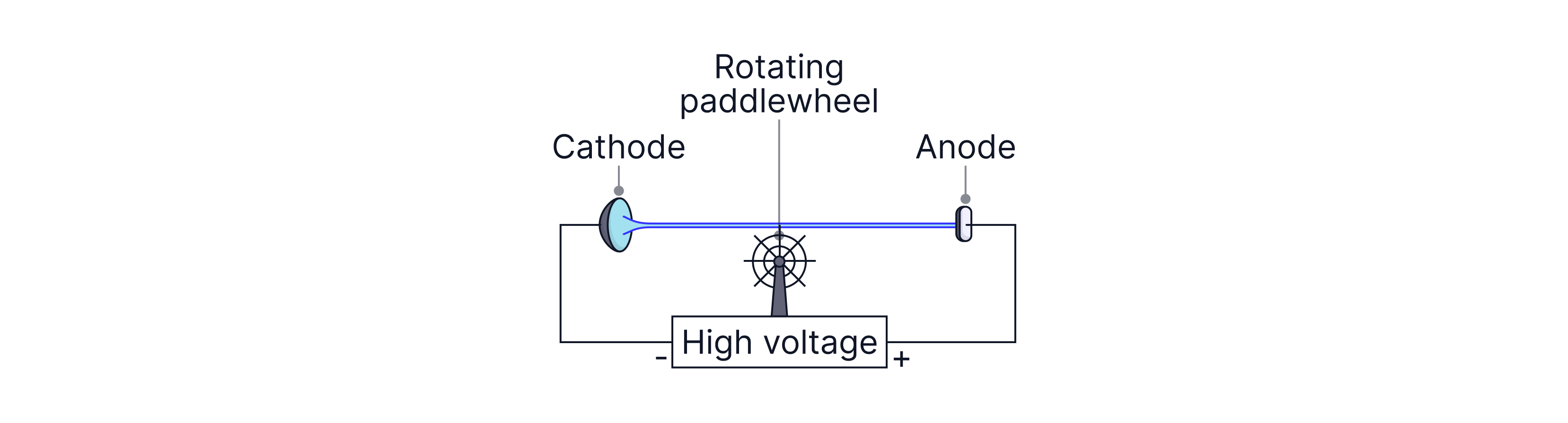
Observation: The wheel rotates when the tube operates.
Conclusion: The beam must transfer momentum (and kinetic energy), so cathode rays behave like particles.
Evidence cathode rays are charged (deflection by electric and magnetic fields)
Set-up: A cathode ray beam is produced in a tube. The beam passes through a region where an electric field can be applied (e.g. between charged plates). A magnetic field can also be applied across the beam using magnets/coils.
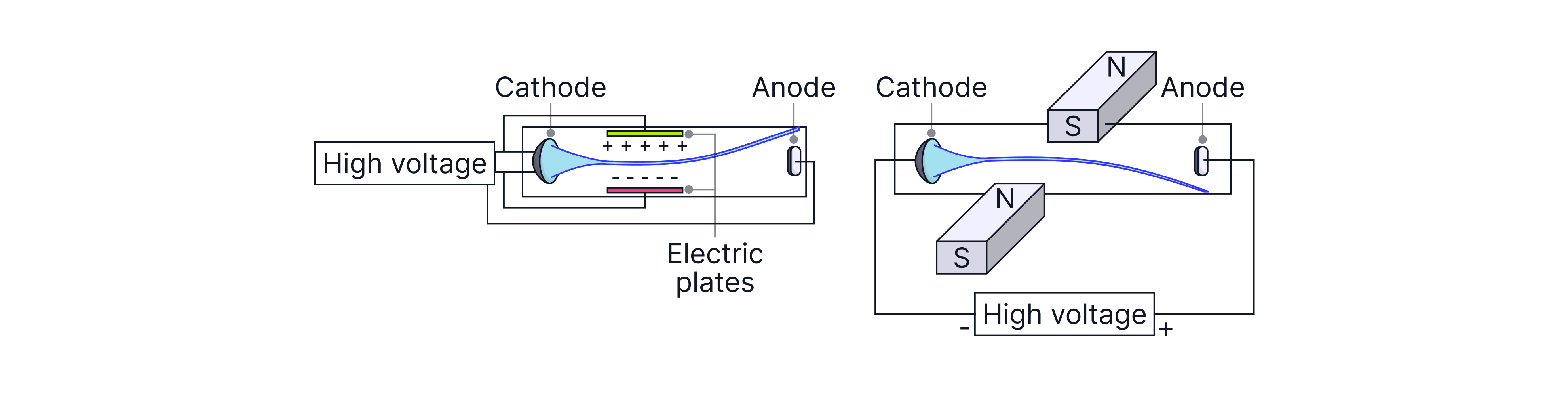
Observation:
With an electric field, the beam bends toward the positive side.
With a magnetic field, the beam curves, and the direction changes if the magnetic field direction is reversed.
Conclusion: Electric deflection shows the beam is charged, and bending toward the positive plate shows it is negative. Magnetic deflection shows the particles are moving charges. Overall: cathode rays are moving negative particles (electrons).
Producing and accelerating electrons
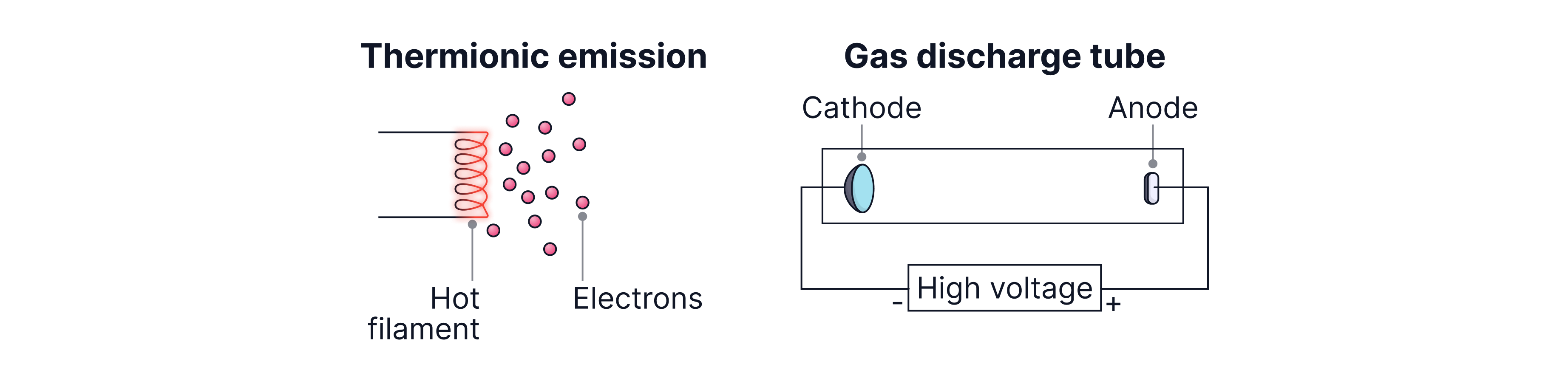
Electrons can be produced in two key ways:
Gas discharge / ionization tubes: At low pressure, a high voltage produces a strong electric field which pulls the electrons out of the gas atoms. (i.e. ionises the gas). The freed electrons are then accelerated towards the anode by the electric field.
Thermionic emission: Heating a metal cathode gives electrons enough energy to escape the surface.
Kinetic energy gained from an accelerating potential difference:
Once electrons are available, a potential difference accelerates them. Assuming the electrons starts at rest, we can find the speed using:
Glowing discharge tubes
The glow in a discharge tube happens because fast electrons collide with gas atoms.

• High voltage creates some free electrons (ionisation as mentioned above).
• These electrons accelerate and collide with gas atoms, causing excitation (atomic electrons move to higher energy levels).
• When atoms de-excite, they emit photons (light) — many emissions produce the visible glow.
Pressure effect: more gas (higher pressure) causes more frequent collisions (brighter glow), but the beam becomes less sharp because collisions scatter the electrons. Very low pressure gives a clearer beam and sharper shadows/deflection.
Worked Example
Electrons are accelerated from rest to a speed of . Calculate the accelerating potential difference .
Worked Example
A student investigates cathode rays using four discharge tubes:
Tube 1 contains a Maltese cross
Tube 2 contains a paddle wheel
Tube 3 passes the beam through an electric field region
Tube 4 passes the beam through a magnetic field
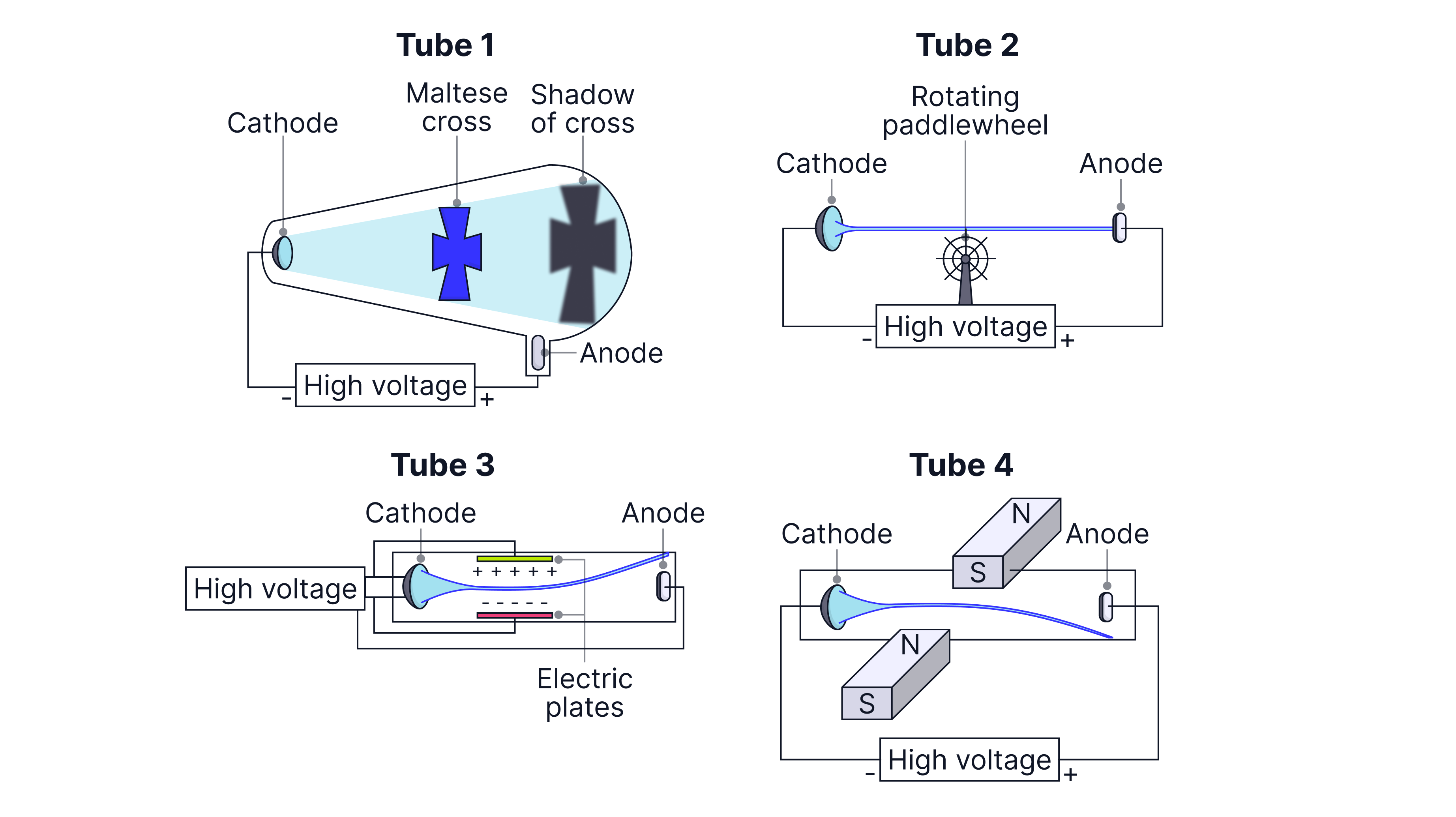
Describe the observation in Tube 1 and what it shows about the path of cathode rays.
Explain how Tube 2 supports the conclusion that cathode rays are particles.
In Tube 3 the beam deflects toward the positive side in the electric field region. What does this show about the charge of cathode rays?
The beam is also deflected by a magnetic field in tube 4. Explain why this supports the idea that cathode rays are moving charged particles.
State two reasons why the tube is evacuated / at very low pressure.
Sharp shadow; rays travel in straight lines.
Wheel rotates; momentum transfer → particle behavior.
Deflects toward positive plate → cathode ray is negative charge.
Magnetic deflection only for moving charges → moving charged particles.
Fewer collisions / clearer beam / less scattering / longer mean free path.
Practice Questions
What is the evidence that cathode rays are made of electrons?
-> Check out Hannah's video explanation for more help.
Answer:
Observation: A Maltese cross produced a sharp shadow.
Conclusion: Cathode rays travel in straight lines.
Observation: A paddle wheel moved / spun when placed in the beam.
Conclusion: Cathode rays transfer momentum / have kinetic energy / behave like particles.
Observation: Cathode rays were deflected by electric or magnetic fields.
Conclusion: Cathode rays are charged.
Observation: Cathode rays were deflected towards the positive plate.
Conclusion: Cathode rays are negatively charged.
How do we produce a controlled beam of electrons?
What potential difference is required to accelerate electrons to a speed of ?
-> Check out Hannah's video explanation for more help
Answer:
Cathode is heated.
Electrons are released from the metal surface.
This is thermionic emission.
A potential difference is applied between cathode and anode.
Electrons are accelerated by the electric field towards the anode.
Energy gained by the electrons is , so for an electron , and this becomes kinetic energy: .