Required Practical 2 - Titrations Strong Acid + Strong Alkali (Triple Only)
Emmanuel Opoku
Teacher

Aim
To determine the reacting volumes of solutions of a strong acid and a strong alkali using a titration.
Example Reaction:
Scientific Background
A titration is an accurate practical method used to find the exact reacting volumes of an acid and an alkali that react completely (neutralisation).
Purpose:
To find an unknown concentration of an acid or alkali.
To check the purity or strength of a solution.
Type of reaction: Neutralisation
Apparatus
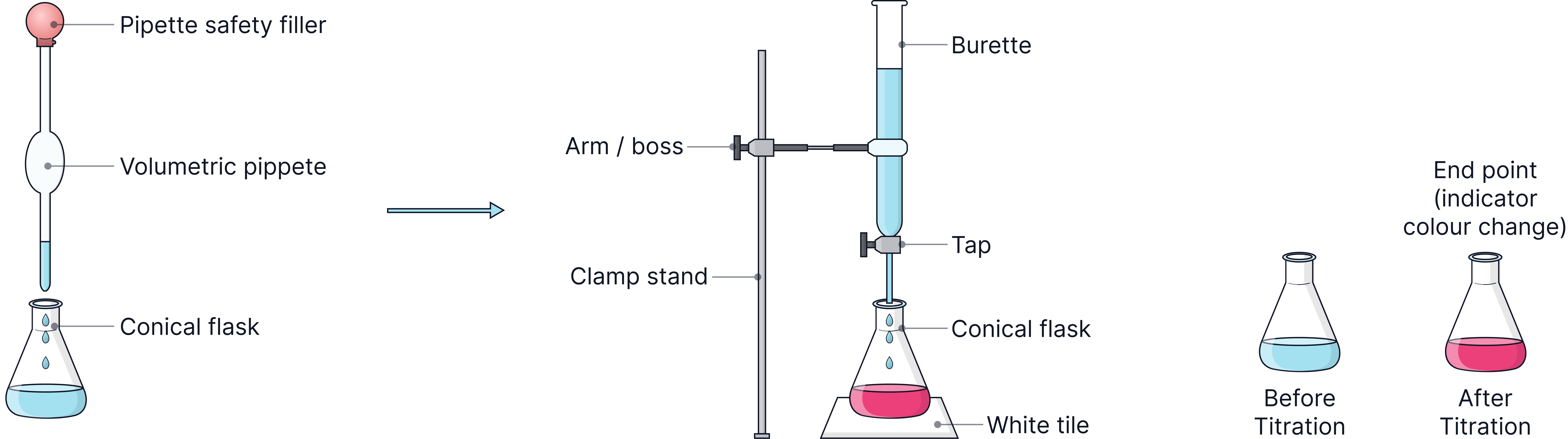
Equipment | Purpose |
Burette | Delivers variable volumes accurately (±0.1 cm³). |
Volumetric Pipette | Delivers a fixed volumes accurately. |
Pipette filler | Used to safely draw liquid into the pipette. |
Conical flask | Holds the alkali; can be swirled without spillage. |
White tile | Helps observe the colour change at the end-point clearly. |
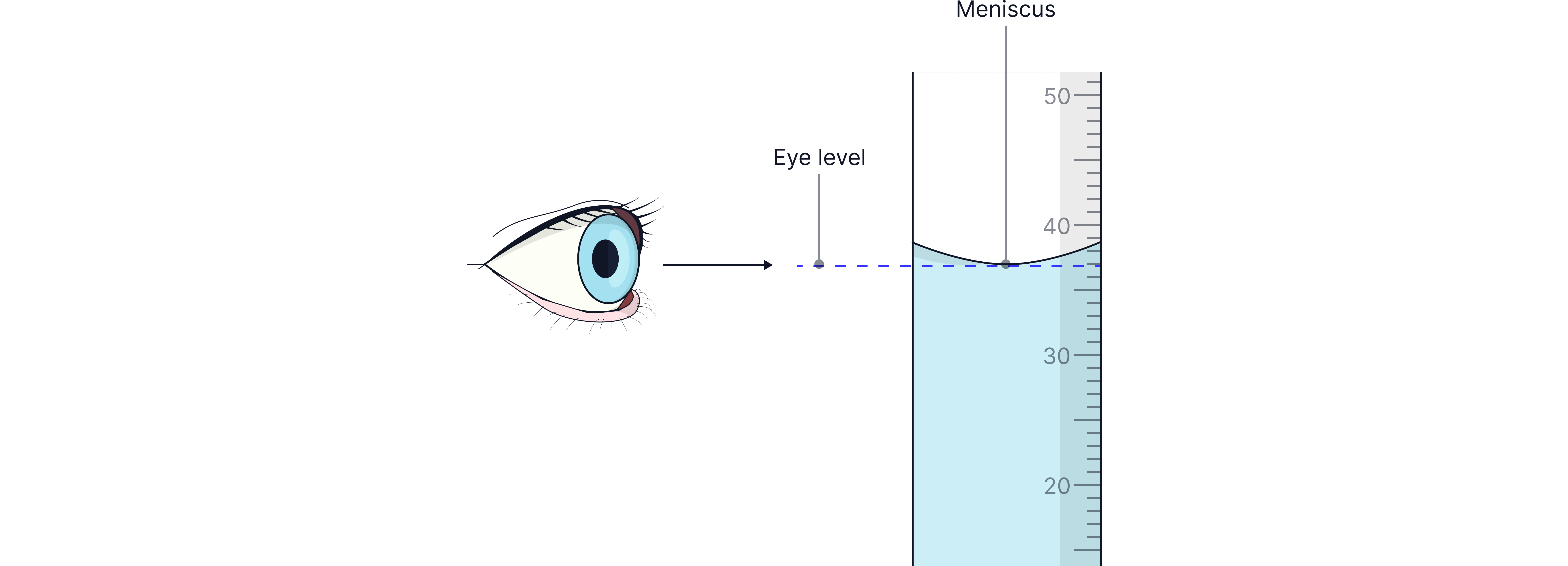
Always read burette and pipette measurements at eye level from the bottom of the meniscus to avoid parallax error – an error caused by not reading at eye level.
Method
1) Use a pipette and filler to add 25.0 cm³ of alkali into a conical flask.
2) Add a few drops of indicator (phenolphthalein or methyl orange).
3) Place the flask on a white tile for clear visibility.
4) Fill a burette with the acid and note the starting reading.
5) Add acid slowly while swirling until the indicator changes colour - this is the end-point.
6) Record the final burette reading.
7) Repeat the titration until results are concordant (within 0.10 cm³).
8) Calculate the mean titre using concordant results only.
This gives an accurate value for the volume of acid required to neutralise the alkali.
Common Indicators
Indicator | In Acid | In Alkali | End-Point Colour Change (acid (in burette) added to alkali (in conical flask)) |
Phenolphthalein | Colourless | Pink | Pink → Colourless |
Methyl Orange | Red | Yellow | Red → Orange |
Universal indicator is not suitable as it has a gradual colour change, not a sharp end-point.
Note that the colour changes will be the other way round if alkali is being added to acid.
Concordant Values
When repeating titrations, concordant values show your results are precise and reliable.
Definition:
Concordant values are titre results that are within 0.10 cm³ (±0.10 cm³) of each other.
Only use concordant titres when calculating the average (mean).
Example
Titration No. | Titre (cm³) |
1 | 24.00 |
2 | 24.50 |
3 | 24.40 |
Titration 2 and 3 are concordant because:
Average (mean) titre:
Concordant results improve accuracy and reduce random error.
If results differ by more than 0.10 cm³, they are not concordant.
Example of Titration Data
Trial | Initial (cm³) | Final (cm³) | Titre (cm³) |
1 | 0.50 | 24.50 | 24.00 |
2 | 2.50 | 27.00 | 24.50 |
3 | 1.55 | 25.95 | 24.40 |
Concordant results: 24.50 cm³ and 24.40 cm³
Mean titre: 24.45 cm³
Accuracy & Good Practice
Add acid dropwise near the end-point.
Swirl continuously for even mixing.
Record burette readings to 2 decimal places (e.g. 24.45 cm³).
Repeat until concordant results (±0.10 cm³) are obtained.
Rinse glassware with distilled water, not tap water, between uses.
Calculations in Titrations (HT Only)
Once the mean titre is found, Higher Tier students must calculate the unknown concentration using:
(where n= moles, c= concentration in mol/dm³, v= volume in dm³)
Example 1 (HT Only)
25.0 cm³ of HCl is neutralised by 22.4 cm³ of 2.00 mol/dm³ NaOH.
Find the concentration of HCl.
1) Moles of NaOH:
2) Ratio (HCl : NaOH) = 1 : 1
So, moles of HCl = 0.0448 mol
3) Concentration of HCl:
Example 2 (HT Only)
What volume of 0.10 mol/dm³ HCl is needed to neutralise 25.0 cm³ of 0.14 mol/dm³ NaOH?
Ratio 1:1 → moles of HCl = 0.0035 mol
Volume of HCl required = 35.0 cm³
General Formulae (HT Only)
Formula | Meaning |
Moles = Concentration × Volume (dm³) | |
Concentration = Moles ÷ Volume | |
Volume = Moles ÷ Concentration |
Volume Conversion (HT Only)
Example: 25.0 cm³ = 25.0 ÷ 1000 = 0.025 dm³
Concentration Conversion (HT Only)
To convert between mol dm⁻³ and g dm⁻³:
Unit analysis:
Example:
If concentration = 0.05 mol dm⁻³ and Mᵣ = 98 g mol⁻¹
→ 0.05 × 98 = 4.9 g dm⁻³
Key Ideas Summary
Concept | Foundation Tier | Higher Tier (HT Only) |
Purpose of titration | To measure reacting volumes of acid and alkali. | To calculate unknown concentrations. |
Indicator used | Phenolphthalein or methyl orange. | Select indicator based on acid/base type. |
Apparatus | Burette, pipette, conical flask, white tile. | Record titres to 2 d.p. and identify concordance. |
Equation | Word equation for neutralisation. | Molar equation n=c×vand stoichiometric ratios. |
Accuracy | Add acid dropwise, swirl, repeat for precision. | Use concordant titres and mean value for reliability. |
Recall
a) What apparatus is used to measure a fixed volume of alkali?
a) Pipette (25 cm³)
b) Why is a conical flask used?
b) It can be swirled without spilling.
c) What colour change does phenolphthalein show?
c) Pink → Colourless (end point).
d) Define concordant values.
d) Results within ±0.10 cm³ of each other, showing high precision.
e) Why is universal indicator unsuitable for titrations?
e) Universal indicator changes through many colours, not a sharp end point.
2) Apply Your Knowledge (HT Only)
f) 25.0 cm³ of NaOH is neutralised by 22.5 cm³ of 0.10 mol/dm³ HCl. Calculate the concentration of NaOH in mol dm³.
f)
g) Write the ionic equation for all titration neutralisation reactions.
g)
3) Challenge (HT Only)
h) 25.0 cm³ of HCl reacts with 24.0 cm³ of 0.20 mol/dm³ NaOH. Calculate the concentration of HCl in g dm³.
h)
n(NaOH) = 0.20 × (24.0 ÷ 1000) = 0.0048 mol
NaOH : HCl = 1 : 1 → n(HCl) = 0.0048
c = n ÷ v = 0.0048 ÷ (25 ÷ 1000) = 0.192 mol/dm³
Concentration in g dm³ = 0.192 × 36.5 = 7.01 g dm³