Water purification
Emmanuel Opoku
Teacher

Aim
Testing, analysing and purifying water samples from different sources
(including pH testing, dissolved solids, distillation and comparing different water sources)
General Scientific Background
1. What Is Potable Water?
Potable water is safe to drink, but not necessarily pure.
Potable water must have:
Low levels of dissolved salts
No harmful microbes
pH close to neutral (≈7)
Potable water is created by removing toxic substances, microbes, and excess ions.
2. Sources of Water
Freshwater (rivers, lakes)
Usually contains low levels of dissolved salts.
Requires filtration and sterilisation (chlorine, ozone or UV).
Seawater
Very high salt content.
Requires distillation or reverse osmosis, both energy-intensive.
Wastewater (sewage, industrial water)
Contains organic matter, microbes, toxic substances.
Requires multiple stages of treatment before safe use.
3. The Practical – Testing and Purifying Water
You will:
1. Measure pH
2. Test for dissolved solids
3. Distil the water to remove salts
4. Retest the purified water
Apparatus
For Testing Water Samples
Beaker (50/100 cm³)
Measuring cylinder (10 cm³ and 50 cm³)
pH meter (calibrated) or pH paper or Universal Indicator
Evaporating basin
Bunsen burner + heatproof mat
Tripod + gauze
Balance (2 d.p. accuracy preferred)
For Testing Ions
Dropping pipettes
Test tubes
Nitric acid (dilute)
Silver nitrate solution
Nichrome wire loop (for flame tests)
Bunsen burner
For Distillation
Round-bottom flask
Condenser (Liebig condenser)
Delivery tube
Beaker to collect distillate
Stand + clamp
Heat source (Bunsen burner or electric heater)
Method
Part A – Testing the Original Water Sample
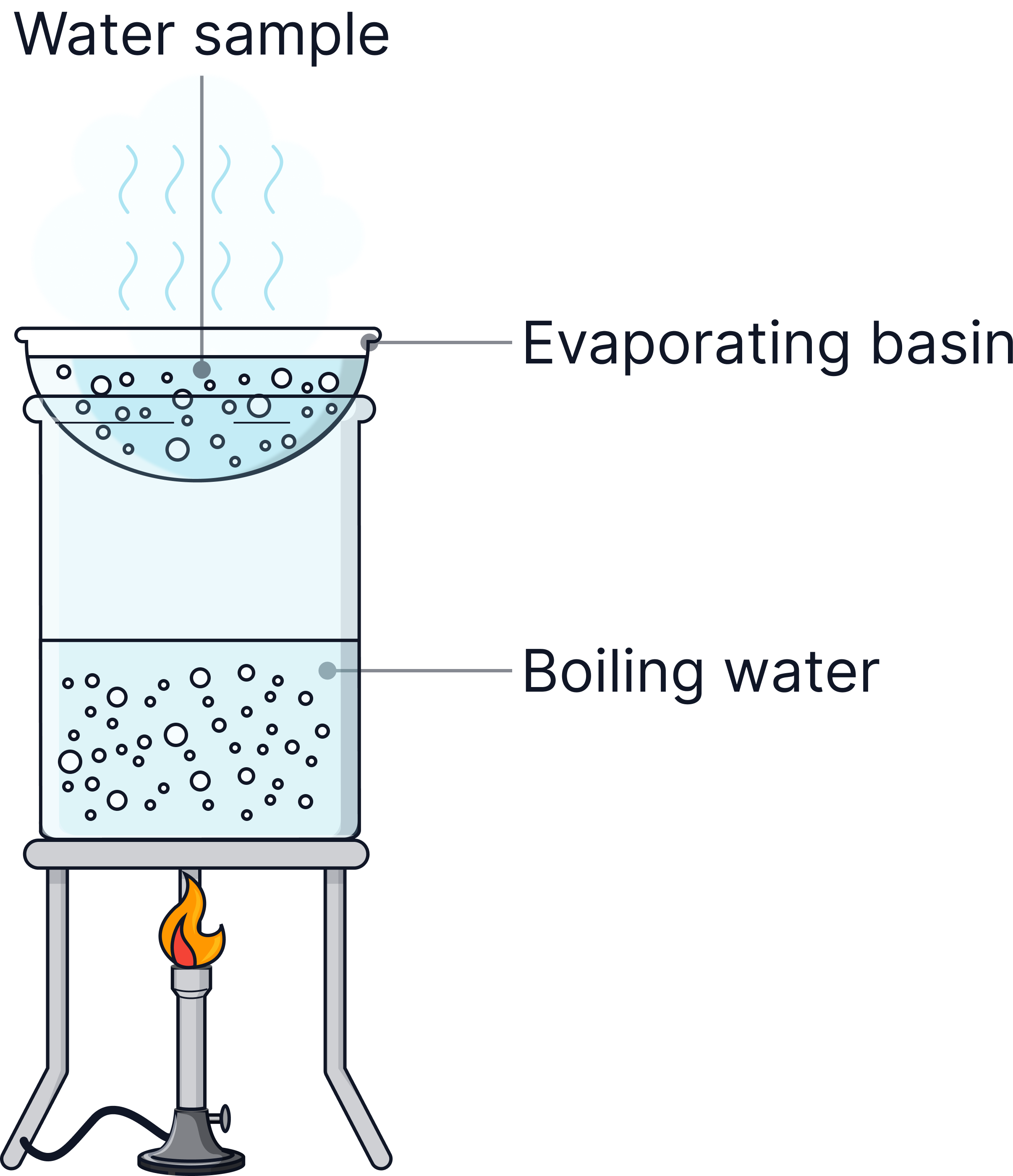
1) Pour roughly 10 cm³ of your water sample into a beaker.
2) Measure its pH using a pH meter (more accurate than indicator).
3) Test for dissolved solids:
Weigh an empty evaporating basin.
Add 5 cm³ of the water sample.
Heat gently using a water bath until all water evaporates.
Reweigh the basin → increase in mass = mas of dissolved solids.
Part B – Testing for chloride and sodium ions – HT ONLY
1) Test for chloride ions (Cl⁻):
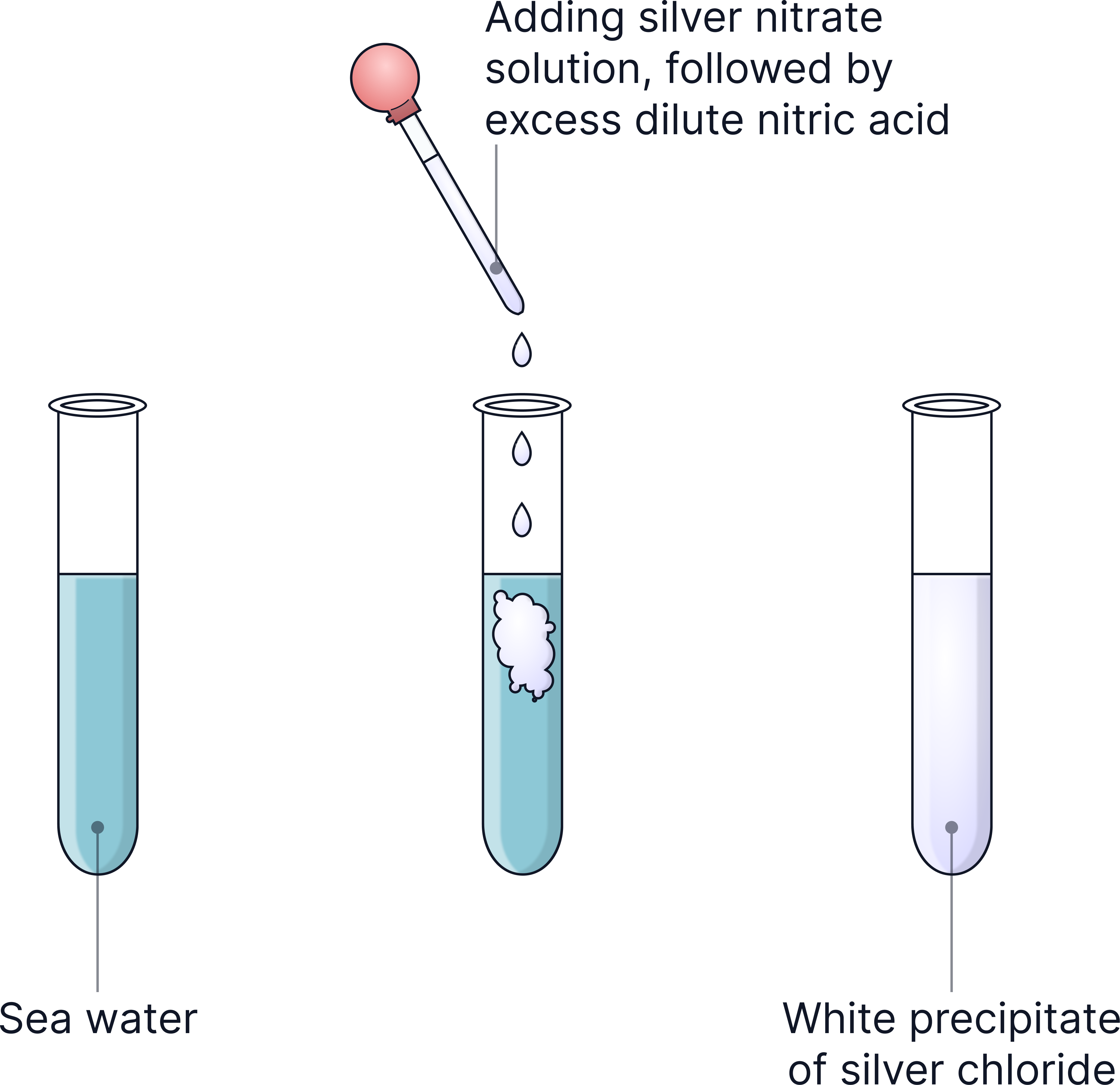
Add nitric acid (removes other ions).
Add silver nitrate solution.
White precipitate = chloride ions present.
2) Test for sodium ions (Na⁺):
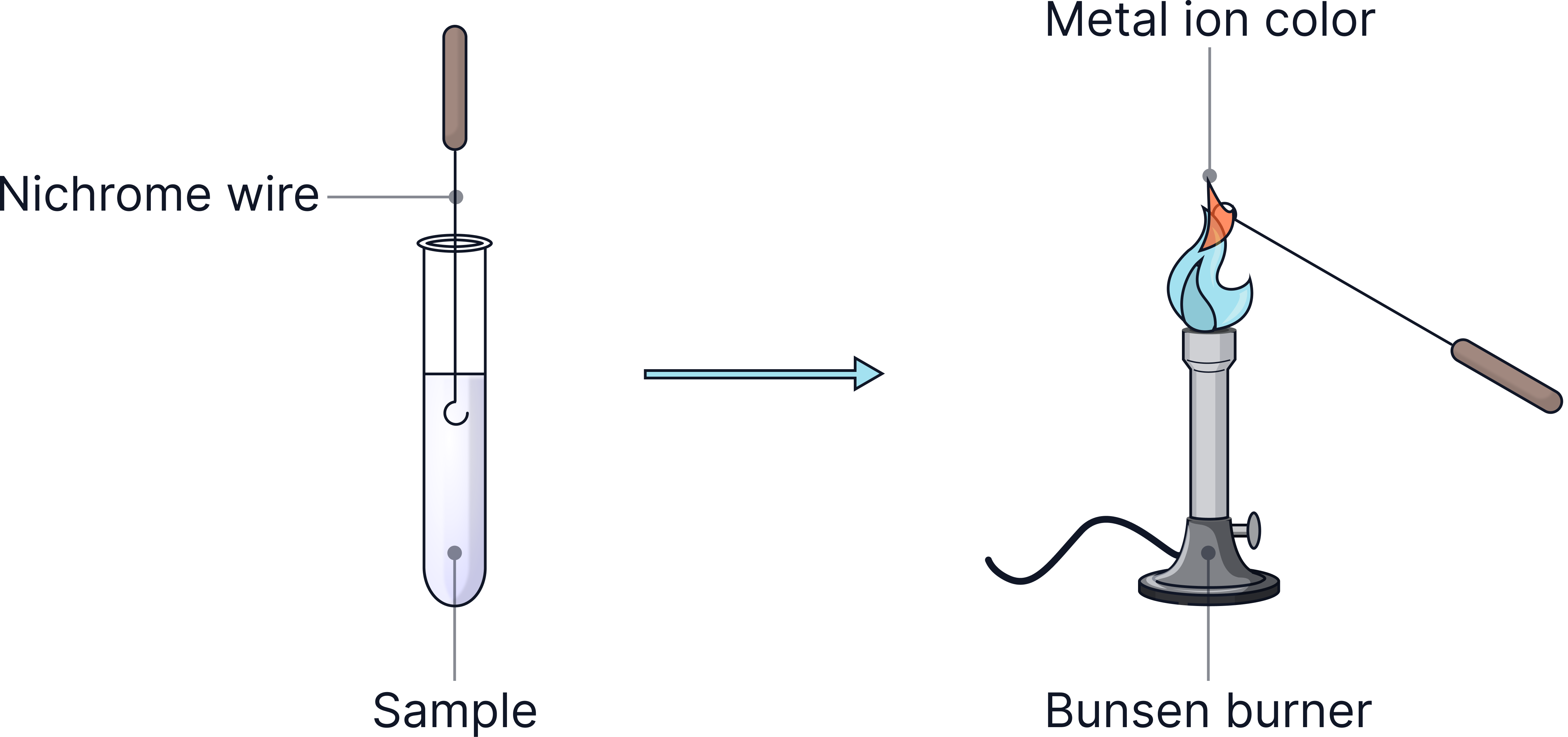
Do a flame test → yellow flame = sodium present.
Part C – Purifying the Water (Distillation)
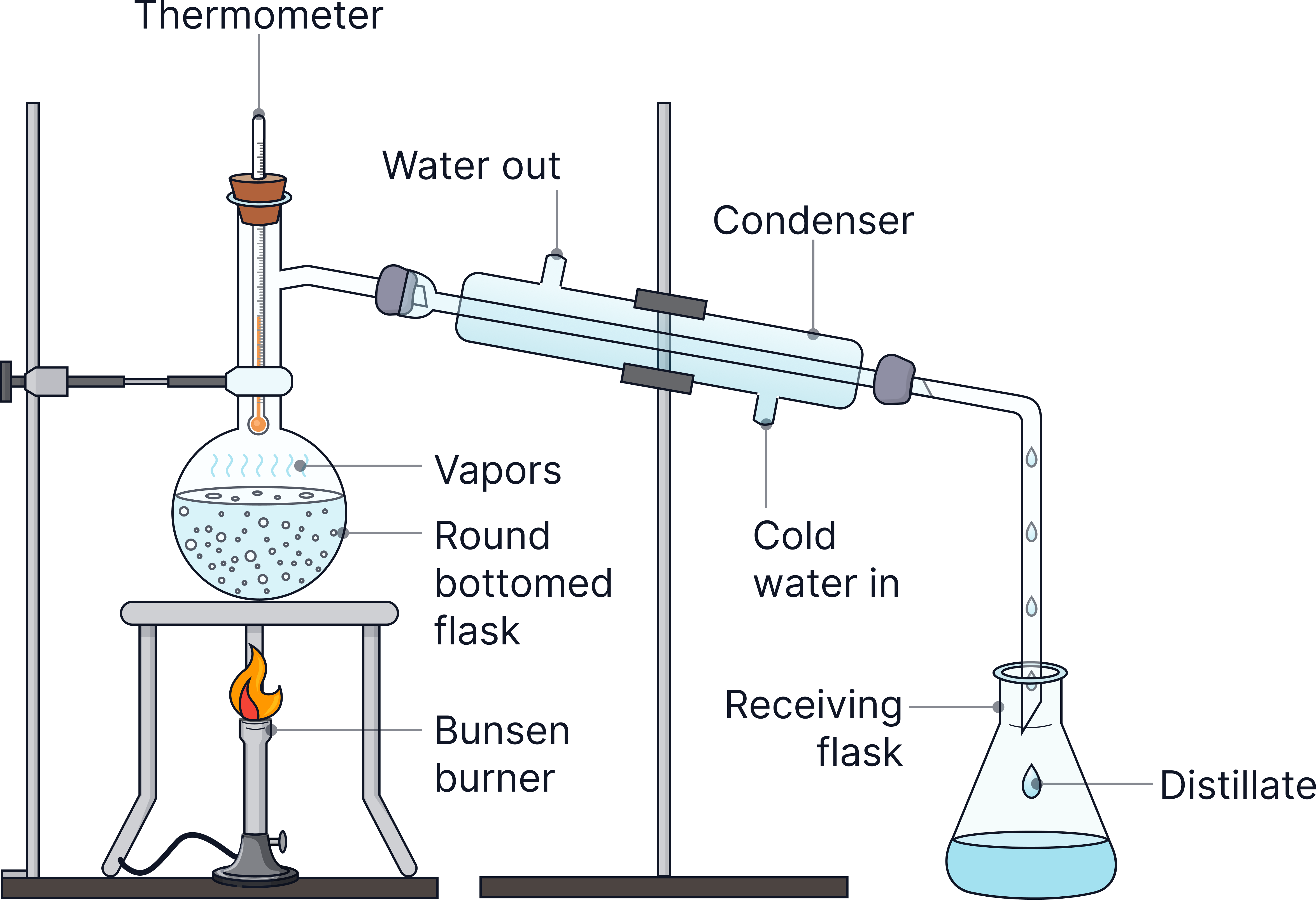
1) Pour the sample into a round-bottom flask.
2) Heat the flask → water boils → steam rises.
3) Steam enters the condenser → cools → condenses into pure water.
4) Collect the distilled water in a beaker.
5) Retest the pH and dissolved solids of the distilled water from Part A.
Example Results
pH results
Water Sample | Initial pH | Final pH (after distillation) |
|---|---|---|
Tap Water | 7.3 | 7.0 |
Sea Water | 8.2 | 7.0 |
Pond Water | 6.4 | 7.0 |
Dissolved Solids
Water Sample | Mass of basin (g) | Mass after heating (g) | Increase (g) |
|---|---|---|---|
Tap Water | 35.00 | 35.02 | 0.02 |
Sea Water | 35.00 | 35.89 | 0.89 |
Pond Water | 35.00 | 35.07 | 0.07 |
Distilled Water | 35.00 | 35.00 | 0.00 |
Ion Tests Summary – HT ONLY
Sample | Na⁺ Flame Test | Cl⁻ (AgNO₃ Test) | Conclusion |
|---|---|---|---|
Tap | Yellow | White ppt | Contains sodium chloride traces |
Sea | Strong yellow | Thick white ppt | High salt content |
Pond | No colour | Slight white | Small amount of dissolved ions |
Distilled | No colour | No ppt | Pure water – no chloride ions |
Expected Conclusions
Distillation removes dissolved salts completely.
Distilled water returns to pH ≈ 7.
Sea water contains very high chloride and sodium ion levels – HT ONLY
Tap water contains low but detectable ions – HT ONLY.
Pond water contains some impurities but fewer than seawater.
Sources of Error
Type of Error | Definition | Effect on Results |
|---|---|---|
Random error | Unpredictable changes in readings (e.g., pH meter drifting) | Causes results to scatter; reduces reliability |
Systematic error | Equipment consistently incorrect (e.g., uncalibrated pH meter/mass balance) | Shifts all measurements in one direction → inaccurate |
Random Error (Human error) | Observer mistakes (parallax, mis-reading mass, misinterpreting pH value from pH paper) | Produces incorrect individual values |
Practice Questions
Recall Questions
a) What is potable water?
a) Water that is safe to drink but not necessarily pure; low salts and no harmful microbes.
b) What pH should potable water have?
b) Around 7 but between 6.5 and 8.5 is acceptable.
Application Questions
c) Explain why distillation removes impure solids from a water sample.
c) Distillation removes impure solids because the water boils and evaporates, leaving the dissolved solids behind. The steam is then condensed back into pure liquid water, so the solids do not transfer into the distillate.
d) Why is a pH meter used rather than universal indicator?
d) A pH meter is more accurate, not subjective, and does not contaminate the water.
e) A student evaporates 10 cm³ of water and finds the basin mass increased by 0.25 g.
What does this tell you?
e) Increase = mass of dissolved solids → 0.25 g of salts per 10 cm³.
Challenge Questions – HT Only
f) Explain why silver nitrate is used in chloride testing.
f) It forms a white precipitate with chloride ions → visible evidence of chloride.
g) What does the flame test for sodium show?
g) Gives a yellow flame indicating that sodium ions present.
h) Evaluate distillation as a method for producing potable water in a hot, dry country.
h) Pros: removes all salts/microbes → very pure.
Cons: expensive, requires energy, not sustainable in poorer countries.
i) Explain how systematic errors could affect the pH results in this practical.
i) If the pH meter is not calibrated, all pH values will be consistently too high/too low.
j) Why must nitric acid be added before silver nitrate in the chloride test?
j) To remove carbonate ions, which would otherwise give a false positive.