Identifying ions tests – cations and anions (Triple Only)
Emmanuel Opoku
Teacher

Contents
Aim
Testing solutions of known ionic compounds and using the results to deduce the identity of an unknown ionic compound.
General Overview
Ionic compounds contain positive ions (cations) and negative ions (anions).
Cations tested
Li⁺, Na⁺, K⁺, Ca²⁺, Cu²⁺, Fe²⁺, Fe³⁺, Al³⁺ and Mg²⁺ ions.
Anions tested
CO₃²⁻ (carbonate), SO₄²⁻ (sulphate), Cl⁻, Br⁻ and I⁻ - the halides.
You will use:
Flame tests (positive ions)
Precipitation tests (metal hydroxides)
Gas test for carbonate
Barium chloride test for sulphates
Silver nitrate test for halides
Apparatus and Chemicals
Bunsen burner
Nichrome wire
Test tubes and rack
Dropping pipettes
Bung attached to a delivery tube
Sodium hydroxide solution, NaOH – 0.5 mol dm-3
Dilute hydrochloric acid, HCl – 0.05 mol dm-3
Dilute nitric acid, HNO₃ – 0.5 mol dm-3
Barium chloride solution, BaCl2 – 0.1 mol dm-3
Silver nitrate solution, AgNO₃ – 0.05 mol dm-3
Limewater, Ca(OH)2
Known labelled solutions of ionic compounds
Unknown sample
Safety & Risk Assessment
Wear safety goggles throughout.
Acids are irritants – avoid skin/eye contact.
Silver nitrate stains skin and clothes.
Keep flammables away from the Bunsen flame.
Use the nichrome wire carefully – it becomes red hot.
Tests for Positive Ions
Flame Tests
Metal ion | Flame colour |
|---|---|
Lithium, Li⁺ | Crimson red |
Sodium, Na⁺ | Yellow/Orange |
Potassium, K⁺ | Lilac |
Calcium, Ca²⁺ | Orange-red |
Copper(II), Cu²⁺ | Green |
Method
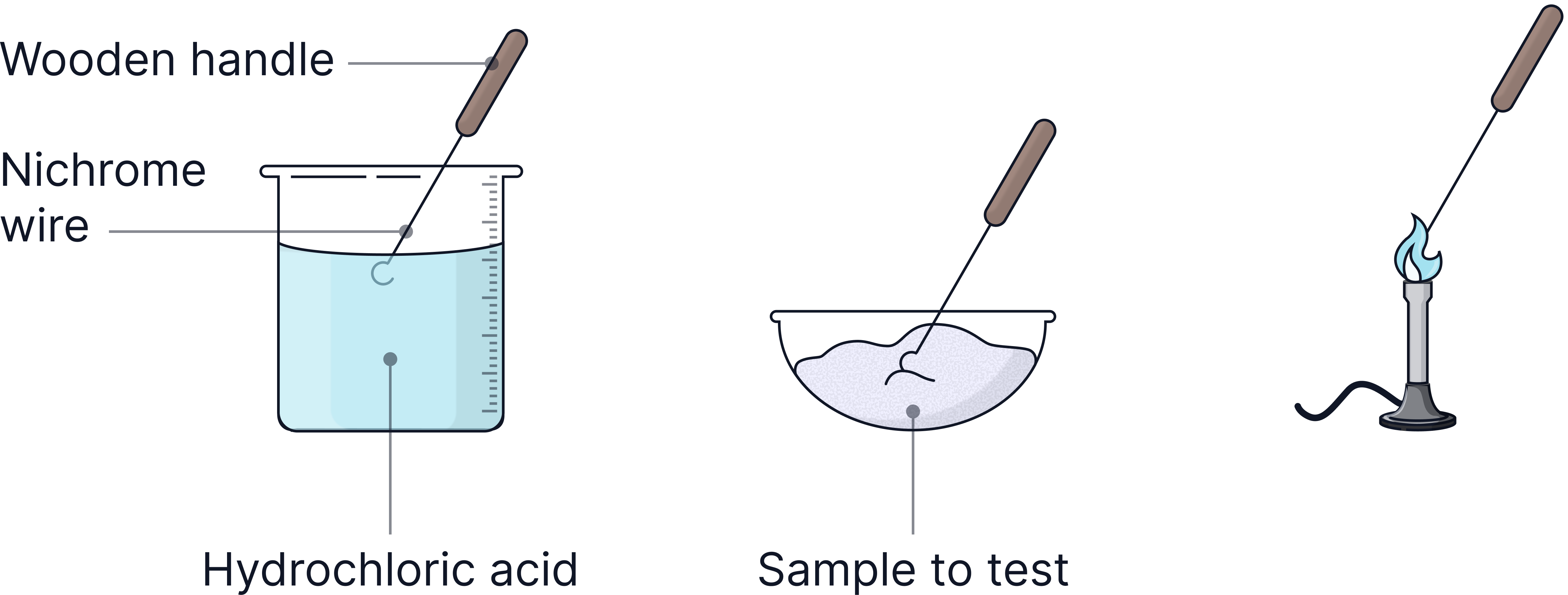
1) Dip a clean Nichrome wire into the solution – cleaned using hydrochloric acid.
2) Hold in blue Bunsen flame.
3) Record flame colour.
4) Clean the wire between tests and repeat for all known solutions of ionic compounds.
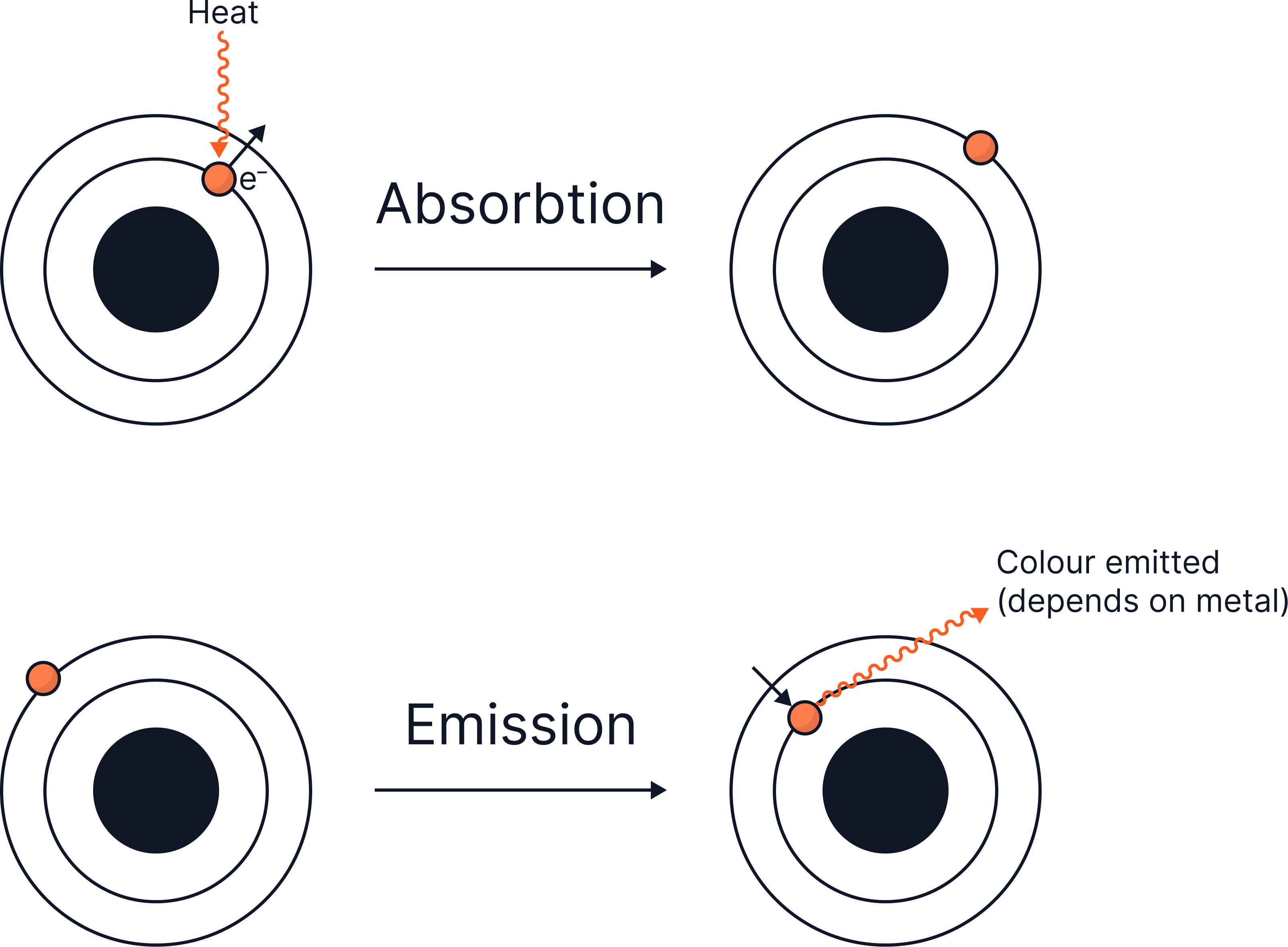
Scientific background:
Heating ions excites electrons and they absorb energy (absorption). When electrons fall back to their ground state – lowest energy level, they emit light of a characteristic wavelength, producing a specific, unique flame colour (emission).
Precipitation Tests for Metal Hydroxides
Ion | Colour of precipitate | Ionic equation |
|---|---|---|
Cu²⁺ | Blue | Cu²⁺ + 2OH⁻ → Cu(OH)₂ |
Fe²⁺ | Green | Fe²⁺ + 2OH⁻ → Fe(OH)₂ |
Fe³⁺ | Brown | Fe³⁺ + 3OH⁻ → Fe(OH)₃ |
Ca²⁺ | White | Ca²⁺ + 2OH⁻ → Ca(OH)₂ |
Mg²⁺ | White | Mg²⁺ + 2OH⁻ → Mg(OH)₂ |
Al³⁺ | White, dissolves in excess NaOH | Al³⁺ + 3OH⁻ → Al(OH)₃ |
Method
1) Pour about 1 cm depth of the sample solution into a clean test tube.
2) Using a dropping pipette, add a few drops of sodium hydroxide (NaOH) solution to the sample.
3) Swirl gently and observe the colour and formation of any precipitate:
4) Add more NaOH (about 10–15 more drops) to check if the precipitate dissolves:
5) Record all observations - note colour, texture (gel-like, grainy, thick), and solubility in excess NaOH.
Scientific background:
Many metal hydroxides are insoluble, forming coloured solids when OH⁻ ions are added. Only the transition metal ions produce coloured precipitates.
Tests for Negative Ions
Carbonate Ions (CO₃²⁻)
Method
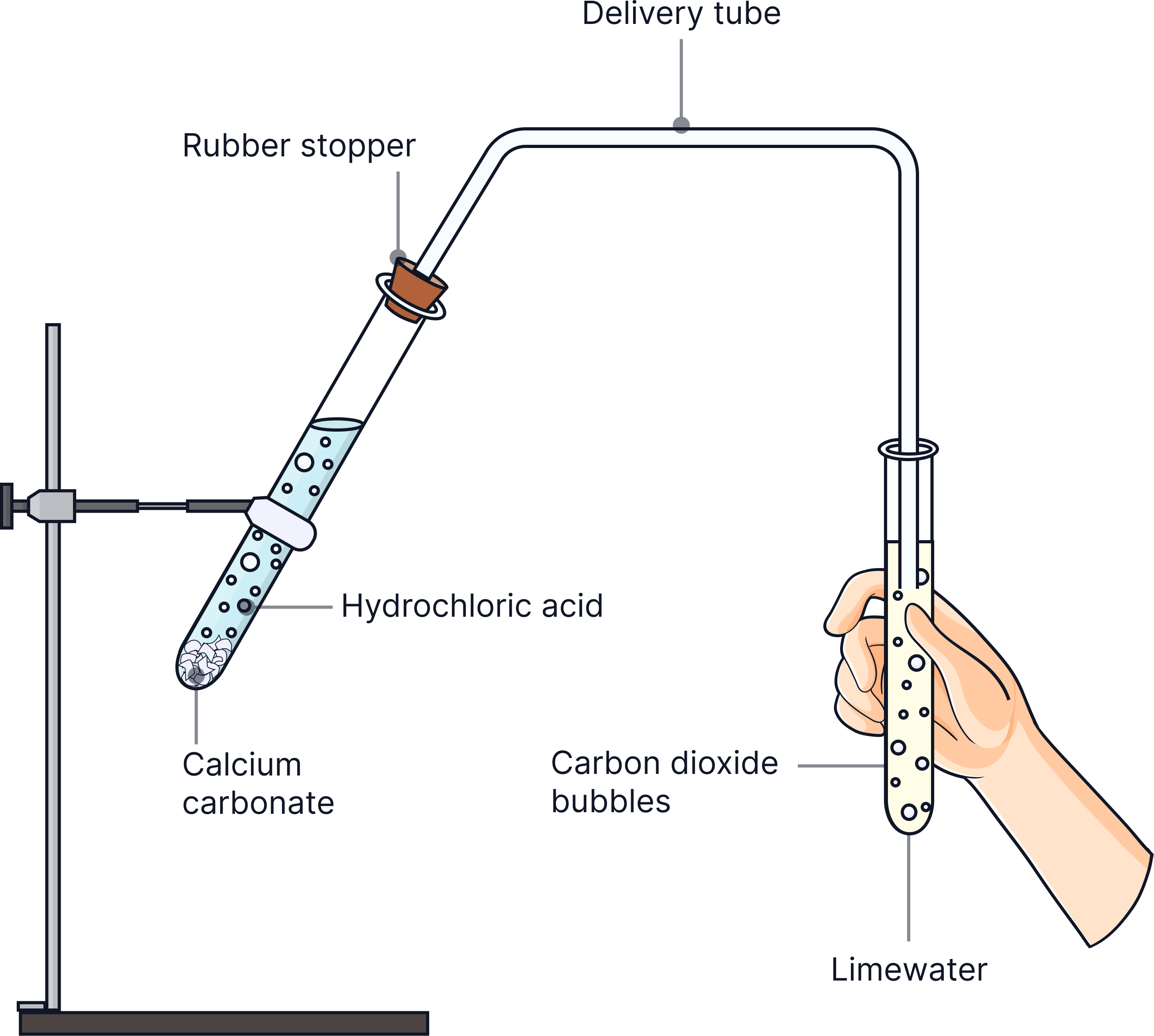
1) Add about 5 cm depth of dilute acid (hydrochloric or nitric), HCl/HNO3, to a sample of the solution into a clean test tube.
2) Look for bubbles of gas and bubble gas through limewater by connecting a bung attached to a delivery tube
3) If limewater turns cloudy, CO₂ is present and it will contain carbonate ions
Scientific background:
The carbonate ions react with the acid to produce carbon dioxide gas (Equation 1). The carbon dioxide reacts with the limewater, Ca(OH)2, to produce insoluble solid calcium carbonate, CaCO3, which turns the solution cloudy.
Equation 1: CO₃²⁻ (aq) + 2H⁺ (aq) → CO₂ (g) + H₂O (g)
Equation 2: Ca(OH)₂ (aq) + CO₂ (g) → CaCO₃ (s) + H₂O (l)
Sulfate Ions (SO₄²⁻)
Method
1) Add about 5 cm depth of dilute hydrochloric/nitric acid, HCl/HNO3, to a sample of the solution into a clean test tube (removes carbonates and prevents a false positive).
2) Add about 5 cm depth of barium chloride solution, BaCl2.
3) If a white precipitate forms then sulphate ions are present.
Scientific background:
The sulphate ions in the solution react with barium ions from barium chloride solution to form barium sulphate, BaSO₄ (Equation 1). Barium sulphate is insoluble in water, so it forms a white precipitate that makes the solution turn cloudy.
This precipitate confirms the presence of sulphate ions (SO₄²⁻) in the original solution.
Before adding barium chloride, dilute acid (hydrochloric or nitric) is added to remove carbonate ions (Equation 2), because carbonates would also form a white precipitate with barium ions and give a false positive.
Equation 1: Ba²⁺ (aq) + SO₄²⁻ (aq) → BaSO₄ (s)
Equation 2: CO₃²⁻ (aq) + 2H⁺ (aq) → CO₂ (g) + H₂O (l) - Removal of carbonate impurities (If present)
Halide ions (X- = Cl⁻, Br⁻, I⁻)
Halide ion (X-) | Colour of precipitate (AgX) |
|---|---|
Chloride (Cl⁻) | White (AgCl) |
Bromide (Br⁻) | Cream (AgBr) |
Iodide (I⁻) | Yellow (AgI) |
Method
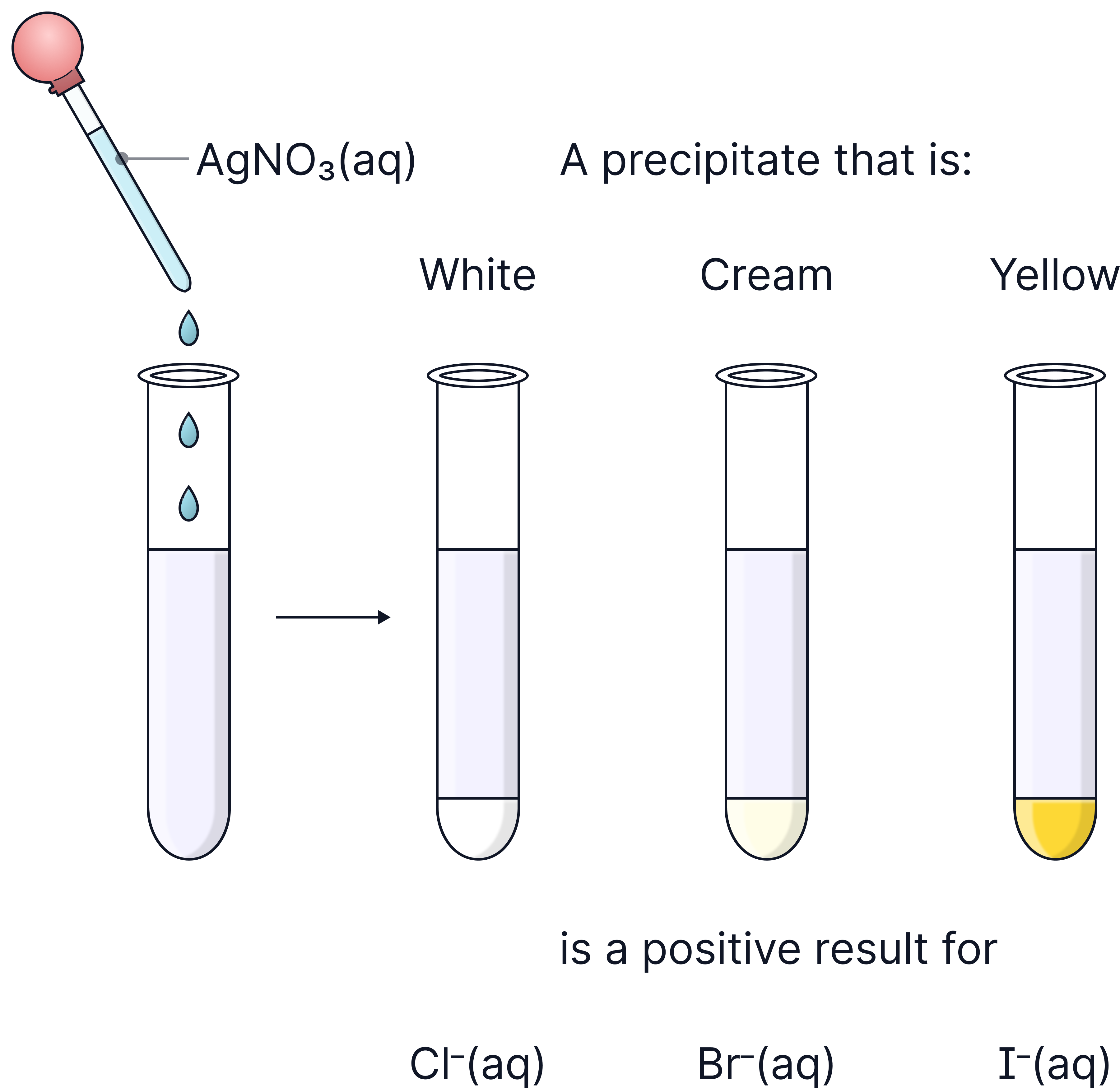
1) Add about 5 cm depth of dilute nitric acid, HNO3, to a sample of the solution in a clean test tube (removes carbonates and prevents a false positive).
2) Add about 5 cm depth silver nitrate solution, AgNO3.
3) Observe and note down the colour.
Scientific background:
The halide ions in the solution react with silver ions from silver nitrate solution to form insoluble silver halides (AgCl, AgBr or AgI). Each silver halide forms a distinctive precipitate colour, which identifies the halide present (as seen in the table above)
Because silver halides are insoluble in water, they form solid precipitates that turn the solution cloudy. This precipitate confirms the presence of halide ions in the original solution (Equation 1).
Before adding silver nitrate, dilute nitric acid is added to remove carbonate ions (Equation 2). This prevents a false positive because silver ions react with carbonate ions to form silver carbonate, another white precipitate.
Hydrochloric acid cannot be used, because it contains chloride ions, which would immediately form white silver chloride with the silver nitrate.
This would cause a false positive even if no halide ions were present in the sample.
Equation 1:
Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s)
or
Ag⁺ (aq) + Br⁻ (aq) → AgBr (s)
Ag⁺ (aq) + I⁻ (aq) → AgI (s)
Equation 2: Removal of carbonate impurities (if present/only nitric acid is suitable)
CO₃²⁻ (aq) + 2H⁺ (aq) → CO₂ (g) + H₂O (l)
Worked Examples
Example 1
An unknown solution of an ionic compound produces these observations for the following tests:
Flame test: Green flame
Sodium Hydroxide precipitate test: Blue precipitate formed
Barium Chloride Test: No reaction (no precipitate)
Gas Test for carbonates: No reaction (no bubbles)
Silver Nitrate Test: White precipitate formed
Conclusion:
The substance is copper(II) chloride, CuCl₂.
Reasoning:
This is because the observations from the flame test and precipitate test confirms that it contains Cu2+ ions, no reaction during the gas test for carbonates and with barium chloride confirmed that there are no carbonates present. The fact that a white precipitate was produced during the reaction with silver nitrate confirms the presence of chloride ions.
Example 2
An unknown solution of an ionic compound produces these observations for the following tests:
Flame test: Lilac flame
Barium chloride test: White precipitate formed
Gas test for carbonates: No reaction (no bubbles)
Silver nitrate test: No reaction (no precipitate)
Conclusion:
The substance is potassium sulfate, K₂SO₄.
Reasoning:
The lilac flame confirms the presence of potassium ions (K⁺). A white precipitate with barium chloride shows that sulfate ions (SO₄²⁻) are present. No reaction with dilute acid shows that no carbonate ions are present. No precipitate with silver nitrate means there are no halide ions (Cl⁻, Br⁻ or I⁻).
Practice Questions
Recall Questions
a) What colour flame does calcium produce?
a) Orange-red.
b) A solution forms a blue precipitate with NaOH. Which metal ion is present?
b) Copper(II), Cu²⁺.
c) Describe how you would test for carbonate ions.
c) Add dilute acid; if bubbles form, pass gas through limewater. If limewater turns cloudy, carbonate ions are present.
d) A student adds dilute nitric acid then silver nitrate and obtains a cream precipitate. Identify the ion.
d) Bromide, Br⁻.
Application Questions
e) What is the purpose of adding dilute nitric acid before silver nitrate?
e) To remove carbonate ions, which would otherwise produce a white precipitate and give a false positive.
f) How do you distinguish between Al³⁺, Mg²⁺ and Ca²⁺ using NaOH?
f)
All form white precipitates.
Al(OH)₃ dissolves in excess NaOH.
Ca²⁺ can be identified by a flame test (orange-red).
Mg²⁺ gives no flame colour and the precipitate remains white.
g) A gas turns limewater cloudy. Write the ionic equation for its formation from a carbonate test.
g) CO₃²⁻ + 2H⁺ → CO₂ + H₂O.
h) An unknown salt gives a lilac flame and forms a white precipitate with AgNO₃. Identify the salt.
h)
Lilac flame → K⁺
White AgCl → Cl⁻
Salt = potassium chloride.
Challenge Questions
i) Explain why hydrochloric acid is unsuitable for use to remove the carbonate ions before the silver nitrate test for halide ions.
i) Hydrochloric acid would introduce chloride ions which would give a false positive by reacting with silver ions to make insoluble white silver chloride.