Required Practical 3 Electrolysis
Emmanuel Opoku
Teacher

Contents
Aim
To investigate what happens when aqueous solutions are electrolysed using inert (carbon) electrodes, and to identify the elements formed at each electrode.
Scientific Background
Electrolysis is the splitting (decomposition) of an ionic compound using electricity to form its elements.
An electric current passes through an electrolyte (a molten or aqueous ionic compound), causing ions to move and undergo redox reactions at the electrodes.
The electrodes are made of graphite as it can conduct electricity but is also inert (will not react with the electrolyte).
Key Terms
Term | Definition |
Electrolyte | A substance that conducts electricity when molten or dissolved in water because its ions are free to move. |
Anode (+) | The positive electrode — attracts negative ions (anions), where oxidation occurs. |
Cathode (–) | The negative electrode — attracts positive ions (cations), where reduction occurs. |
OIL RIG | Oxidation Is Loss, Reduction Is Gain (of electrons). |
Apparatus
Equipment | Purpose |
Beaker with petri-dish lid (two holes for electrodes) | Contains the electrolyte and electrodes |
Two carbon electrodes (inert) | Conduct electricity without reacting |
Copper (II) chloride, copper (II) sulfate, sodium chloride, sodium sulfate solutions | Electrolytes normally tested |
Low-voltage DC power supply (≈ 6 V) | Provides current for electrolysis |
Crocodile clips | Connect electrodes to the wires and power supply |
Blue litmus paper | Detects chlorine or acidic gases |
Method (Step-by-Step)
1. Pour roughly 50 cm³ of the electrolyte solution into a beaker.
2. Insert carbon rods through holes in the lid (make sure they don’t touch each other).
3. Attach crocodile leads and connect to the DC power supply (red = positive = anode; black = negative = cathode).
4. Set voltage to 6 V and switch on.
5. Observe both electrodes: look for bubbling or coating formation.
6. Hold blue litmus paper near the anode — if it bleaches, chlorine is present; if it stays blue, the gas is oxygen.
7. After 5 minutes, turn off the power. Observe any metal deposit on the cathode.
8. Record your observations and repeat with the other three electrolyte solutions (optional).
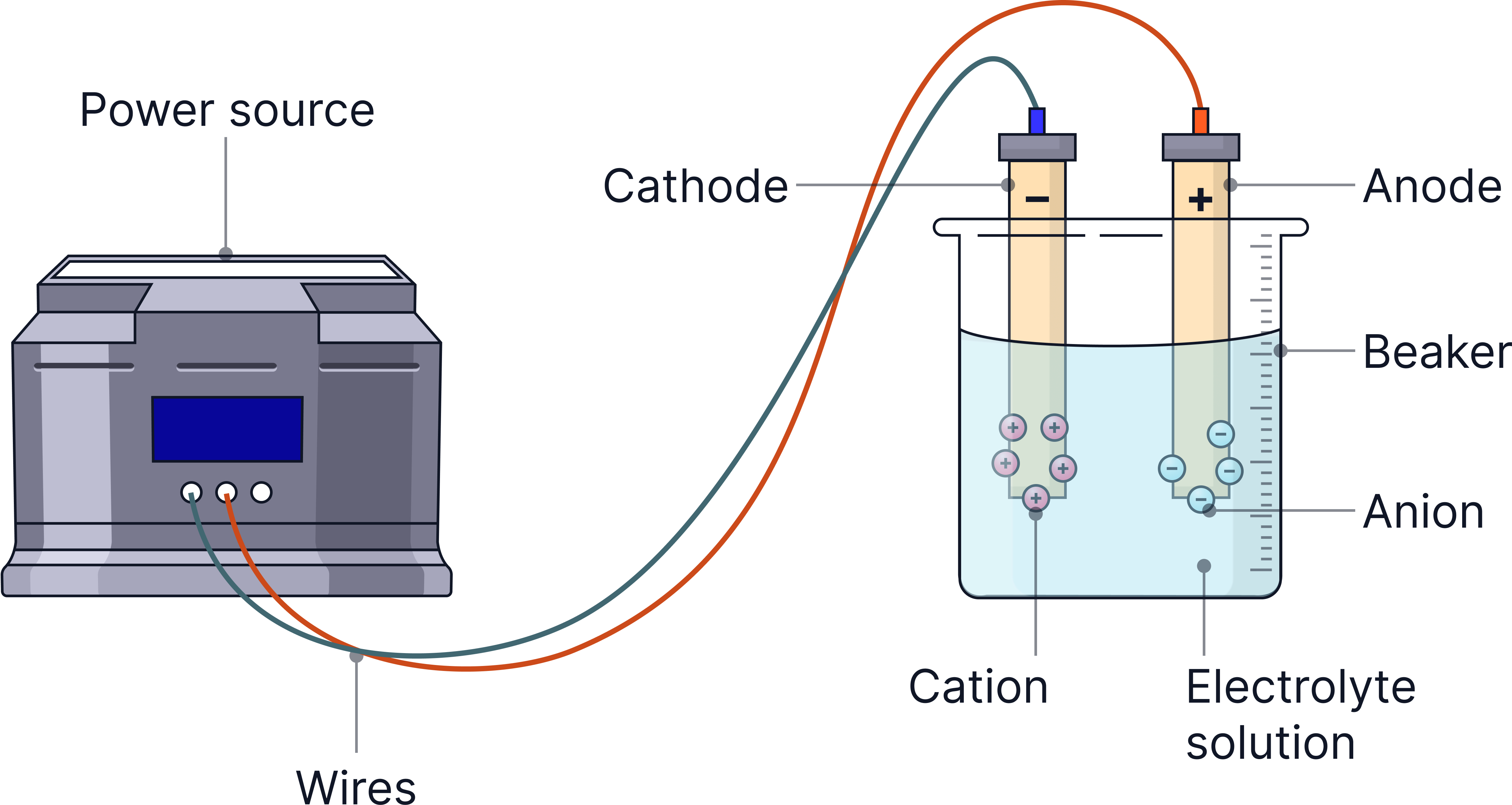
Typical Observations and Products
Anode (Positive Electrode) | Cathode (Negative Electrode) | |||
|---|---|---|---|---|
Electrolyte Solution | Observation | Element Formed | Observation | Element Formed |
Copper (II) chloride | Gas bleaches blue litmus paper | Chlorine (Cl₂) | Pink-brown coating | Copper (Cu) |
Copper (II) sulfate | Bubbles of colourless gas | Oxygen (O₂) | Pink-brown coating | Copper (Cu) |
Sodium chloride | Gas bleaches blue litmus paper | Chlorine (Cl₂) | Bubbles of gas | Hydrogen (H₂) |
Sodium sulfate | Bubbles of colourless gas | Oxygen (O₂) | Bubbles of gas | Hydrogen (H₂) |
Explanation of Results
At the Cathode (–)
Positive ions (cations) are reduced (gain electrons).
Metal ions below hydrogen in the reactivity series (e.g. Cu²⁺) form solid metal.
Metal ions above hydrogen form hydrogen gas instead.
At the Anode (+)
Negative ions (anions) are oxidised (lose electrons).
If halide ions (Cl⁻, Br⁻, I⁻) are present → halogen gas is produced.
Otherwise, oxygen gas is released from the hydroxide ions (4OH⁻ → O₂ + 2H₂O + 4e⁻).
Results Analysis
Metal deposited at cathode: Indicates a less reactive metal than hydrogen (e.g. copper).
Gas bleaches blue litmus: Chlorine released.
Colourless gas bubbles at both electrodes: Usually hydrogen (cathode) and oxygen (anode).
Safety Precautions
Wear goggles — chlorine gas is toxic.
Keep electrodes apart — short circuits can occur.
Turn off the power supply before adjusting electrodes.
Use tweezers when holding litmus paper near gases.
Practice Questions
Recall
a) What is electrolysis?
a) Splitting an ionic compound using electricity.
b) Which ions move to the anode and cathode?
b) Anions → anode (+); cations → cathode (–).
c) Why are inert electrodes used?
c) To prevent the electrodes reacting with products.
d) What does the term “inert” mean?
d) Chemically unreactive.
Apply Your Knowledge
e) Why must ionic compounds be molten or dissolved?
e) So the ions are free to move and carry charge.
f) During electrolysis of copper(II) chloride, what is formed at each electrode?
f) Copper forms at the cathode, chlorine gas at the anode.
g) What would happen if you electrolysed sodium sulfate solution?
g) Hydrogen is produced at the cathode and oxygen at the anode.
Challenge (HT Only)
h) Write half-equations for the electrolysis of molten lead(II) bromide.
h) At cathode: Pb²⁺ + 2e⁻ → Pb
At anode: 2Br⁻ → Br₂ + 2e⁻