Required Practical 6 – Chromatography
Emmanuel Opoku
Teacher

Contents
Aim
Investigate the coloured components of a mixture using chromatography by comparison to known colours.
General Scientific Background
Chromatography is used to separate mixtures and help identify substances. Different chemicals have:
-
Different solubilities in the solvent
-
Different attractions to the chromatography paper
So they separate and travel different distances → creating separate spots.
A chemical that is more soluble in the solvent will travel further up the paper.
A chemical that is more strongly attracted to the paper will travel a shorter distance.
To compare substances we calculate the Rf value:
Rf values are always between 0 – 1, and substances with identical Rf values in the same solvent are likely to be the same compound.
Pure vs Impure Substances in Chromatography
Chromatography is a powerful way to test the purity of a substance.
✔ Pure substances
A pure substance contains only one chemical, so on a chromatogram it produces:
-
Only ONE spot in a vertical column above the sample.
-
No matter how many times the experiment is repeated.
-
And the spot appears in the same position each time (same Rf value).
✔ Impure substances (mixtures)
A mixture contains more than one chemical, so it will produce:
-
Two or more spots in a vertical column above the sample.
-
Each with its own Rf value.
-
Showing that the substance is made of different components.
Variables
Independent variable
Type of dye (A, B, C, D, X - unknown).
Dependent variable
Distance moved by each dye (mm) → used to calculate Rf.
Controlled variables
-
Solvent volume
-
Solvent type
-
Size of the chromatography paper
-
Starting spot size
-
Same pencil baseline height
-
Temperature of solvent
Apparatus
-
250 cm³ beaker
-
Glass rod/wooden splint
-
Chromatography paper
-
Capillary tubes
-
Solvent (e.g. water/ethanol)
-
Known food colours (Sample 1–6)
-
Unknown sample (X)
-
Pencil
-
Ruler
-
Lid (to stop solvent from evaporating)
Method
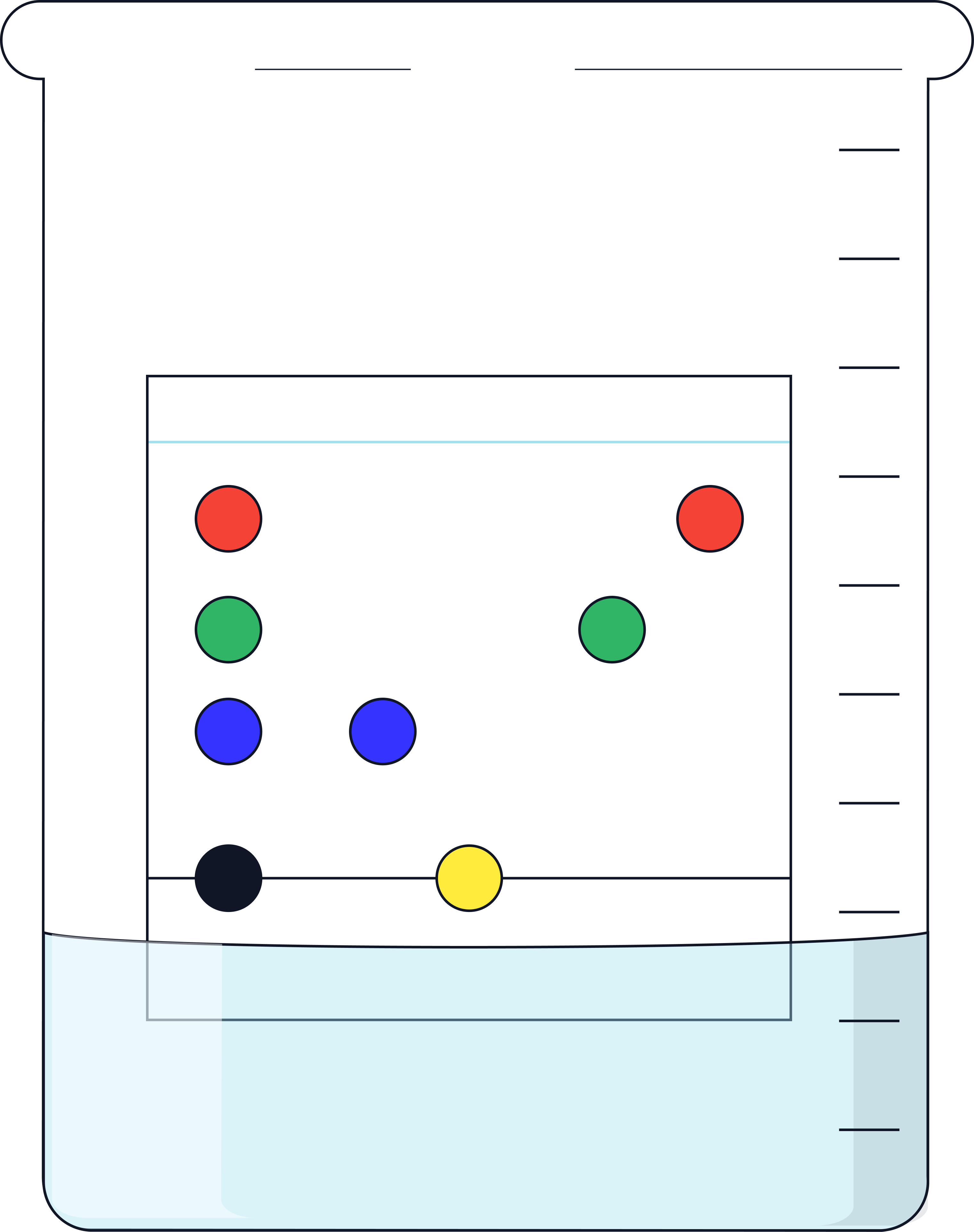
1. Draw a horizontal pencil line 2 cm from the bottom of the chromatography paper.
Mark five pencil spots evenly spaced along the line.
2. Use capillary tubes to place small spots of food colouring 1–6 and X onto the pencil marks (each spot < 5 mm diameter). Allow to dry.
3. Pour water (solvent) into a beaker to a depth of no more than 1 cm.
4. Attach the top of the chromatography paper to a glass rod/wooden split using tape/paper clip and hang it in the beaker so:
-
the baseline is above the solvent,
-
the paper does not touch the sides.
5. Place the lid over the top of the beaker and leave the chromatogram until the solvent front travels about ¾ up the paper. Do not disturb the beaker.
6. Remove the paper and immediately mark the wet solvent front with pencil.
7. Allow the paper to dry and measure the following:
-
Distance moved by solvent (baseline → solvent front).
-
Distance moved by each spot (baseline → centre of each spot).
8. Calculate Rf values using:
Example Chromatogram and Results Table
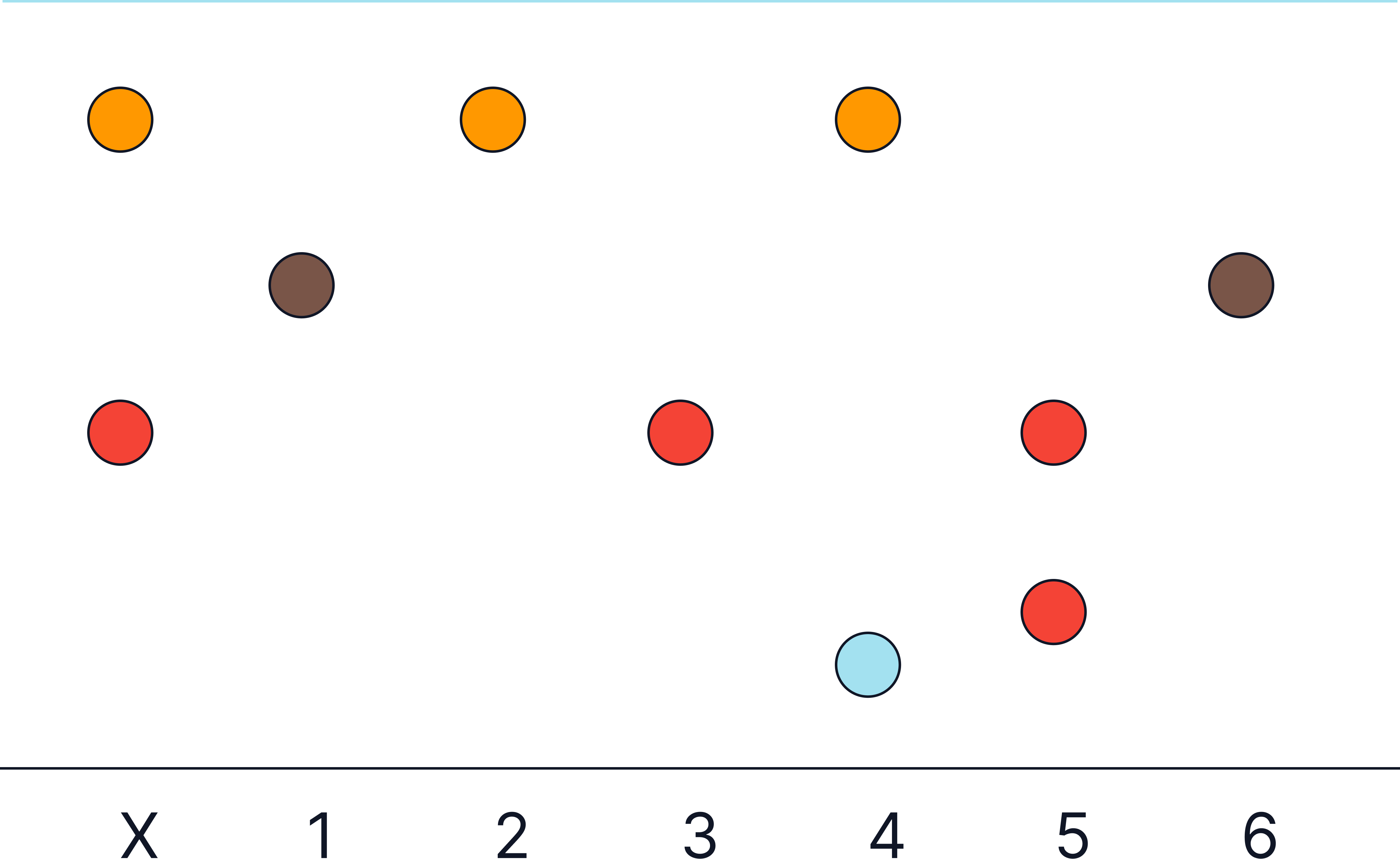
|
Sample |
Colour |
Distance (mm) |
Solvent Distance (mm) |
Rf |
|---|---|---|---|---|
|
X |
orange |
70 |
80 |
0.88 |
|
X |
red (middle) |
30 |
80 |
0.38 |
|
1 |
brown |
45 |
80 |
0.56 |
|
2 |
orange |
70 |
80 |
0.88 |
|
3 |
red (middle) |
30 |
80 |
0.38 |
|
4 |
orange |
70 |
80 |
0.88 |
|
4 |
blue |
10 |
80 |
0.13 |
|
5 |
red (middle) |
30 |
80 |
0.38 |
|
5 |
red (low) |
20 |
80 |
0.25 |
|
6 |
brown |
45 |
80 |
0.56 |
Conclusion: What Does X Contain?
X contains two substances:
-
Orange (Rf 0.88)
-
Red-medium (Rf 0.38)
✔ Substances that match X:
|
Substance in X |
Matches With |
|---|---|
|
Orange (0.88) |
2, 4 |
|
Red (0.38) |
3, 5 |
X Does one sample contain BOTH components of X?
No.
-
Samples 2 and 4 have the orange spot, but not the red (middle height). And 4 has an extra blue spot which is not in X.
-
Samples 3 and 5 have the medium red, but not the orange and sample 5 has an extra red spot (low height).
➡ Therefore: No sample fully matches X but it is a mixture of 2 and 3.
Main Sources of Error in Chromatography
|
Error |
Effect |
|---|---|
|
Baseline drawn inink/ pen |
Ink dissolves and contaminates results (shifts all Rf values) |
|
Solvent level above baseline |
Spots dissolve into solvent → all results invalid |
Practice Questions
Recall
A) What two phases are involved in paper chromatography?
A) Stationary phase = chromatography paper, mobile phase = solvent.
B) What does a pure substance look like on a chromatogram?
B) One spot above the sample in a vertical column.
C) What does Rf stand for?
C) Retention factor.
Apply
D. A spot travels 15 mm and the solvent travels 45 mm. Calculate Rf.
D) Rf = 15/45 =0.33
E. A chromatogram shows three spots for a food dye sample. Is it pure or impure? Explain.
E) Impure – more than one spot in a vertical column above the sample.
F. Two substances have Rf values of 0.33 and 0.67. Which is more soluble in the solvent?
F) The spot with Rf = 0.67 (moves further → more soluble).
Challenge
G) Explain why pencil must be used instead of pen.
G) Pen ink dissolves in solvent and interferes with the results.
H) A student draws the baseline in ink. Predict how this affects the results.
H) Ink dissolves and produces extra spots → cannot identify the sample.
I) Why do different solvents produce different Rf values for the same substance?
I) Different solvents change solubility and strength of attraction → altering how far substances travel.