Rates of reaction - Effect of concentration
Emmanuel Opoku
Teacher

Contents
Aim
Investigate how changing concentration affects the rate of reaction using three different methods:
1) Collecting a gas (gas syringe/downward displacement of water)
2) Measuring colour change/turbidity
3) Measuring mass loss
General Scientific Background – Why Concentration Affects Rate
According to collision theory, a reaction occurs when particles:
Collide with each other
With enough energy to surpass the minimum energy required to make products (activation energy)
And in the correct orientation
Increasing concentration means:
More reactant particles in a fixed volume (measured in cm³)
More frequent collisions (per second)
Higher chance of successful collisions
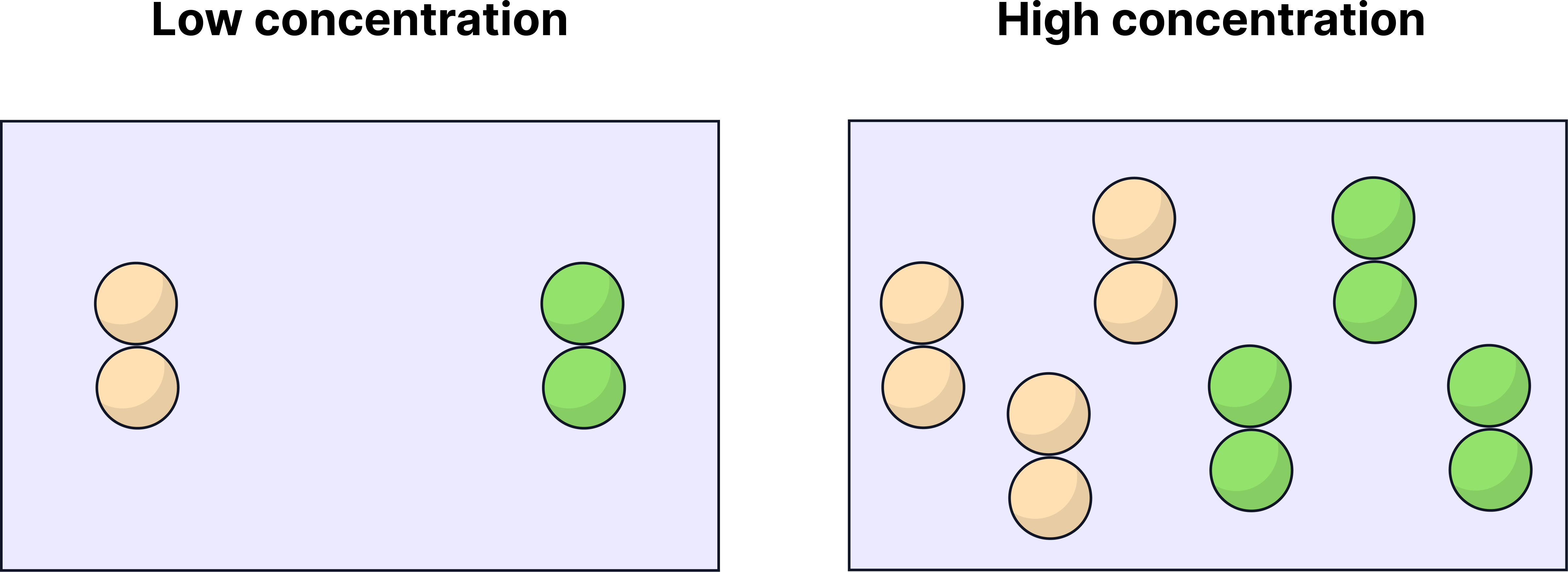
Higher concentration → more particles in a fixed volume → faster reaction rate
On graphs, this appears as a steeper curve.
All other factors that affect the rate of a reaction (temperature, pressure, using a catalyst and surface area) need to be controlled to ensure that this is a fair test. These experiments can be modified to investigate how other factors affect the rate of a reaction; this is often how they appear in exams, but the methods are broadly similar, with variables being changed accordingly.
Reducing the Effects of Errors in Required Practical 5 (Rates of Reaction)
When carrying out rate experiments (gas collection, colour change/turbidity, or mass loss), several types of error can affect the accuracy and reliability of your results. Understanding these helps you explain anomalies and improve your method.
There are two types of errors to consider:
Type of Error | Definition | Overall Effect on Results |
|---|---|---|
Random Error | Unpredictable and random variations in measurements caused by uncontrollable factors (e.g., timing variations, inconsistent observations). | Results scatter around the true value → reduces precision. |
Systematic Error | A consistent, repeatable error caused by faulty equipment or incorrect technique (e.g., balance not zeroed, leaking syringe). | All results shift in the same direction away from the true value → reduces accuracy. |
Where Errors Commonly Occur in the Three Methods
Method | Typical Error Sources | Effect on Results |
|---|---|---|
Gas Collection | Leaking bung or syringe, syringe sticking, slow stopwatch start | Gas volume too low → rate appears slower |
Colour Change / Turbidity | Different judging of when the cross “disappears”, inconsistent swirling | Time too short or too long → rate inconsistent |
Mass Loss | Balance not zeroed, CO₂ escaping before lid is on, drafts affecting balance | Mass loss too high or too low → incorrect rate |
Final Tip
To reduce errors overall, repeat measurements, calculate a mean, and keep all variables except concentration constant (volume, temperature, surface area, mass of reactants, etc.).
Gas Collection Method – Magnesium + Hydrochloric Acid
Scientific Background to Method 1
Magnesium reacts with hydrochloric acid to produce magnesium chloride and hydrogen gas:
Rate depends on collisions between Mg atoms and H⁺ ions (from the acid).
Higher acid concentration → more H⁺ ions → increased collision frequency → faster hydrogen production.
Final volume of gas is the same because same mass of magnesium reacts.
Variables for Method 1 - Gas Collection Method
Type of Variable | Specific variable in the experiment | Explanation/Application |
|---|---|---|
Independent variable | Concentration of hydrochloric acid | This is what you change to see how it affects rate. |
Dependent variable | Volume of hydrogen gas produced over time | Measured using a gas syringe or downward displacement of water at regular time intervals. |
Control variables | - Volume of acid | Must be kept constant to ensure a fair test. |
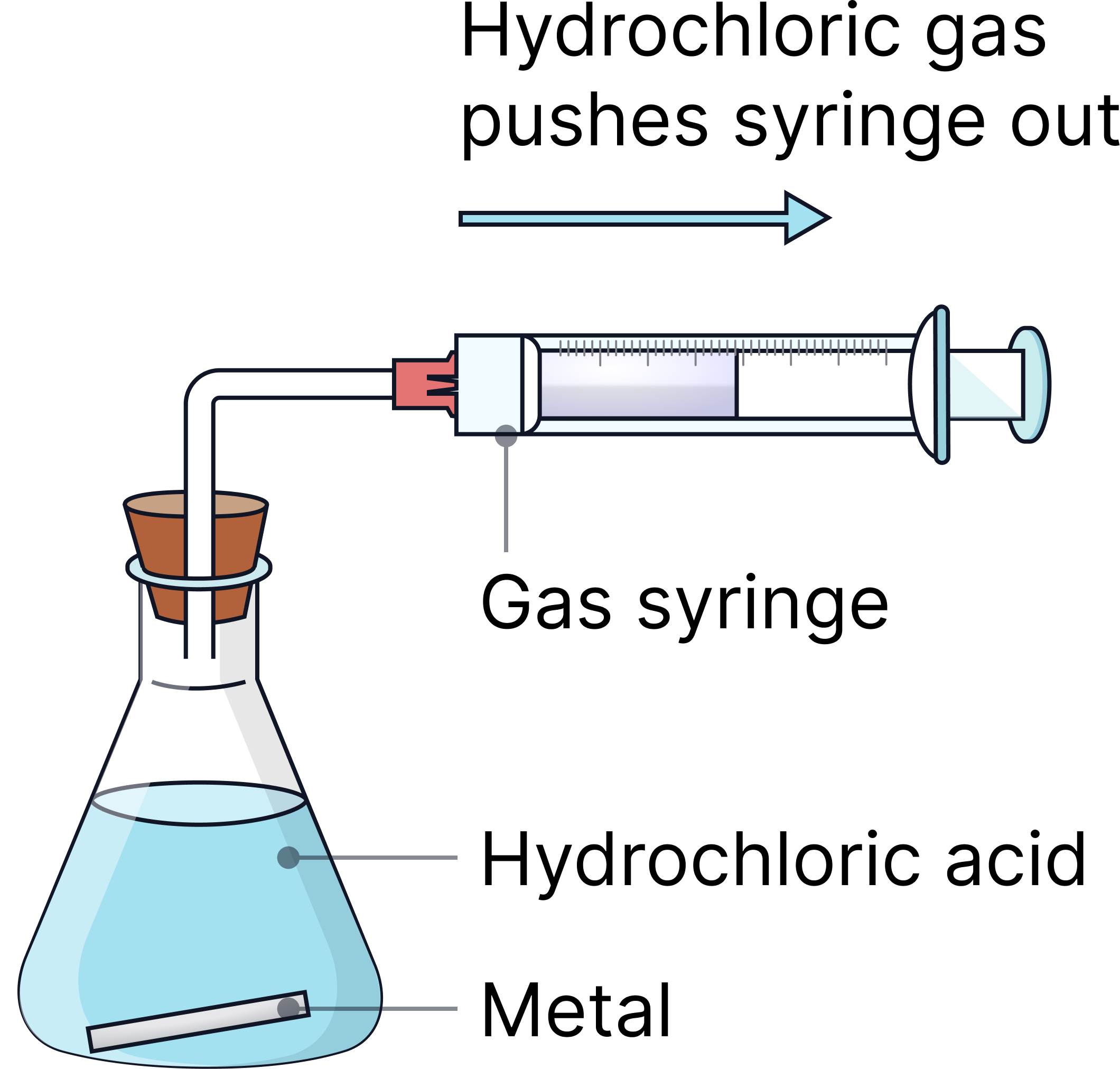
Or
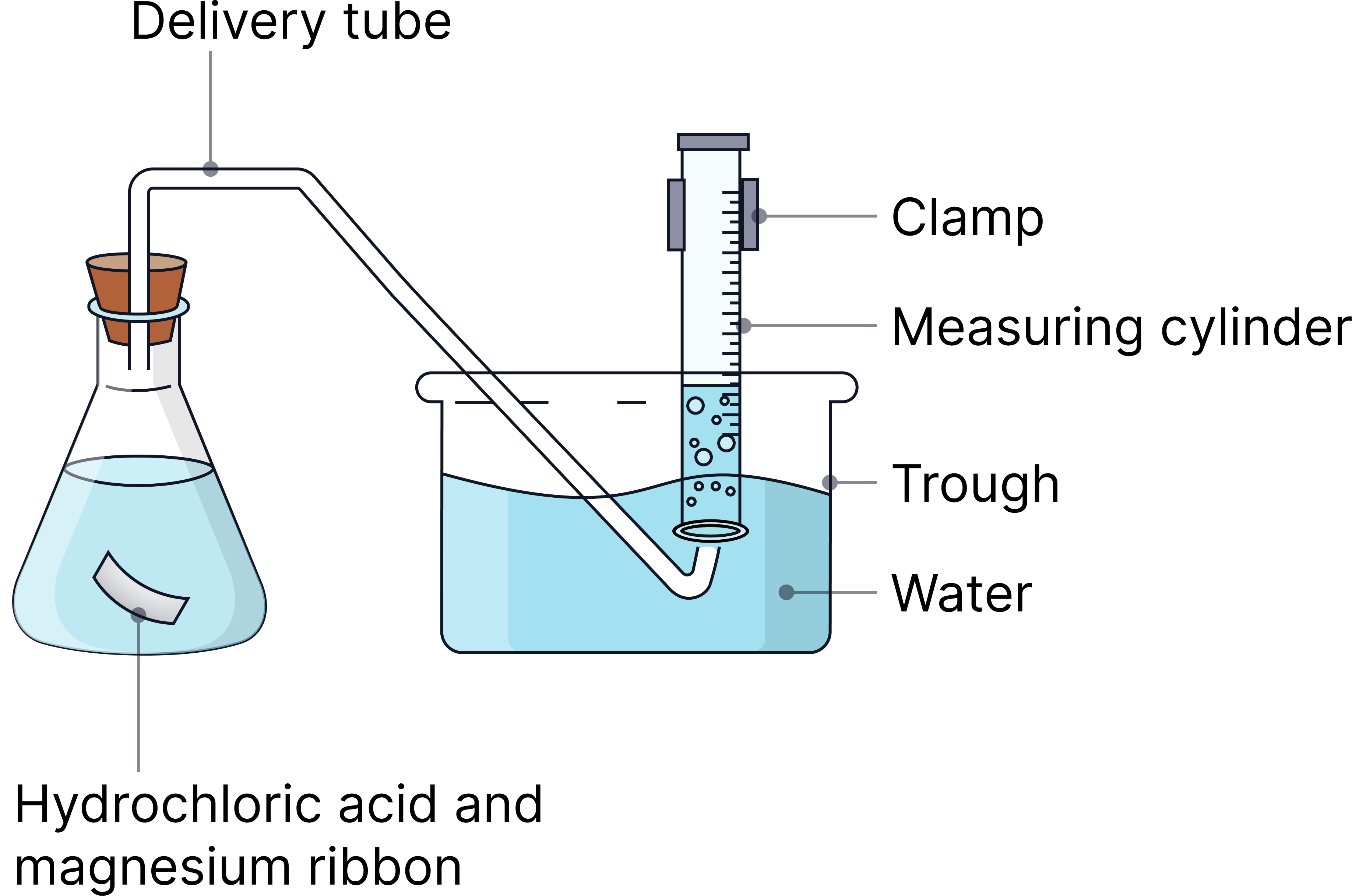
1) Measure 50 cm³ of hydrochloric acid (0.5 mol/dm³) into a conical flask.
2) Add magnesium ribbon (1g) and attach a gas syringe/delivery tube immediately.
3) Start the stopwatch.
4) Record volume of gas every 20 seconds until reaction stops (volume of gas made stops changing).
5) Repeat with a higher concentration of acid (1 mol/dm³).
6) Repeat the whole experiment at least one more time
7) Calculate the average volume of gas (ignoring any anomalies).
This gives an accurate value for the volume of gas produced.
Example Results Table (Two Concentrations)
Average Hydrogen Gas Collected (cm³) | ||||||
|---|---|---|---|---|---|---|
0.5 mol/dm³ (low concentration) |
(high concentration) | |||||
Time (s) | Trial 1 | Trial 2 | Average | Trial 1 | Trial 2 | Average |
0 | 0 | 0 | 0 | 0 | 0 | 0 |
20 | 8 | 12 | 10 | 21 | 23 | 22 |
40 | 17 | 19 | 18 | 32 | 32 | 32 |
60 | 26 | 26 | 26 | 35 | 37 | 36 |
80 | 31 | 33 | 32 | 40 | 40 | 40 |
100 | 37 | 35 | 36 | 40 | 40 | 40 |
120 | 40 | 40 | 40 | 40 | 40 | 40 |
Graph Description
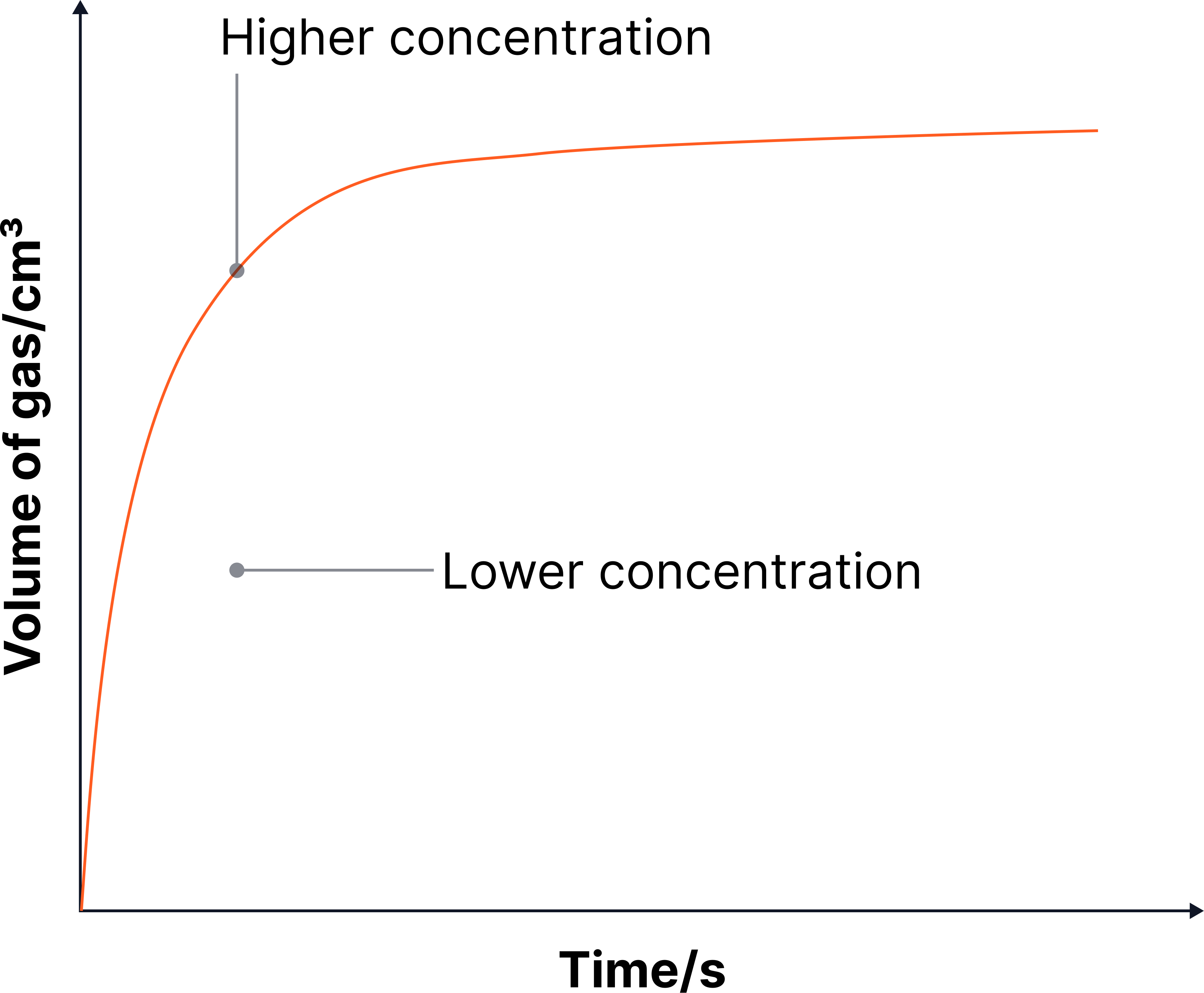
Axes:
x-axis → Time (s)
y-axis → Volume of gas produced (cm³)
The high concentration curve is steeper and reaches the final volume faster.
Both curves level off at the same volume (same amount/mass of Mg, same final yield of gas).
Error Analysis – Gas Collection Method
Source of Error | Effect on Results | Too High/Low? | Type of Error |
|---|---|---|---|
Gas escapes when adding Mg | Less gas collected | Too low | Random |
Leak in gas-syringe/delivery tube connection | Constant gas loss | Too low | Systematic |
Magnesium pieces not identical | Rate varies | Mixed | Random |
Delay starting stopwatch | Early gas missing | Too low rate | Random |
Inaccurate acid concentration | Reaction always too fast/slow | High or low | Systematic |
Colour change/Turbidity Method
– e.g. Sodium Thiosulfate () + Hydrochloric acid (HCl)
Scientific Background to Method 2
The reaction forms a solid sulfur precipitate, causing cloudiness:
Cloudiness increases until the black cross cannot be seen.
Higher concentration → solid sulfur forms faster → shorter time.
Rate is estimated using 1 ÷ time (HT Only).
Different concentrations of sodium thiosulphate solution are often achieved by diluting an original, much stronger solution with water, but ensuring the total volume of solution added stays the same.
Variables for Method 2 – Colour change/Turbidity Method
Method (Numbered Steps)
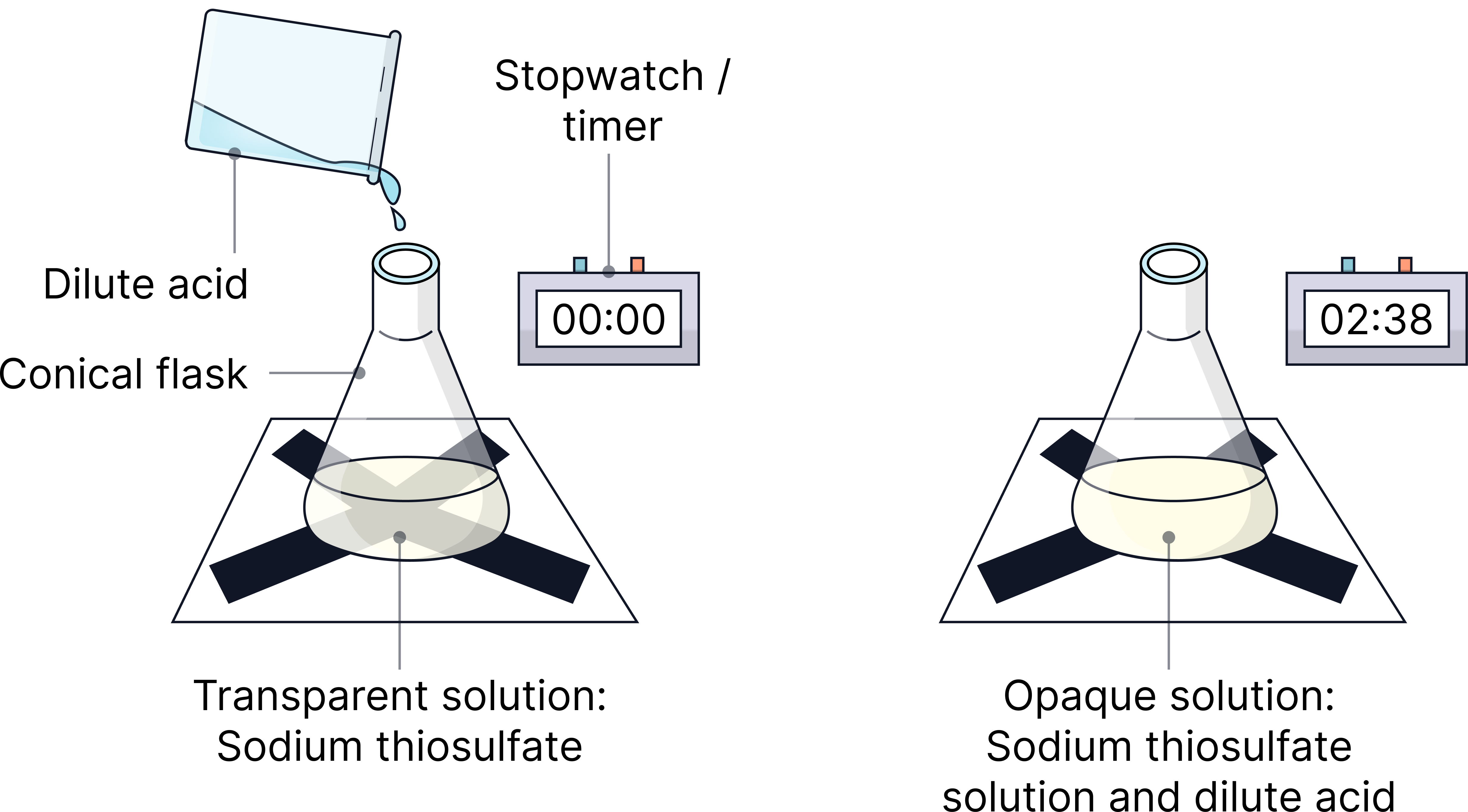
1) Place a conical flask over a black cross.
2) Add 20 cm3 of sodium thiosulfate (0.1 mol/dm³) solution.
3) Add 20 cm3 hydrochloric acid (1 mol/dm³) and start timing immediately.
4) Stop the timer when the cross disappears from view.
5) Repeat with higher concentrations of sodium thiosulphate (0.2, 0.3, 0.4 and 0.5 mol/dm³)
6) Repeat the whole experiment at least one more time.
7) Calculate the average time taken for the cross to disappear (ignoring any anomalies).
Example Results Table
| Time taken for cross to disappear (s) | HT ONLY | ||
|---|---|---|---|---|
Concentration of sodium thiosulphate (mol/dm³) | Trial 1 (s) | Trial 2 (s) | Mean (s) | Rate (1/t) |
0.10 | 108 | 112 | 110 | 0.0091 |
0.20 | 56 | 54 | 55 | 0.0182 |
0.30 | 37 | 39 | 38 | 0.0263 |
0.40 | 27 | 25 | 26 | 0.0385 |
0.50 | 21 | 19 | 20 | 0.0500 |
Graph Description
Graph A — Time vs Concentration
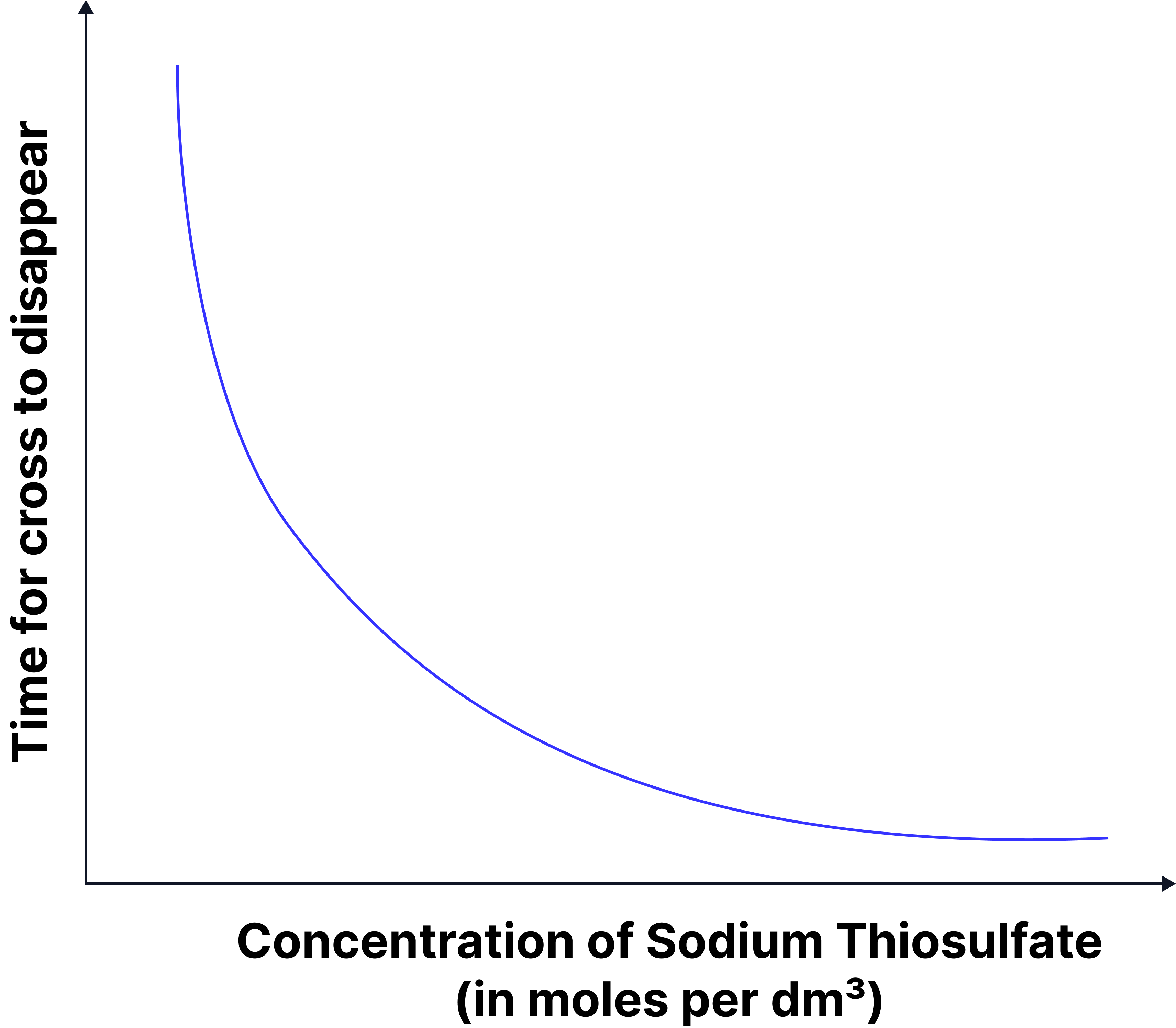
x-axis → Concentration of sodium thiosulphate (mol dm3)
y-axis → Time (s)
Decreasing curve (higher concentration → shorter time for cross to disappear)
Graph B — Rate vs Concentration - HT-ONLY
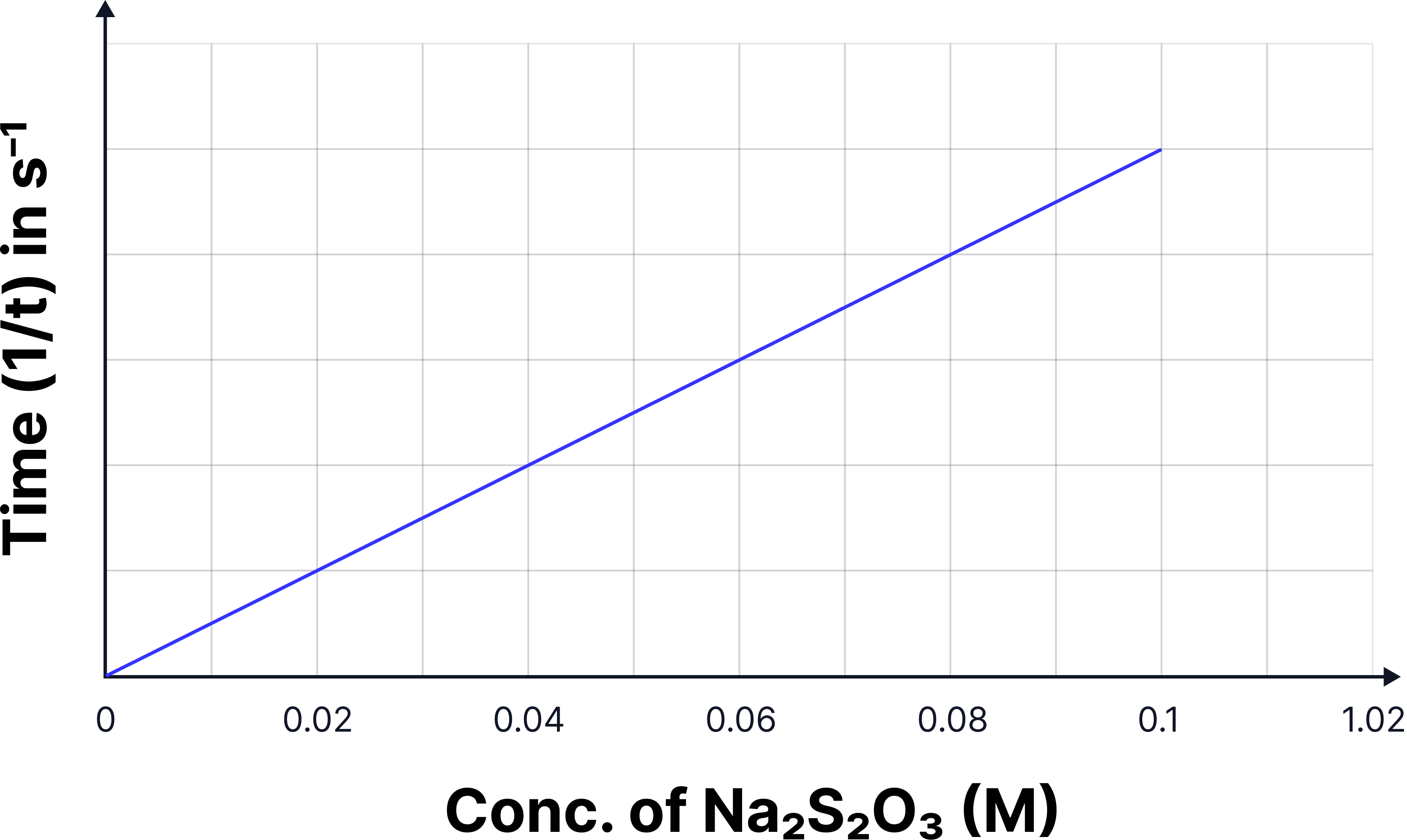
x-axis → Concentration of sodium thiosulphate (mol dm3)
y-axis → Rate (s-1)
Straight line through the origin (directly proportional relationship between concentration and rate)
Error Analysis – Colour change/Turbidity Method
Source of Error | Effect on Results | Too High/Low? | Type |
|---|---|---|---|
Human judgement of cross disappearing | Results vary | High or low | Random |
Temperature changes of solutions | Higher temp → faster rate | Too fast (lower time) | Systematic |
Total volume not constant | Changes concentration | High or low | Systematic |
Flask not mixed evenly initially | Uneven sulfur formation | Mixed | Random |
Greasy/dirty glassware | Cross obscured too early/late | High or low | Random |
Mass Loss Method – Calcium Carbonate (CaCO) + Hydrochloric Acid
Scientific Background to Method 3
Calcium carbonate reacts with acid to release CO₂ gas:
As gas escapes, the mass decreases.
Higher concentration → faster CO₂ formation → steeper mass-loss curve.
Cotton wool prevents acid spray escaping so that the only mass lost is CO₂.
Variables for Method 2 – Mass-Loss Method
Type of Variable | Specific variable in the experiment | Explanation/Application |
|---|---|---|
Independent variable | Concentration of hydrochloric acid | Changed to see how CO₂ production rate varies. |
Dependent variable | Mass of reaction mixture over time | Measured every 30 s as CO₂ escapes from the flask. |
Control variables | - Mass and size (surface area) of CaCO₃ chips - Volume of hydrochloric acid - Temperature of reaction mixture - Same balance and flask - Cotton wool bung used consistently | Controls ensure only concentration affects the rate. |
Method
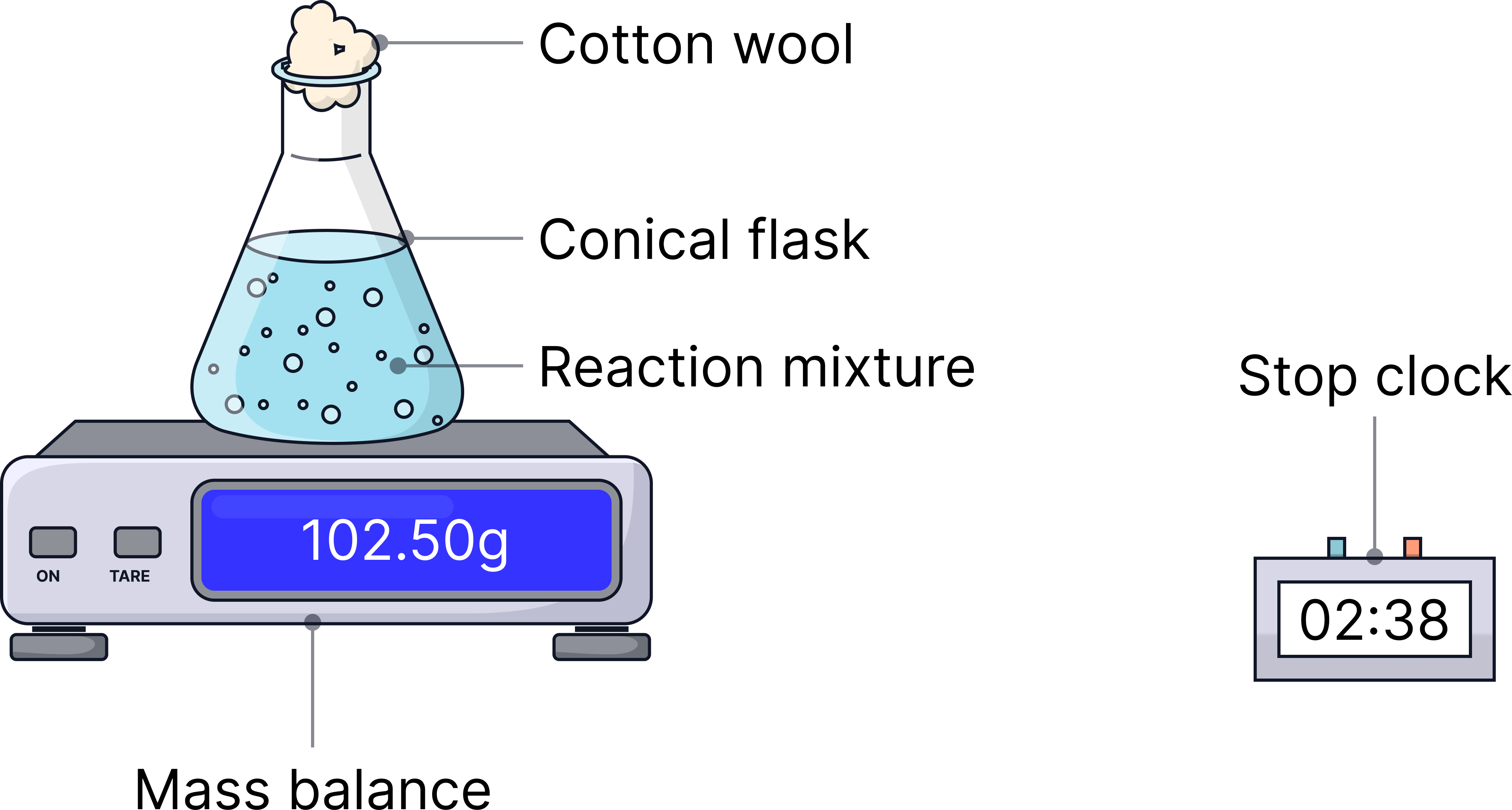
1) Place conical flask on a balance and zero it.
2) Add 5 g of calcium carbonate chips.
3) Add 20 cm3 hydrochloric acid (0.5 mol/dm³) and replace cotton wool.
4) Record mass every 30 s.
5) Repeat with higher concentration of hydrochloric acid (1 mol/dm³).
6) Repeat the whole experiment at least one more time.
7) Calculate the average mass lost (ignoring any anomalies).
Example Results Table
Mass of reaction mixture (g) | ||||||
|---|---|---|---|---|---|---|
0.5 mol/dm³ (low concentration) | 1 mol/dm³ (high concentration) | |||||
Time (s) | –Trial 1 (g) | Trial 2 (g) | Mean (g) | Trial 1 (g) | Trial 2 (g) | Mean (g) |
0 | 156.1 | 155.9 | 156.0 | 156.0 | 156.0 | 156.0 |
30 | 155.4 | 155.2 | 155.3 | 154.8 | 154.6 | 154.7 |
60 | 154.9 | 154.7 | 154.8 | 154.1 | 154.3 | 154.2 |
90 | 154.5 | 154.3 | 154.4 | 153.7 | 154.0 | 154.0 |
120 | 154.2 | 154.0 | 154.1 | 154.0 | 154.0 | 154.0 |
150 | 154.0 | 154.0 | 154.0 | 154.0 | 154.0 | 154.0 |
180 | 154.1 | 153.9 | 154.0 | 154.1 | 153.9 | 154.0 |
Graph Description
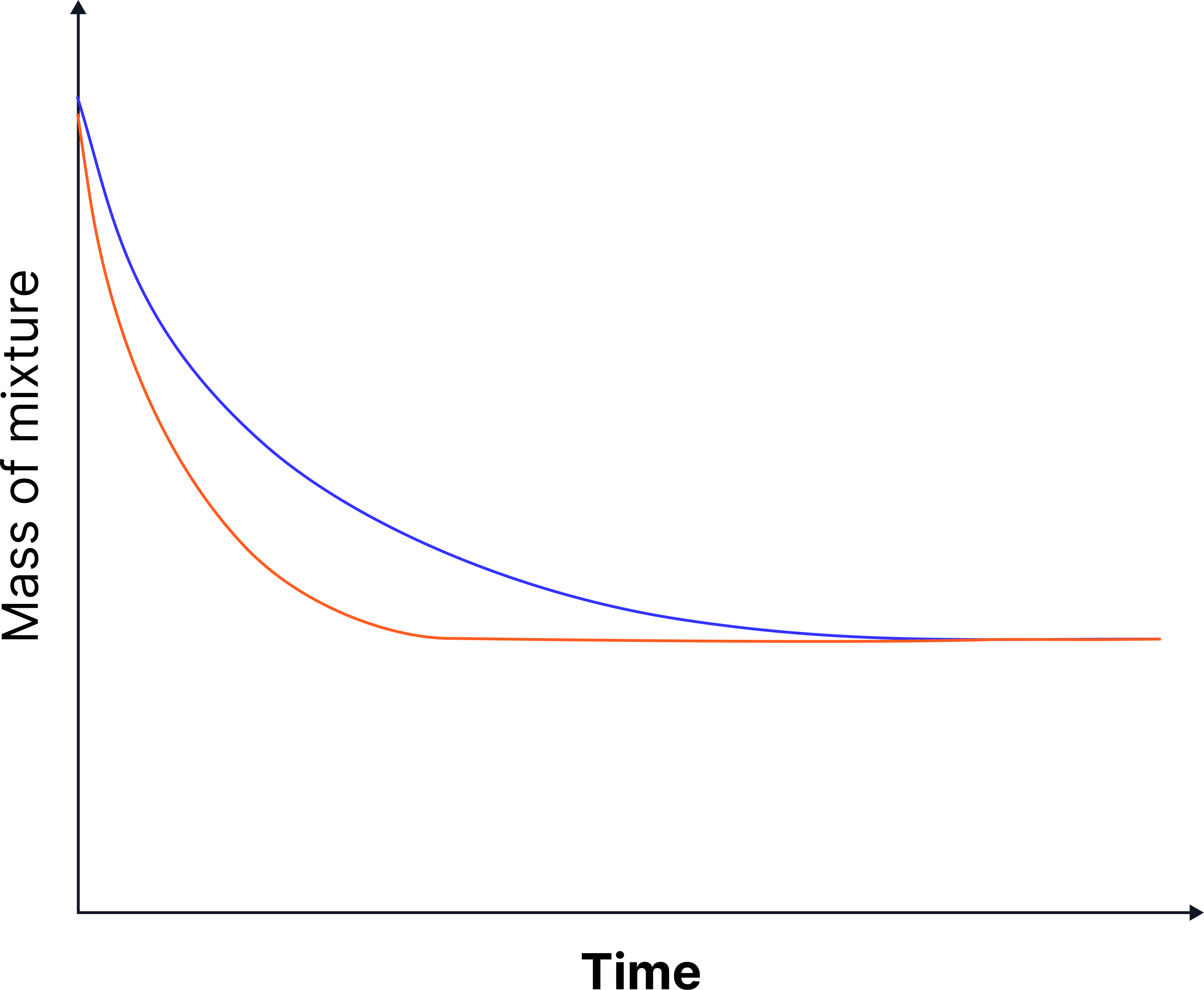
Both curves fall from the same starting mass
Higher concentration → steeper decline → fater rate of reaction
Both reach the same final mass loss (same amount/mass of calcium carbonate used)
It is common to see mass loss calculated (final mass of reaction mixture – initial mass of reaction mixture) and plotted against time.
In this case your graph will start from 0 and curve upwards – much like the graph seen in Method 1.
Error Analysis – Mass-Loss Method
Source of Error | Effect on Results | Too High/Low? | Type |
|---|---|---|---|
Cotton wool too loose → droplets escape | Mass drops too much | Too low | Systematic |
Cotton wool too tight → CO₂ trapped | Mass doesn’t fall enough | Too high | Systematic |
Balance affected by air currents | Jumping readings | Variable | Random |
CaCO₃ chips with different surface area | Rate varies | Mixed | Random |
Delay starting balance reading | Early CO₂ unmeasured | Too low rate | Random |
Practice Questions
Recall
a) State the three experimental methods used to measure reaction rate in this practical.
Answers:
a) Gas collection, colour change/turbidity, mass loss.
b) Why does increasing concentration increase the rate of reaction?
Answer:
b) Higher concentration = more particles per cm³ → more frequent successful collisions.
c) What is the role of a gas syringe in the magnesium + hydrochloric acid experiment?
Answers:
c) To accurately measure the volume of hydrogen gas produced over time.
d) What visible change occurs in the sodium thiosulphate + hydrochloric acid reaction?
Answer:
d) The solution becomes cloudy and the black cross disappears.
e) Why is cotton wool placed in the flask during the mass-loss experiment?
Answer:
e) To let CO₂ escape while preventing acid spray from leaving the flask.
f) What does the gradient of a rate graph show?
Answer:
f) The rate of reaction (steeper = faster).
g) Define a systematic error and give an example from these experiments.
Answer:
g) A systematic error affects results in the same direction every time. Example: a leak in the gas syringe → all volumes too low.
h) Define a random error and give an example from these experiments.
Answer:
h) A random error causes unpredictable variation. Example: human judgement of when the cross disappears.
Apply
i) A student collects hydrogen gas during the magnesium and hydrochloric acid experiment.
They find the 1.0 mol/dm³ acid curve is steeper than the 0.5 mol/dm³ curve.
Explain why.
Answer:
i) Higher concentration contains more H⁺ ions per cm³, so there are more frequent successful collisions with magnesium.
This produces hydrogen faster → steeper curve.
j) In the thiosulfate experiment, a student records the following times for the cross to disappear:
Trial 1 = 70 s, Trial 2 = 68 s, Trial 3 = 110 s.
Explain what has happened and what they should do.
Answer:
j) One result (110 s) is an outlier, likely due to a mistake or distraction.
They should repeat the experiment and calculate a mean excluding anomalies.
k) A student carries out the mass loss experiment, but the cotton wool completely seals the flask.
Predict how this affects the results and explain why.
Answer:
k) CO₂ cannot escape, so mass will not decrease properly.
The reaction appears slower, giving a shallower mass-loss curve.
l) During the gas-collection method, a student starts the stopwatch 5 seconds late.
Please explain how this affects the shape of their graph.
Answer:
l) The graph will start at a higher gas volume than 0 at time = 0.
The early part of the curve is missing, so the calculated initial rate is too low.
Challenge (HT Only)
m) Explain why plotting rate = 1/time against concentration for the thiosulfate reaction gives a straight-line graph through the origin.
Answer:
m) Rate is directly proportional to concentration for this reaction.
n) A student draws a mass-loss graph and finds the high-concentration curve reaches the final mass at the same time as the low-concentration curve.
Explain two possible experimental errors that could cause this.
n)
The high-concentration acid may have been accidentally diluted → slower reaction.
Different sizes of CaCO₃ chips used → surface area accidentally similar between experiments.
Cotton wool too tight → CO₂ trapped in both cases.
o) Two sets of results from the magnesium + acid experiment are shown below:
Time / s | Student A Gas Volume / cm³ | Student B Gas Volume / cm³ |
|---|---|---|
20 | 15 | 7 |
40 | 28 | 14 |
60 | 40 | 20 |
80 | 48 | 26 |
100 | 50 | 30 |
Both used 1.0 mol/dm³ acid.
Suggest reasons for the difference between A and B’s results.
Answer:
o) Student B has consistently lower gas volumes. Possible causes:
A leak in the gas syringe connection.
Gas escaping before syringe attached.
Acid concentration incorrectly measured.
Using shorter magnesium ribbon, producing less hydrogen.