Temperature changes in reacting solutions (exothermic reaction)
Emmanuel Opoku
Teacher

Contents
Aim
To investigate the variables that affect temperature changes in reacting solutions — such as neutralisation, acid + metals, acid + carbonates, or displacement reactions.
In this practical, we focus on the neutralisation reaction between hydrochloric acid and sodium hydroxide.
Scientific Background
When chemical reactions occur, energy is transferred between the system and surroundings:
Exothermic reactions → release energy to the surroundings (temperature increases).
Endothermic reactions → absorb energy from the surroundings (temperature decreases).
Neutralisation reactions between acids and alkalis are exothermic until the reaction is complete.
Once all the alkali or acid is used up, adding more reactant doesn’t increase temperature — instead, temperature may fall because you are just adding a cool solution.
Apparatus
Equipment | Use |
|---|---|
Polystyrene cup with lid (optional cotton wool) | Acts as an insulator to reduce heat loss to surroundings |
Beaker | Provides stability for the cup |
Measuring cylinders (10 cm³ and 50 cm³) | Measure accurate volumes of liquids |
Thermometer | Measure temperature change (ΔT) |
Hydrochloric acid (2 mol dm3) | Reactant acid |
Sodium hydroxide (2 mol dm3) | Reactant alkali |
Method

1) Measure 25 cm³ of sodium hydroxide into a polystyrene cup using a measuring cylinder.
2) Record the starting temperature of the solution.
3) Add 5 cm³ of hydrochloric acid, stir gently with the thermometer, and record the highest temperature reached.
4) Repeat step 3, increasing the volume of acid by 5 cm³ each time (e.g. 10, 15, 20 cm³, etc.).
5) Record the maximum temperature for each addition and calculate the temperature change (ΔT) before recording it into the table.
6) Repeat the entire experiment (steps 1–5) to obtain a second trial, and record the second set of results in the correct column of the results table.
7) Calculate the mean temperature change for each total volume of acid added and write the average values in the final column of the table.
8) Plot a line graph of:
x-axis: total volume of acid added (cm³)
y-axis: mean temperature change (°C)
Draw two straight best-fit lines:
one through the increasing temperature change points
one through the decreasing temperature change points
Extend both lines until they cross.
9) Use the point where the two lines intersect to estimate the exact volume of acid needed to neutralise the 25 cm³ of sodium hydroxide solution.
Tip – you could also do this by finding the maximum temperature reached
Example Results Table – Temperature Change (ΔT = Final – Start)
(Assuming starting temperature = 20.0 °C)
Total volume of acid added (cm³) | Temperature change (°C) | ||
|---|---|---|---|
| First Trial | Second Trial | Mean |
0 | 0.0 | 0.0 | 0.0 |
5 | +3.0 | +2.5 | +2.8 |
10 | +6.0 | +5.5 | +5.8 |
15 | +9.0 | +9.0 | +9.0 |
20 | +11.0 | +10.5 | +10.8 |
25 | +10.5 | +10.0 | +10.3 |
30 | +8.0 | +7.5 | +7.8 |
35 | +5.0 | +4.5 | +4.8 |
40 | +2.0 | +2.0 | +2.0 |
Results Analysis
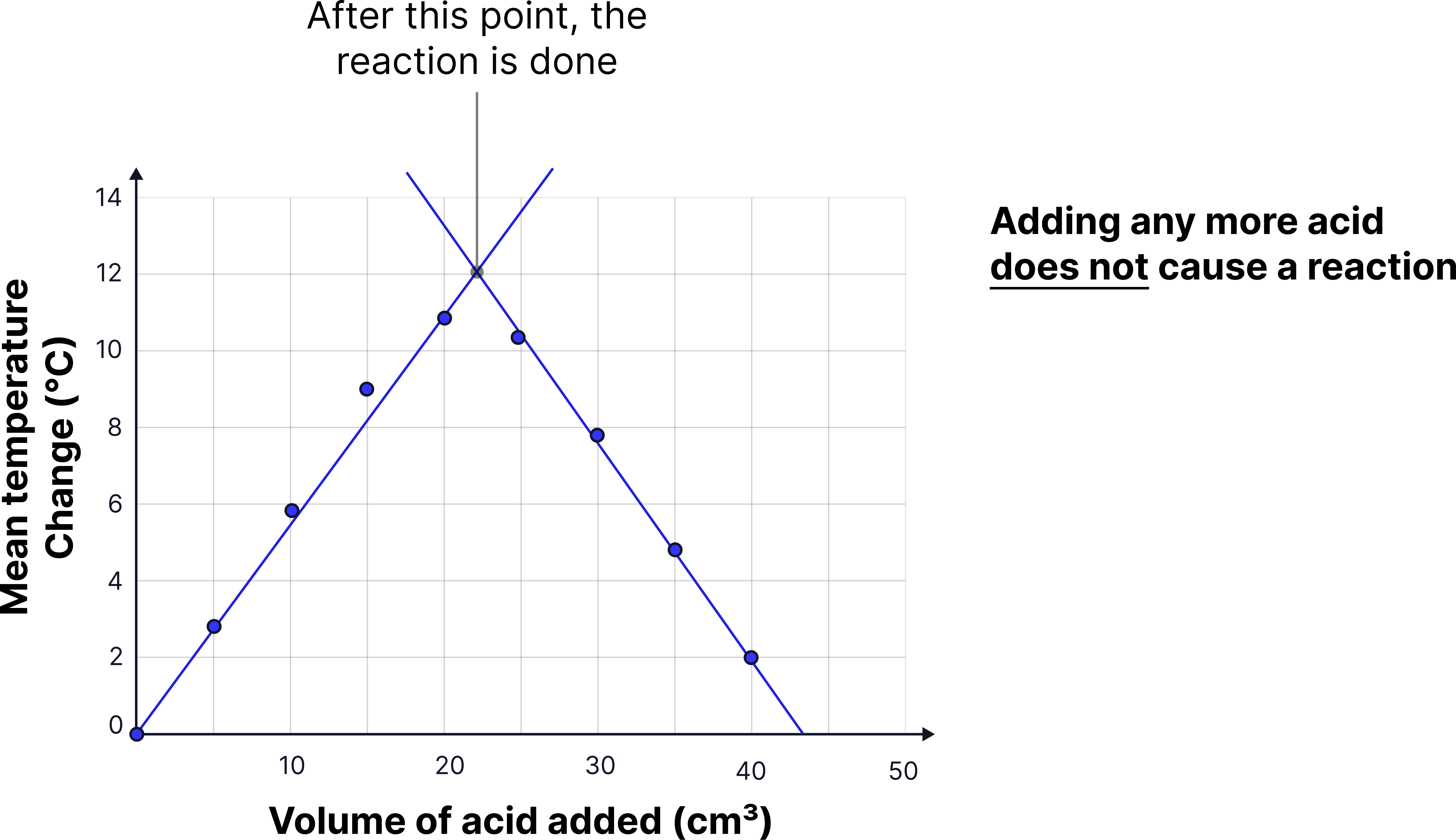
The temperature rises as acid is added because neutralisation is exothermic.
The maximum temperature occurs when all the sodium hydroxide is neutralised.
Beyond this point, adding more acid causes a fall in temperature, as the reaction is complete and the added acid simply dilutes the mixture.
Graph shape:
Upward curve to a peak (maximum temperature), then gentle downward slope.
Variables
Type | Variable | How it’s controlled or measured |
|---|---|---|
Independent variable | Volume of acid added | Measured using a measuring cylinder |
Dependent variable | Temperature change (°C) | Measured with a thermometer |
Control variables | Volume and concentration of alkali, concentration of acid, size of cup | Keep constant for a fair test |
Tip – you could also add alkali to acid instead, in which case your variables would switch acid for alkali and vice versa.
Example Calculation of temperature change
Example:
If the temperature increased from 21°C to 28°C:
Reaction type = Exothermic
Key Considerations
Use a polystyrene cup with a lid → reduces heat loss (insulation).
Stir the mixture gently → ensures even temperature distribution.
Record maximum temperature quickly → before it starts cooling.
Repeat the experiment → to calculate a reliable mean value.
Risk Assessment
Hazard | Risk | Precaution |
|---|---|---|
Hydrochloric acid / Sodium hydroxide - Irritant to skin and eyes | Medium | Wear goggles and handle carefully |
Hot solution - Burns from exothermic reaction | Medium | Do not touch cup after reaction |
Spillage – causing slippery surface | Low | Clean immediately |
Common Reactions and Their Energy Changes
It is possible to do this experiment for many reactions; the most common ones seen are listed below:
Reaction Type | Example | Energy Change | Temperature Effect |
|---|---|---|---|
Neutralisation | HCl + NaOH → NaCl + H₂O | Exothermic | Increases |
Acid + Metal | HCl + Mg → MgCl2 + H₂ (H₂ gas produced) | Exothermic | Increases |
Acid + Carbonate | 2 HNO3 + CaCO3 → Ca(NO3)2 + H₂O + CO₂ (CO₂ gas produced) | Exothermic | Increases |
Dissolving ammonium nitrate | NH₄NO₃ (s) → NH₄⁺ (aq) + NO₃⁻ (aq) | Endothermic | Decreases |
Practice Questions
1) Recall
a) What is meant by an exothermic reaction?
Answer:
A reaction that releases energy to the surroundings.
b) Why is a polystyrene cup used instead of a beaker?
Answer:
It reduces heat loss to the surroundings (acts as insulation).
c) How is the temperature change (ΔT) calculated?
Answer:
ΔT = final temperature – starting temperature.
2) Apply Your Knowledge
d) Why does the temperature eventually start to fall after the peak point?
Answer:
The reaction is complete, and extra acid just cools the mixture.
e) Suggest one improvement to reduce heat loss further.
Answer:
Use a lid or cotton wool insulation.
f) If a reaction causes the temperature to drop, what type of reaction is it?
Answer:
f) Endothermic reaction.
3) Challenge Question (HT Only)
g) The reaction between hydrochloric acid and sodium hydroxide releases 57.3 kJ per mole of water formed.
Explain why the temperature change depends on both the energy released and the mass of the solution.
Answer:
g) The larger the mass of solution, the smaller the temperature change for the same energy released - because the energy is spread through more particles.
Temperature rise depends on:
where Q is energy released, m is mass, and c is specific heat capacity.