Preparation of a pure, dry sample of a soluble salt from an insoluble oxide or carbonate
Emmanuel Opoku
Teacher

Required Practical 1 – Making Soluble Salts
Aim
To prepare a pure, dry sample of copper(II) sulphate crystals by reacting sulfuric acid with copper(II) oxide.
Scientific Background
Soluble salts can be made by reacting an acid with an insoluble base such as a metal oxide, hydroxide, or carbonate.
This is a neutralisation reaction producing a salt and water.
Example: Copper (II) oxide (base) and sulphuric acid (acid)
Word equation:
Sulfuric acid + Copper(II) oxide → Copper(II) sulphate + Water
Balanced symbol equation:
Apparatus
40–50 cm³ dilute sulfuric acid (specific concentration i.e. 0.5 mol dm-3)
Copper(II) oxide powder
Beaker
Tripod
Gauze
Bunsen burner – for heating
Glass rod
Filter funnel, paper, conical flask – for filtration
Evaporating basin
Beaker (for water bath)
crystallising dish – for crystallisation
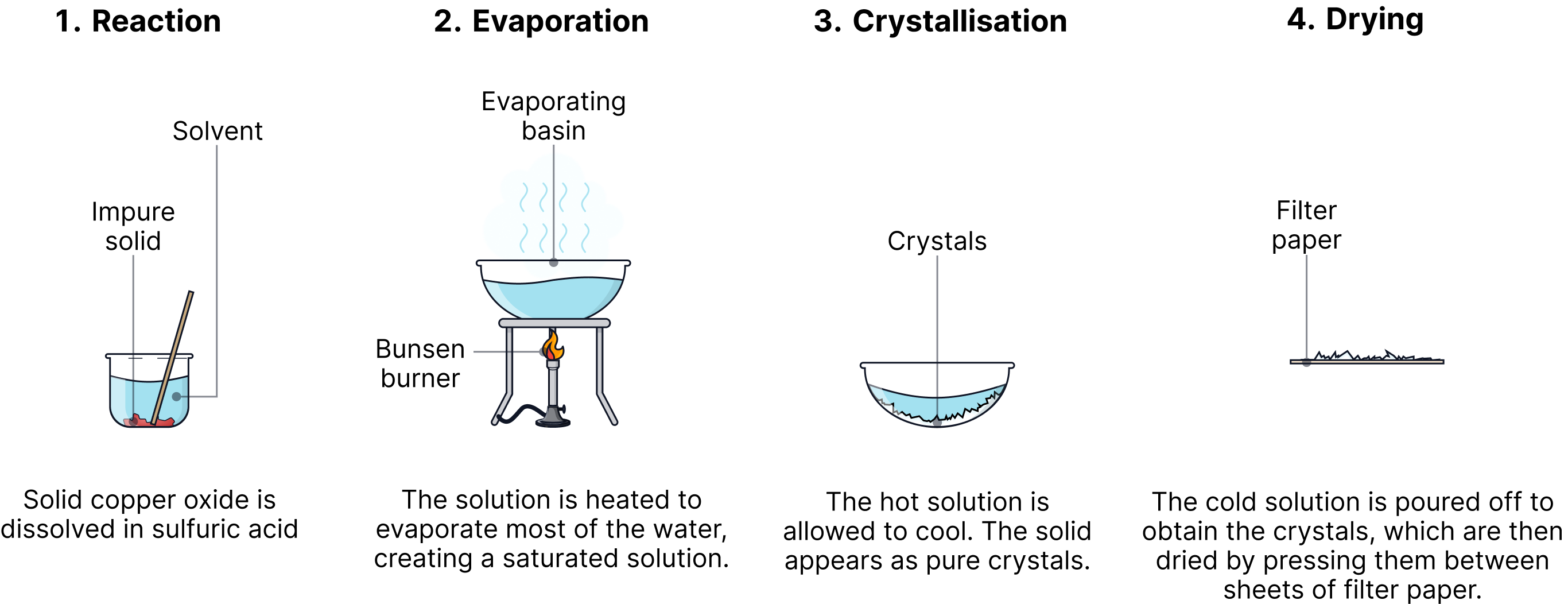
Method
1. Measure 40 cm³ of dilute sulfuric acid into a beaker.
2. Gently warm the acid using a Bunsen burner.
3. Add small spatulas of copper(II) oxide to the warm acid while stirring.
4. Continue adding (in excess) until some black solid remains.
5. Filter the mixture to remove unreacted solid and collect the blue filtrate.
6. Heat the filtrate gently using a water bath until about half the liquid evaporates.
7. Allow the hot solution to cool slowly and form crystals.
8. Remove and dry the crystals using filter paper.
Key Steps
1. Adding the Base in Excess
Copper(II) oxide is added until no more dissolves and solid remains at the bottom (in excess).
This ensures all the acid reacts completely, preventing any leftover acid that could make the final crystals impure.
Using an excess of the base guarantees the salt solution is neutral, containing only copper(II) sulphate and water.
2. Filtration
The reaction mixture contains unreacted solid copper(II) oxide and copper(II) sulphate solution.
Filtration separates these by passing the mixture through filter paper.
The filtrate is the clear blue solution of copper(II) sulphate, while the residue is the unreacted oxide.
This step ensures purity - only the soluble salt remains.
3. Partial Evaporation
The filtrate is heated gently in a water bath (ensures gentle evaporation) until about half the water has evaporated, forming a saturated solution.
Heating directly with a Bunsen flame can cause spitting/popping of the crystals or overheating, leading to decomposition of copper(II) sulphate (which breaks down into black copper oxide and sulphur oxides which are toxic).
After partial evaporation, the solution is left to cool slowly, allowing large, pure blue crystals to form.
Safety
Wear goggles - sulfuric acid is corrosive.
Handle hot apparatus carefully.
Avoid skin contact with copper(II) sulphate or pouring down the sink - toxic if ingested. And harmful to aquatic life.
Use a water bath to heat gently and safely.
Practice Questions
Recall
a) Write the balanced symbol equation for this reaction.
a) H₂SO₄ + CuO → CuSO₄ + H₂O
b) Why is the base added in excess?
b) To ensure all the acid reacts and no acid remains to contaminate the salt.
c) What type of reaction forms the salt?
c) A neutralisation reaction.
Apply Your Knowledge
d) Why is the solution filtered before evaporation?
d) To remove unreacted solid base and ensure a pure salt solution.
e) Why is a water bath used for heating instead of a direct flame?
e) To heat gently and avoid spitting or decomposition of copper(II) sulphate.
f) What happens if the solution is overheated during evaporation?
f) The salt decomposes, turning black (forming copper oxide) and becoming impure.
Challenge Question (HT Only)
g) Explain how the rate of cooling affects the size and purity of the crystals formed.
g) Slow cooling allows ions to arrange into an orderly lattice, producing larger, purer crystals. Rapid cooling traps impurities and forms smaller, irregular crystals.