Addition Polymerisation (Triple Only)
Lajoy Tucker
Teacher

Contents
What Are Addition Polymers?
Key idea:
Many plastics are made from small alkene molecules that link together to form extremely long chains. These long chains are known as addition polymers.
Why alkenes?
Alkenes contain C=C double bonds.
These double bonds can open up, allowing each alkene molecule (a monomer) to connect with its neighbours.
When thousands of monomers join together, they form a polymer.
Result:
A polymer contains exactly the same atoms as the monomers it came from - nothing is gained or lost in the reaction.
How Addition Polymerisation Works
Step-by-step idea
1. A monomer with a double bond approaches others under conditions such as high pressure and a catalyst.
2. The double bond breaks, giving each carbon an extra bonding position.
3. These new bonding positions create a continuous chain as many monomers link together.
4. The chain produced is the addition polymer.
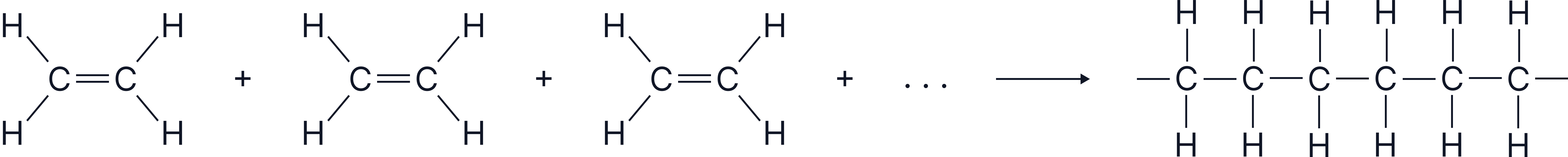
A simplified representation of ethene monomers connecting to form a long-chain addition polymer.
Example: Making Poly(ethene)
Ethene molecules join together to form poly(ethene). The repeating unit comes from the ethene molecule after its C=C bond opens.
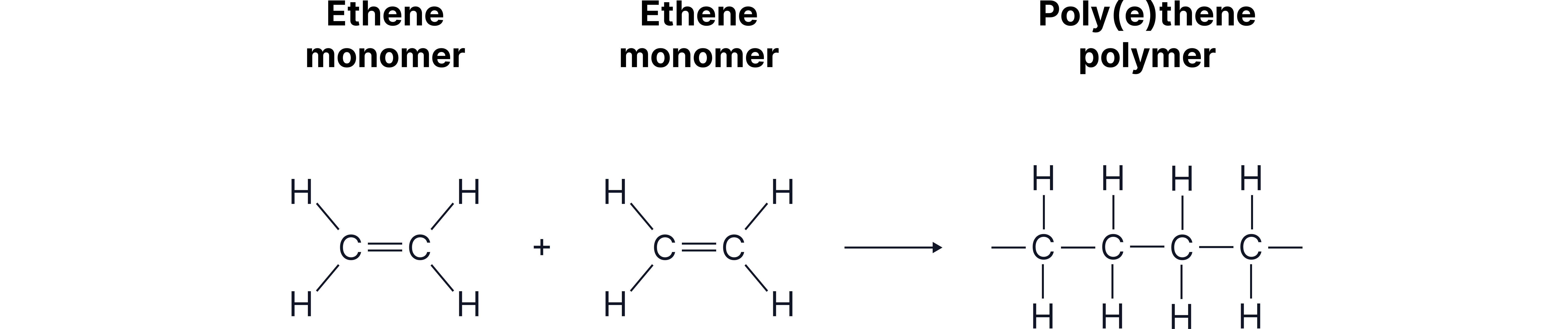
Ethene molecules link through their opened double bonds to produce the polymer poly(ethene).
Example: Making Poly(propene)
Propene monomers undergo the same process.
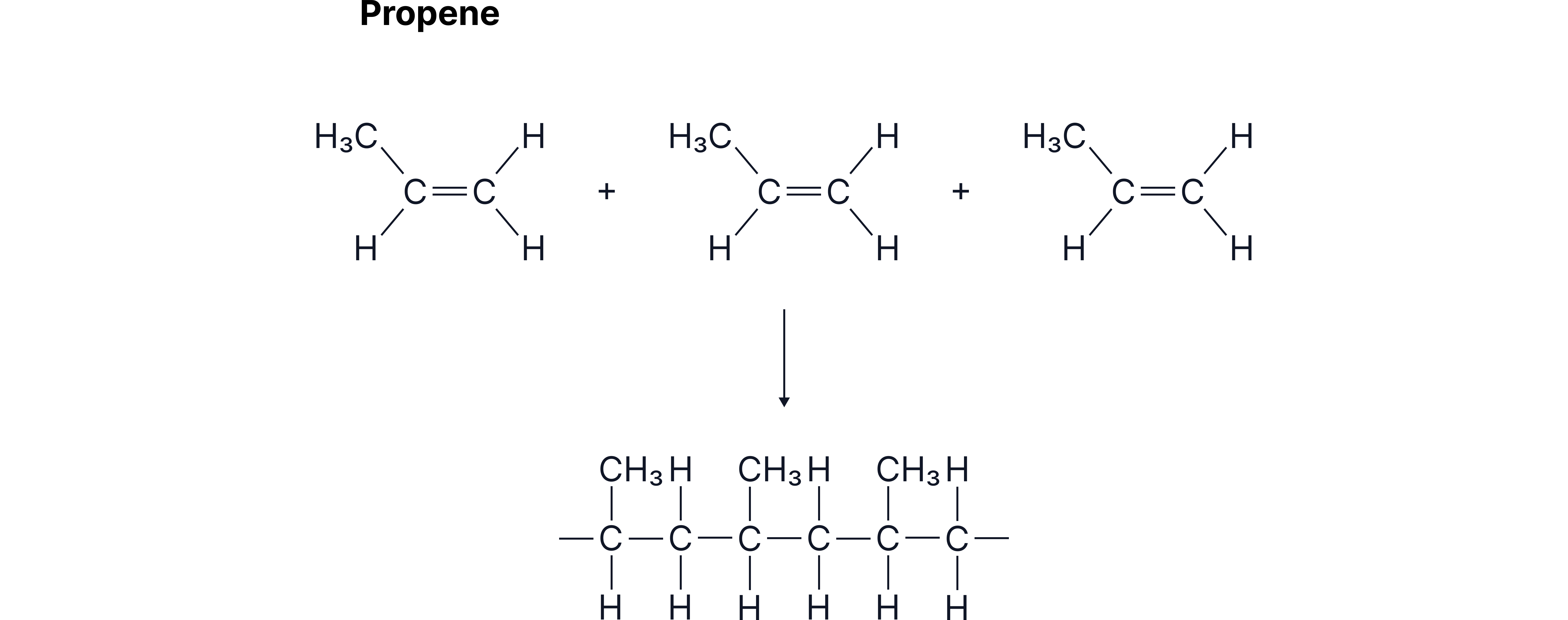
When many propene molecules react, they form the polymer known as poly(propene).
Naming addition polymers
Take the name of the monomer
Add “poly(_____ )” in front
e.g., propene → poly(propene)
Sometimes the brackets are dropped: polypropene
Drawing the Displayed Formula of Polymers
You can convert a monomer into its polymer repeating unit by following a consistent method.
Process for drawing a polymer from a monomer
1. Locate the C=C double bond in the monomer and change it to a single bond.
2. Add one extra bond to each carbon where the double bond was opened - these bonds will connect to the next repeating units in the chain.
3. Copy all other groups exactly as they appear around the double bond.
4. Place brackets around the repeating section and add ‘n’ to the right-hand side.
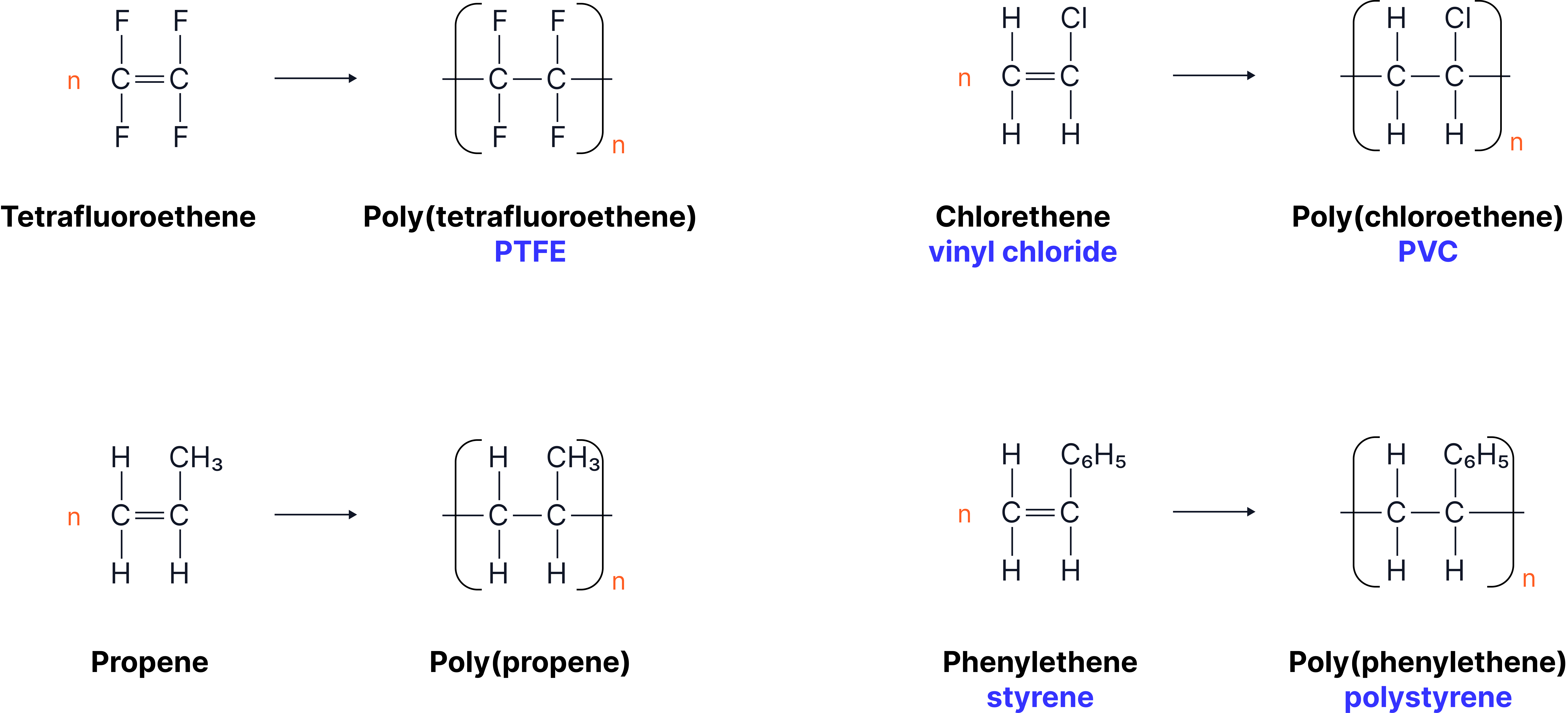
A monomer’s double bond becomes a single bond in the polymer, and the structure inside the brackets shows the repeating section of the chain.
Drawing the Monomer from the Polymer
Sometimes you are given the polymer and must work backward.
Method
1. Identify the repeating section inside the brackets.
2. Remove the two connecting bonds at each end.
3. Restore the double bond between the main carbon atoms.
This gives you the original monomer.

Check for Understanding
Recall
a) What type of monomer forms addition polymers?
Answer
Alkene / molecule with C=C.
b) What happens to the C=C bond during polymerisation?
Answer
Double bond opens / becomes single bond.
c) What is meant by a repeating unit?
Answer
Section that repeats along the chain.
d) Why are addition polymers described as saturated?
Answer
No double bonds remain after polymerisation.
Application
e) Name the polymer formed from ethene.
Answer
Poly(ethene).
f) Describe the repeating unit formed from propene.
Answer
–CH₂–CH(CH₃)– in brackets with n.
e.g.

g) Explain why addition polymerisation produces only one product.
Answer
No small molecule eliminated; only polymer formed.