Condensation Polymerisation (Triple Only)
Lajoy Tucker
Teacher

Contents
What Condensation Polymers Are
Condensation polymers are long-chain molecules made when monomers with two reactive functional groups join together.
Each time a link forms, a small molecule is released (often water, sometimes hydrogen chloride or methanol).
This loss of a small molecule gives the process its name: condensation polymerisation.
Key idea: Every monomer must be able to bond twice so the chain can continue growing.
How Condensation Polymerisation Works
Monomers involved
A condensation polymer can be made from:
1. Two different monomers, each with two identical functional groups
(e.g., a molecule with two groups reacts with a molecule with two –OH groups), or
2. One monomer with two different functional groups
(e.g. an amino acid which contains both and groups).
Step-by-step process
1. Functional groups on neighbouring monomers react.
2. A covalent link forms between the monomers.
3. A small molecule is expelled (commonly water).
4. The product still contains a reactive group at each end, so it can react again.
5. This repeats thousands of times → a polymer chain.
Polyester Formation
Polyesters
Polyesters are one of the most important families of condensation polymers.
Functional groups needed
To make a polyester, you need:
A dicarboxylic acid (contains two groups), and
A diol (contains two groups)
When these react, they form an ester linkage each time the group reacts with an group.
Example 1:
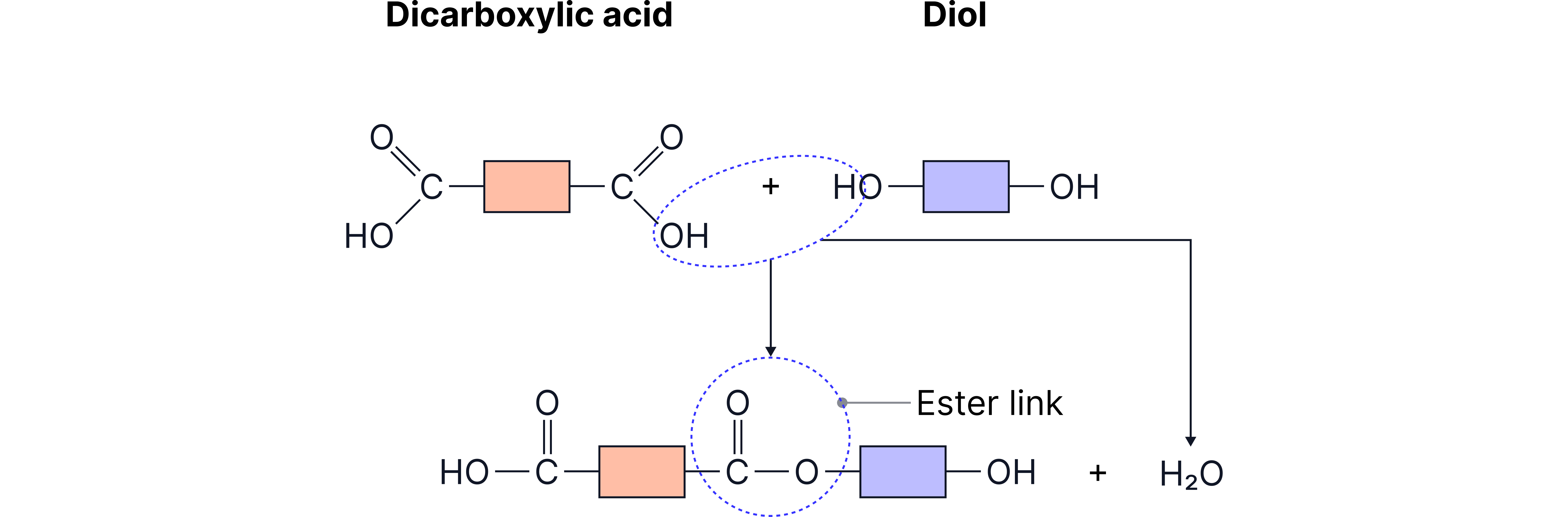
Figure 1: The formation of an ester link.
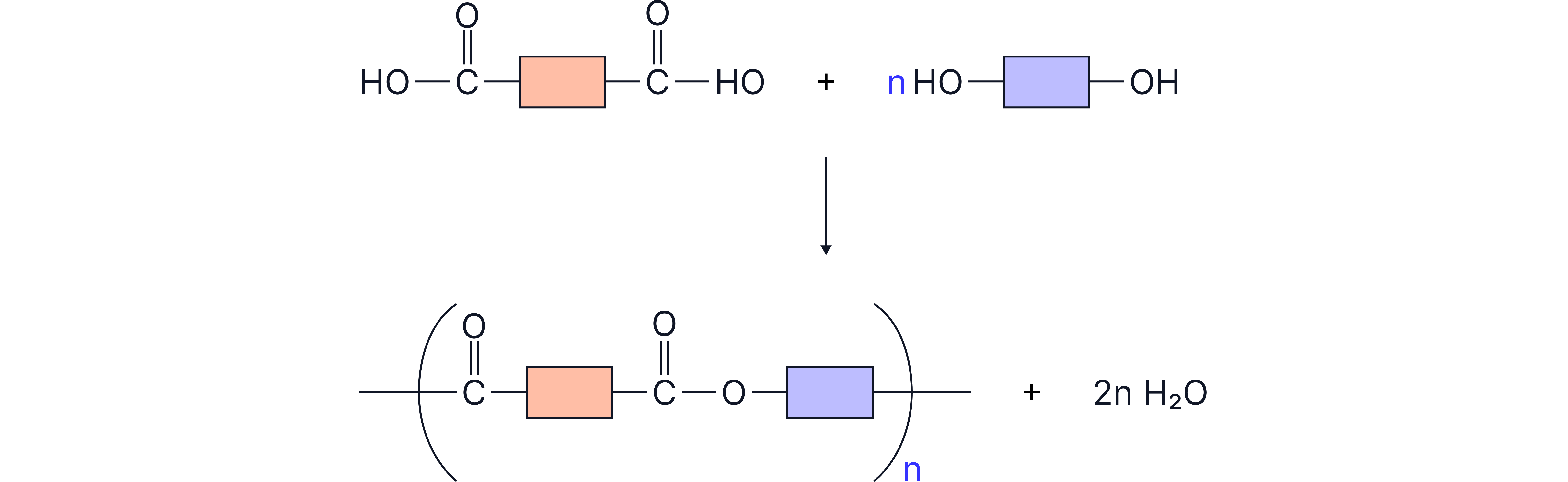
Figure 2: Formation of a polyester.
Example 2: Terylene (polyethylene terephthalate (PET)) used in textiles and plastic drinks bottles
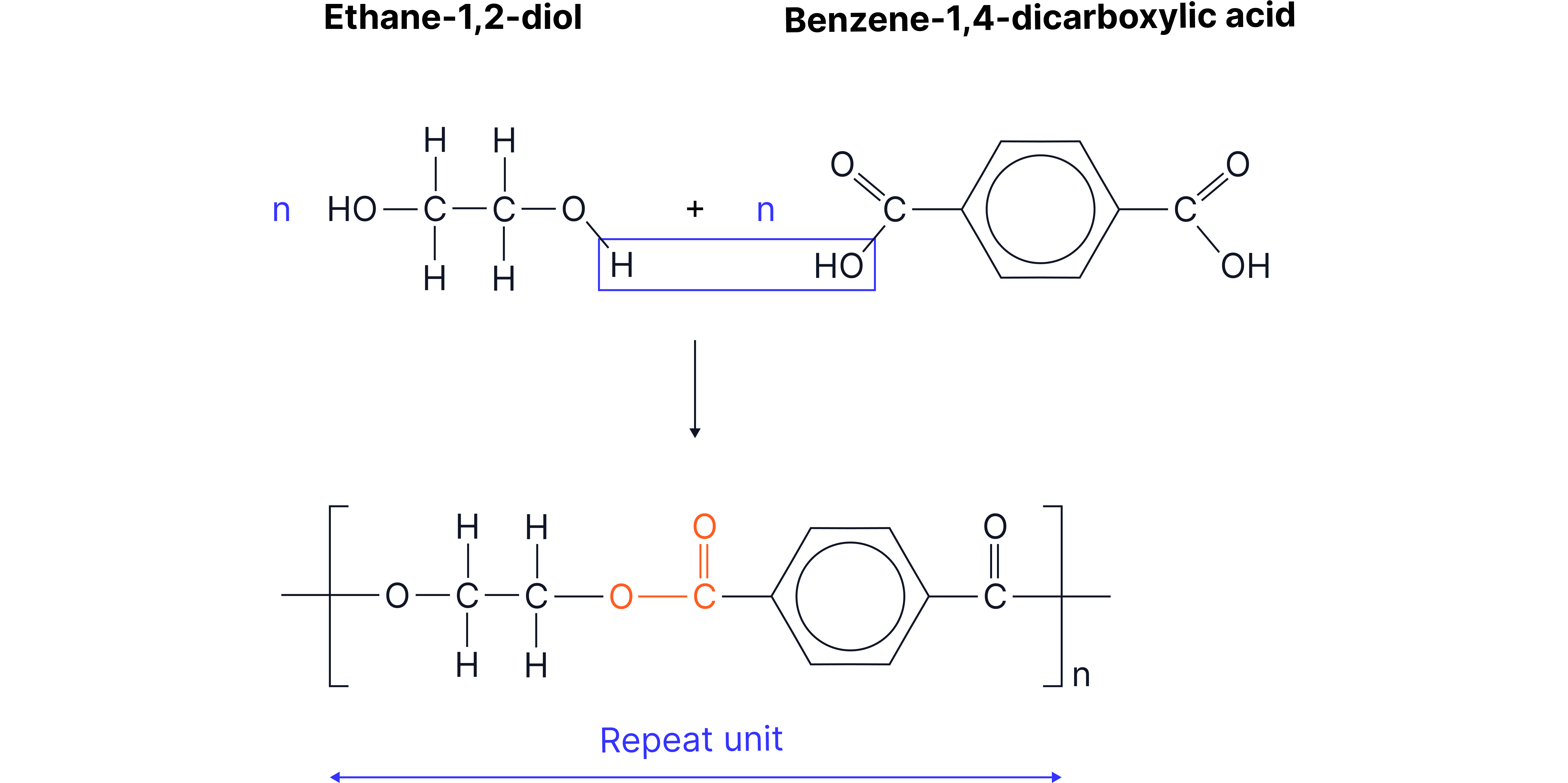
How Condensation Polymerisation Differs from Addition Polymerisation
Both processes create polymers - but the similarities end there.
Types of monomers
Addition polymerisation
Requires alkenes (monomers with double bonds).
Uses one monomer type.
Condensation polymerisation
A single molecule contains two different types of functional groups.
E.g. amino acids with and . Because the two groups can react with each other between molecules, one type of monomer is enough to form a chain.
You use two different molecules, and each one has a single type of functional group — but it has two copies of that group. E.g. a dicarboxylic acid and a diol
Products formed
Addition polymerisation
Only the polymer is produced (no by-products).
Condensation polymerisation
Produces the polymer AND a small molecule (water, , etc.).
Functional groups involved
Addition
Only the bond is reactive.
Condensation
Uses functional groups such as:
These determine the type of polymer (polyester, polyamide, etc.).
Check for Understanding
Recall
a) What small molecule is released during condensation polymerisation?
Answer
Water
b) What two functional groups react to form a polyester?
Answer
–COOH and –OH
c) What linkage is formed in a polyester?
Answer
Ester linkage.
Application
d) Explain why monomers must have two functional groups.
Answer
So chain can continue at both ends.
e) Describe how a polyester is formed from a dicarboxylic acid and a diol.
Answer
Condensation reaction; ester links formed between –COOH and –OH groups; water removed.
Challenge (HT)
f) Explain how polyamides are formed and identify the bond formed.
Answer
Diamine reacts with dicarboxylic acid; condensation reaction occurs; amide linkage forms between –COOH and –NH₂ groups; water removed.