Naturally Occurring Polymers (Triple Only)
Lajoy Tucker
Teacher

Contents
Amino Acids & Proteins
What Are Amino Acids?
Amino acids are small organic molecules that act as the building blocks of proteins.
Each amino acid contains two key functional groups:
Amino group (–NH₂) - basic / alkaline
Carboxyl group (–COOH) - acidic
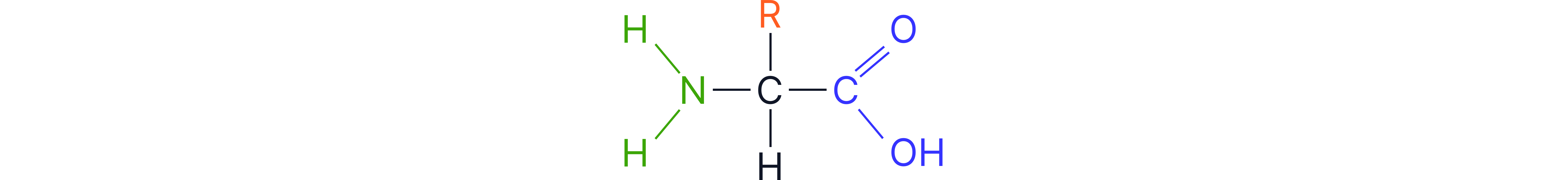
Structural outline of a generic amino acid showing the amino group, carboxyl group, central carbon, and side chain (R group).
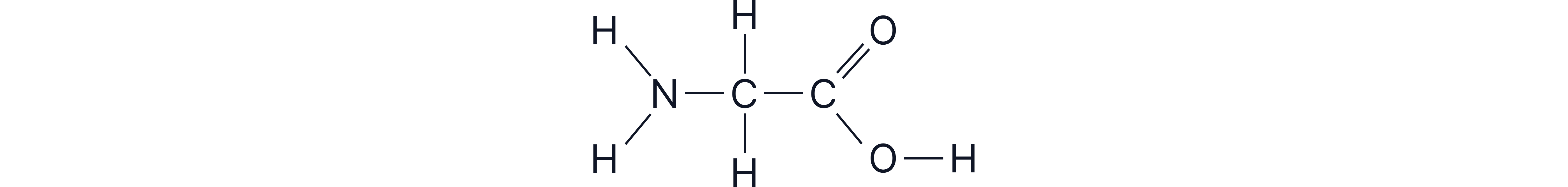
Example
Glycine is the simplest amino acid. It has both the amino and carboxyl groups. The R group in this case is a hydrogen atom.
Forming Polypeptides
Amino acids can chemically join together through a condensation reaction.
During this process:
The amino group of one amino acid reacts with the carboxyl group of another.
A molecule of water is released.
A new C–N bond, known as a peptide bond, forms.
Repeated reactions form long chains called polypeptides.
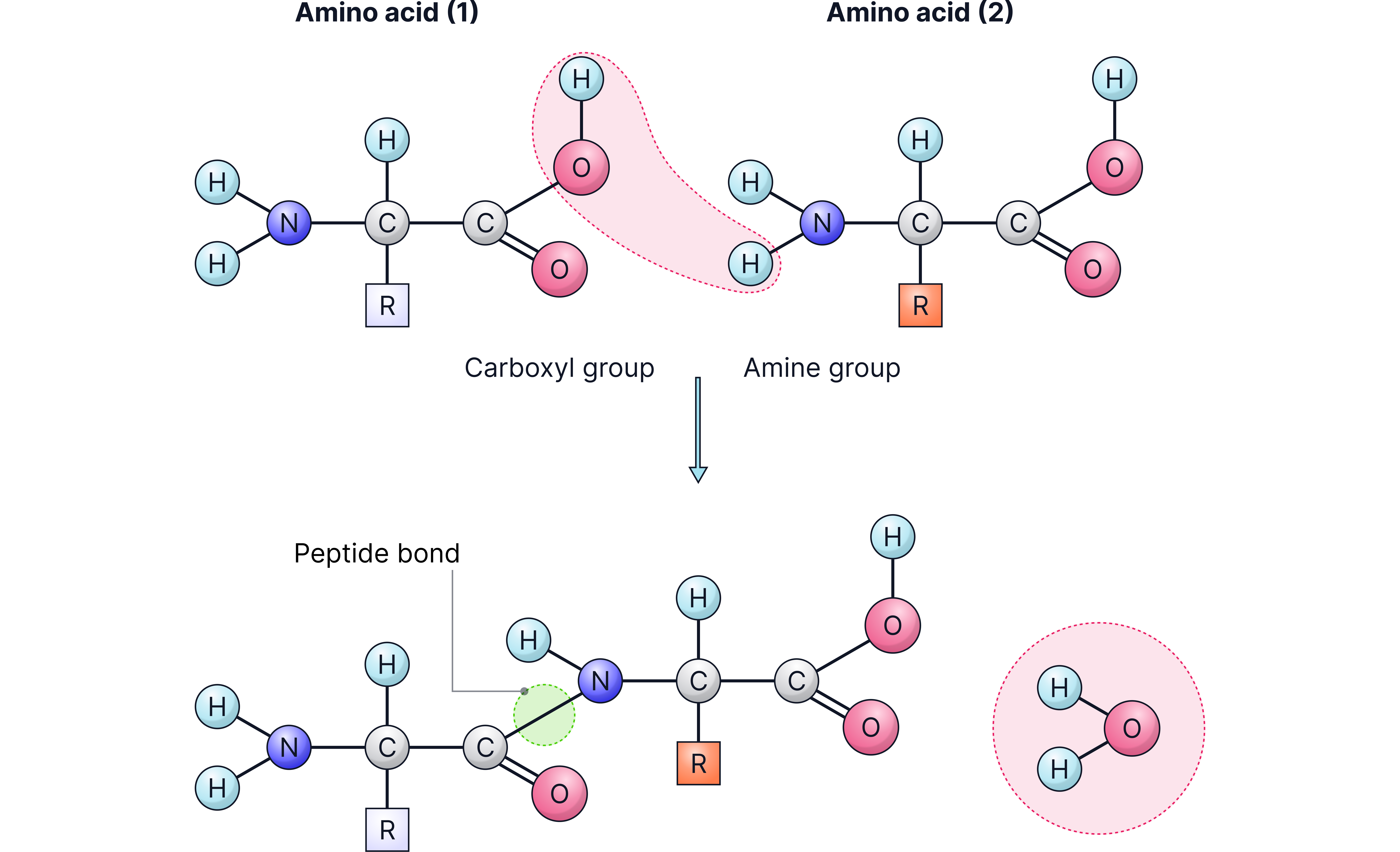
Formation of a peptide link: the carboxyl end of one amino acid bonds with the amino group of another, producing a growing chain.
Proteins
When one or more long polypeptide chains fold into specific shapes, the result is a protein.
Roles of Proteins in Humans
Proteins perform a huge variety of functions, including:
Biological catalysts - enzymes speed up chemical reactions.
Transport - molecules such as those in blood carry gases around the body.
Immune defence - antibodies recognise and neutralise harmful microorganisms.
Structural support - muscle, skin, hair, and many tissues are protein-based.
Why Protein Structure Matters
The order of amino acids determines how the chain folds.
Folding → shape → function.
Different structures = different biological roles.
DNA (Deoxyribonucleic Acid)
What Is DNA?
DNA is the information storage molecule found in almost all living organisms.
It contains coded instructions that control development, function, and inheritance.
DNA has a distinctive double helix shape - two long strands twisted around each other.

A conceptual model of the DNA double helix, showing two linked chains coiled into a stable spiral.
Structure of DNA
DNA is a polymer made from repeating units called nucleotides.
Each nucleotide contains:
1. A sugar molecule
2. A phosphate group
3. A nitrogen-containing base
There are four types of bases:
Adenine (A)
Cytosine (C)
Guanine (G)
Thymine (T)
Base Pairing
Bases form specific pairs across the two strands:
A pairs with T
C pairs with G
These pairings hold the strands together and maintain the helix structure.
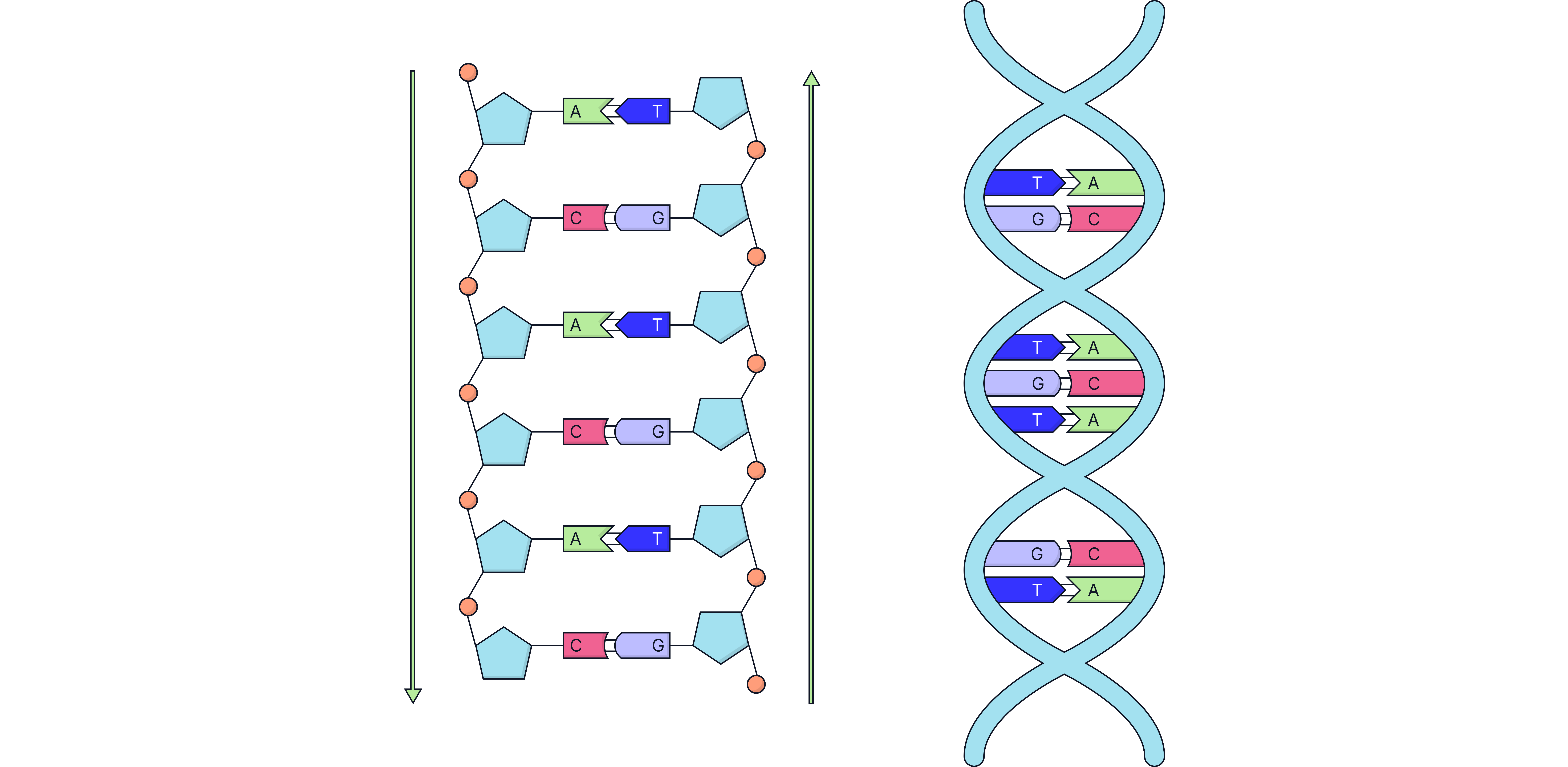
Complimentary bases forming links between the two DNA chains, stabilising the double-helix arrangement.
Genetic Code
The sequence of bases acts like a set of coded instructions for building proteins.
Other Naturally Occurring Polymers
Sugars as Monomers
Certain small carbohydrate molecules (simple sugars like glucose) can act as monomers.
When linked together, they form polysaccharides, which are natural polymers.
Examples of Sugar-Based Polymers
Starch - plants store chemical energy in this form
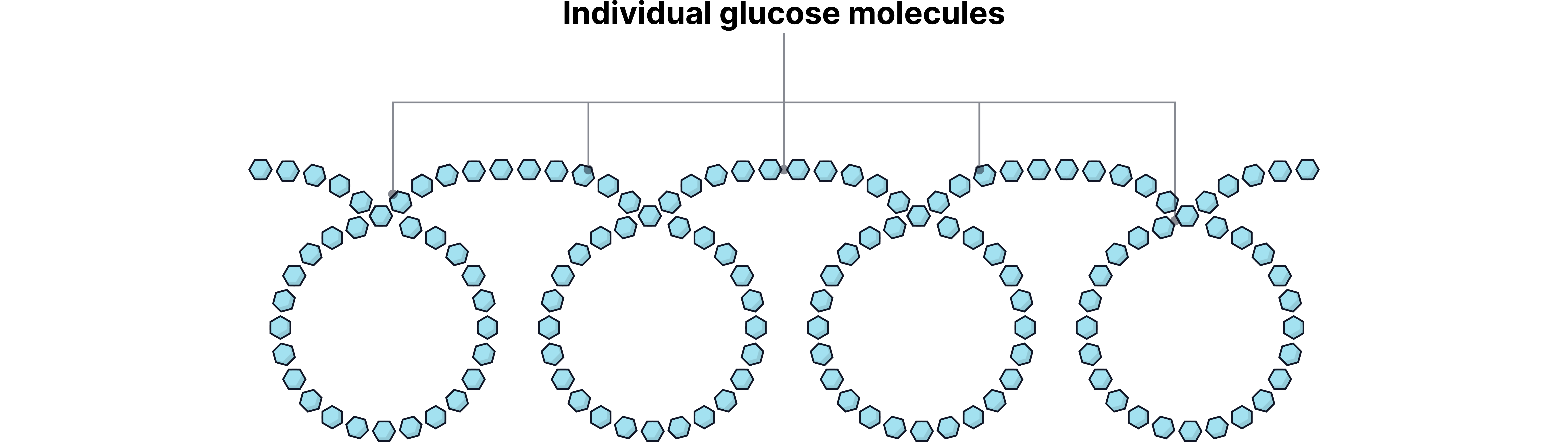
Cellulose - provides strength in plant cell walls
Check for Understanding
Recall
a) Name the two functional groups in amino acids.
Answer
–NH₂ and –COOH
b) What type of bond links amino acids?
Answer
Peptide bonds.
c) State the complementary base pairs in DNA.
Answer
A–T and C–G.
Application
d) Explain how two amino acids form a dipeptide.
Answer
Condensation reaction; peptide bond formed between –NH₂ and –COOH groups; water removed.
e) Describe the basic structure of DNA.
Answer
Polymer of nucleotides; sugar-phosphate backbone; base pairs; double helix structure.
Challenge (HT)
f) Explain how the sequence of bases in DNA determines protein structure.
Answer
Base sequence codes for amino acid order; order determines folding; folding determines function of the protein.