Alkenes & Their Reactions (Triple only)
Lajoy Tucker
Teacher

What Are Alkenes?
Alkenes are a group of hydrocarbons that belong to a family called a homologous series. Every alkene contains:
Only carbon and hydrogen atoms
A carbon–carbon double bond somewhere in its structure
This double bond is the key feature that makes alkenes much more reactive than alkanes.
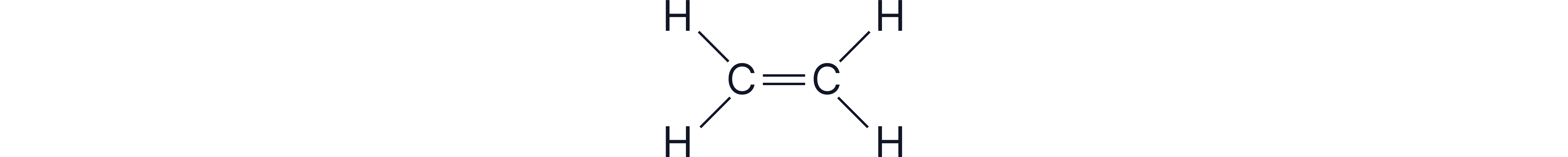
The diagram above shows an example hydrocarbon chain with one double bond. The diagram shows that every carbon still forms four total bonds, while hydrogens form only one.
Why the double bond matters
The double bond has one “normal” bond + one weaker bond layered on top.
That weaker bond can break open, allowing new atoms to attach easily.
This makes alkenes important starting materials in industry for solvents, plastics, and many other chemicals.
Why Alkenes Are Called Unsaturated
Alkenes are said to be unsaturated because the presence of a bond means the molecule contains fewer hydrogen atoms than the equivalent alkane.
Saturated = full of hydrogen (no double bonds)
Unsaturated = fewer hydrogens because of a double bond
If the double bond were “opened up,” the carbon atoms could hold additional hydrogen atoms. This is why alkenes readily take part in addition reactions.
General Formula & Recognising Alkenes
All straight-chain alkenes follow the formula:
Examples:
Hexene
Heptene
Octene
To check if a compound could be an alkene:
1. Does the number of hydrogens equal twice the number of carbons?
2. Or does its structural formula show a bond?
If yes it is an alkene.
Structures of the First Alkenes
Here are verbal descriptions (instead of diagrams) using different examples from the original source:
1. Ethene ()
Two carbons double-bonded, with each carbon holding two hydrogens.
2. Propene ()
Three-carbon chain, double bond between carbon 1 and carbon 2.
3. butene ()
Four-carbon chain with the double bond starting at carbon 2.
4. pentene ()
Five-carbon chain; double bond position can vary (e.g. carbon 3).
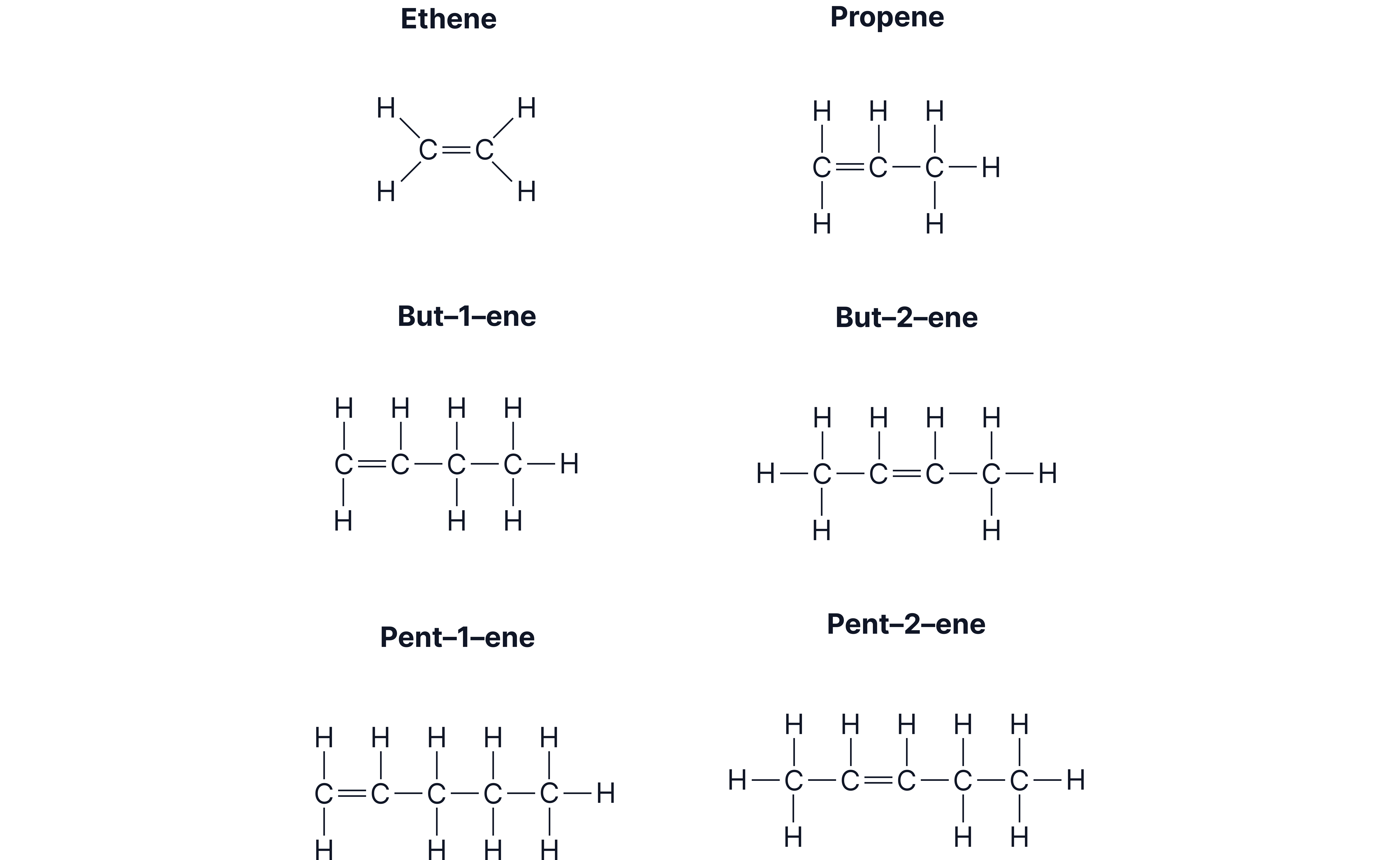
For butene and pentene the double bond can appear in different places.
Distinguishing Alkenes From Alkanes (Bromine Water Test)
Because alkenes react easily at the bond, they can be identified using bromine water.
Procedure:
Add a small amount of orange bromine water to the substance being tested.
If the solution quickly loses its orange colour, an alkene is present.
Why this happens
The double bond reacts with bromine, forming a colourless product.
Alkanes do not do this - they leave the orange colour unchanged.
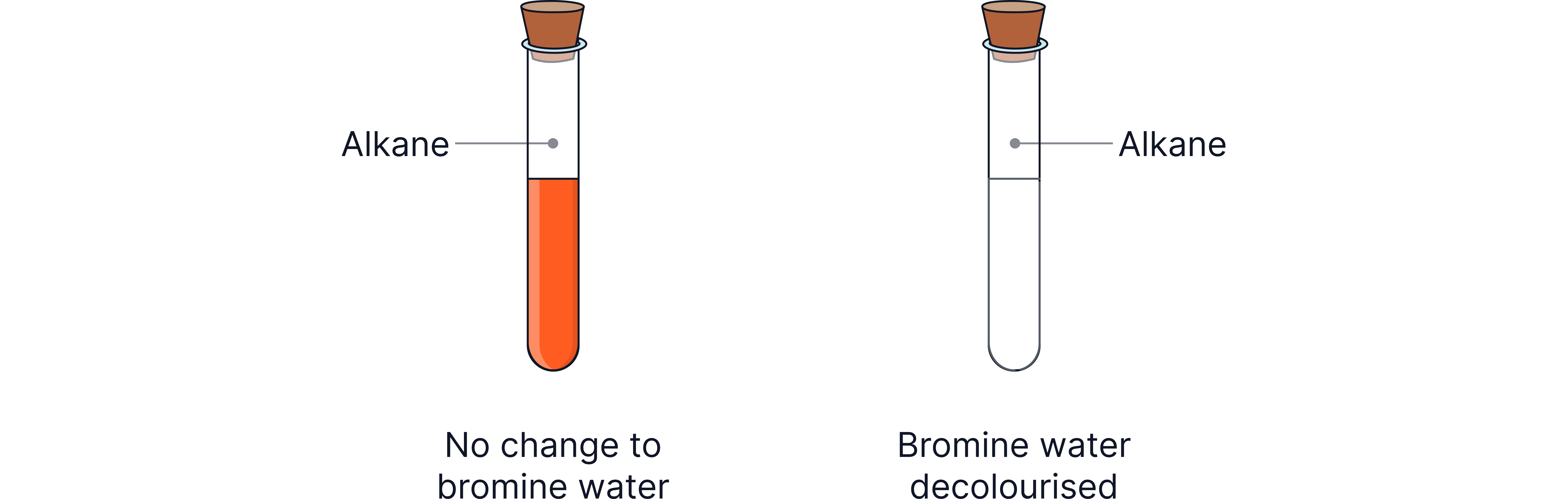
The diagram above shows two small containers side by side: one showing the starting orange bromine solution, and another showing the mixture turning colourless after contacting an alkene.
Combustion of Alkenes
Complete combustion
With plenty of oxygen, alkenes burn to form:
Carbon dioxide
Water
Incomplete combustion
When oxygen supply is limited, alkenes burn with a smoky flame, producing:
Carbon monoxide
Solid carbon (soot)
Possibly some unburned hydrocarbons
This is typical for all hydrocarbons with high carbon content.
Reactivity of Alkenes: The Double Bond in Action
Most reactions of alkenes involve addition reactions.
An addition reaction occurs when:
The bond opens → two new atoms/groups attach to the formerly double-bonded carbons.
This mechanism is the foundation for:
Hydrogenation
Hydration
Addition of halogens
Many polymerisation reactions
Addition Reactions in Detail
Hydrogenation (Adding Hydrogen)
When an alkene reacts with hydrogen:
The double bond opens
Two hydrogen atoms are added
A saturated alkane is formed
Typical conditions
Hydrogen gas
Metal catalyst (nickel, platinum, or palladium)
Warm conditions
Example:
Propene → Propane
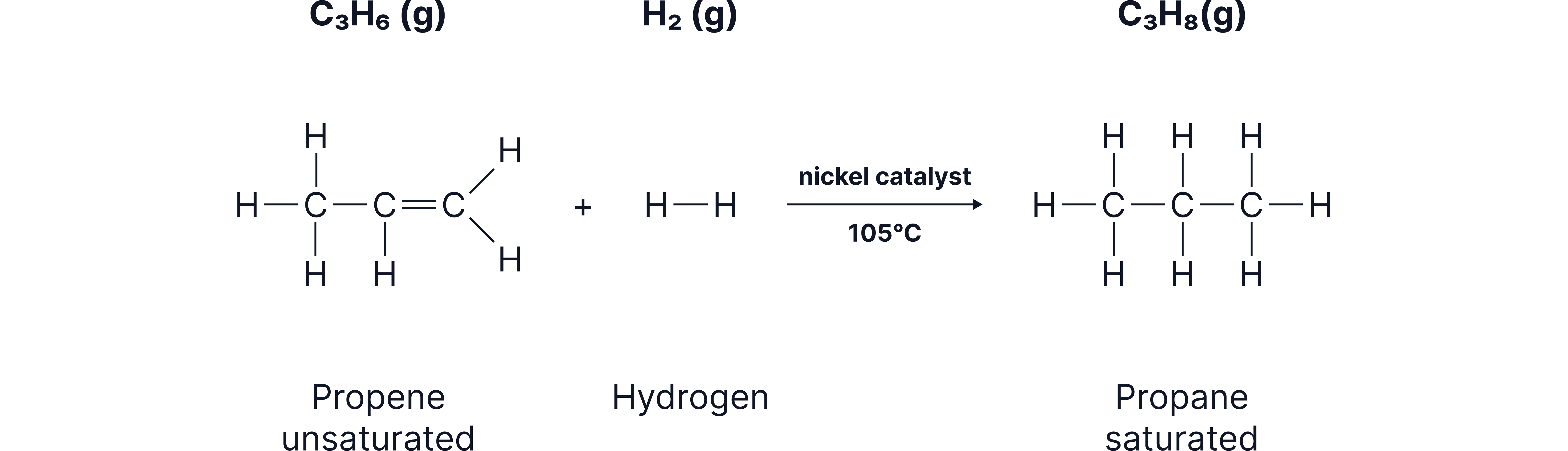
A before-and-after diagram showing an alkene becoming an alkane as hydrogen atoms attach to each carbon of the previous double bond.
Hydration (Adding Water to Form Alcohols)
Steam can be added across the double bond to produce an alcohol.
Conditions
Steam
Catalyst (often phosphoric acid)
High temperature and pressure
Example Reaction
Ethene + steam → ethanol
The -OH group can attach to either carbon of the original double bond.
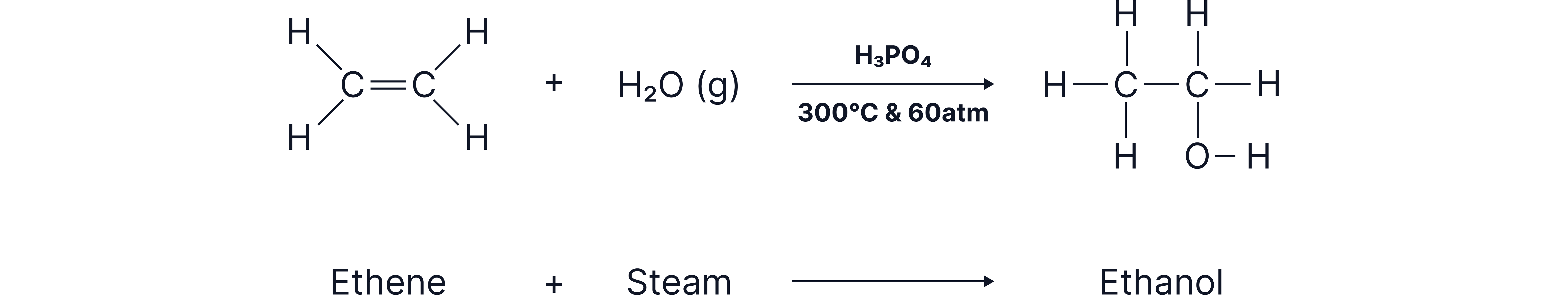
Addition of Halogens
Alkenes react rapidly with halogens such as:
Chlorine
Bromine
Iodine
What happens
The double bond breaks
Each carbon attaches to a halogen atom
A saturated dihaloalkane is formed
Different Example
Pentene + chlorine → dichloropentane
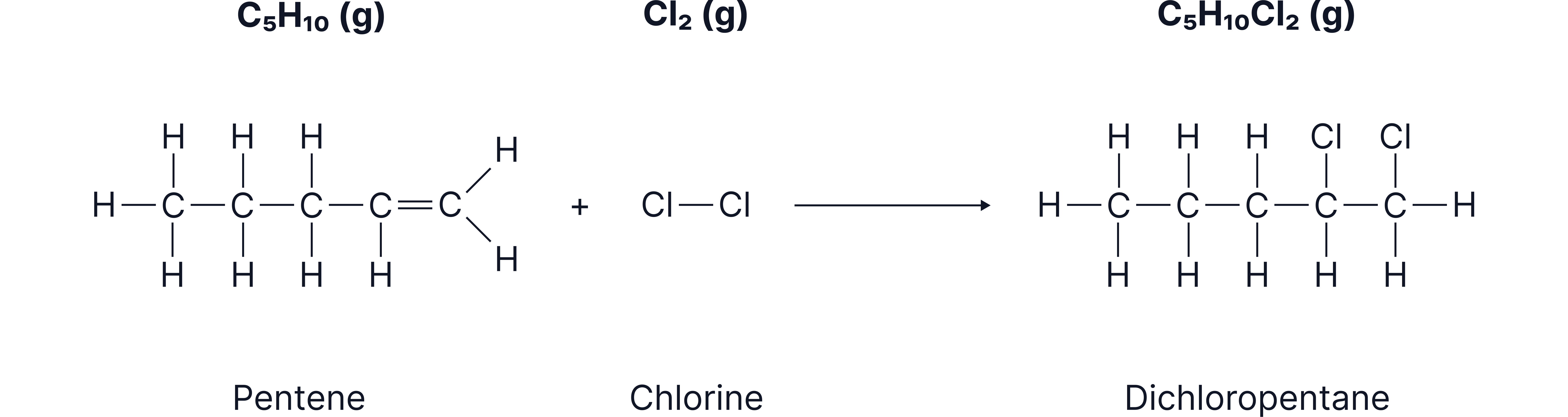
Diagram showing a halogen molecule approaching the double bond, followed by the final product in which both halogen atoms are attached to the carbon chain where the double bond was.
Summary Table of Key Reactions
Reaction Type | What Is Added? | Product Formed | Conditions Needed |
Hydrogenation | Alkane | Metal catalyst, warmth | |
Hydration | (steam) | Alcohol | Catalyst + high T/P |
Addition of Halogens | Dihaloalkane | No special conditions | |
Combustion | CO₂ + H₂O (or CO/soot) | Plenty/limited oxygen |
Check for Understanding
Recall
a) State the general formula for alkenes.
Answer
CₙH₂ₙ
b) What functional group do alkenes contain?
Answer
C=C double bond.
c) What is the result of the bromine water test?
Answer
Colour changes from orange to colourless.
Application
d) Predict the product when ethene reacts with hydrogen.
Answer
Ethane.
e) Name the type of reaction when alkenes form polymers.
Answer
Addition polymerisation.
Challenge (HT)
f) Explain why alkenes are more reactive than alkanes.
Answer
Double bond; high electron density; breaks to form two bonds.