Alcohols (Triple Only)
Lajoy Tucker
Teacher

Contents
Introduction to Alcohols
Alcohols are an important class of organic molecules widely used as fuels, solvents, disinfectants and in beverages.
All alcohols share one key feature: the –OH functional group (also called a hydroxyl group).
What Makes an Alcohol an Alcohol?
The Functional Group
Every alcohol contains an -OH group attached to a carbon atom.
This -OH group gives alcohols their characteristic reactions and properties.
Diagram Placeholder 1
Title: Structure of the hydroxyl (-OH) group
Caption: “A simplified sketch showing an oxygen atom bonded to a hydrogen atom, highlighting the functional group that defines alcohols.”
The Homologous Series of Alcohols
Alcohols follow a general formula:
As you go down the series:
The number of carbon atoms increases by one each step.
Chemical behaviour is very similar because the -OH group stays the same.
First Four Alcohols You Need to Know
| Name | Number of Carbons | Chemical Formula | Displayed Formula |
| Methanol | 1 | 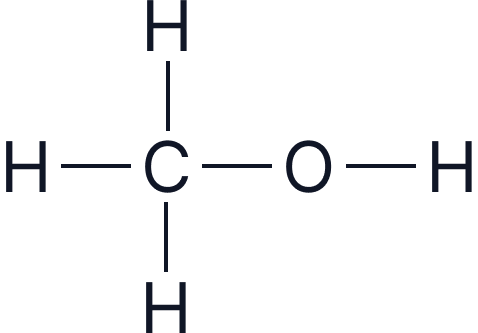 |
|
| Ethanol | 2 | 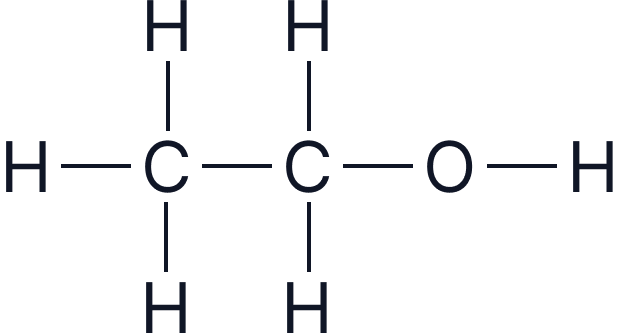 |
|
| Propanol | 3 | 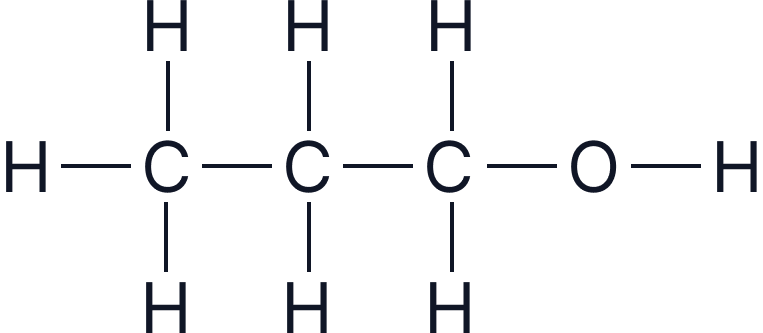 |
|
| Butanol | 4 | 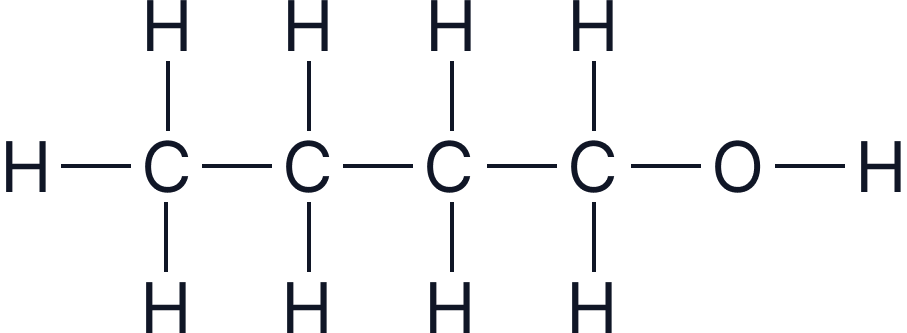 |
Caption: “Four separate structural diagrams showing carbon chains of increasing length with an –OH group attached at the end, demonstrating the pattern in the homologous series.”
Naming Alcohols
Alcohol names end in –ol.
Steps for Naming
1. Count the longest carbon chain.
2. Use the alkane prefix (meth-, eth-, prop-, etc.).
3. Replace the ending -ane with –ol.
4. If the -OH group can appear in different positions, include a number (e.g., butan-2-ol).
Representing Alcohols
Alcohols can be shown in:
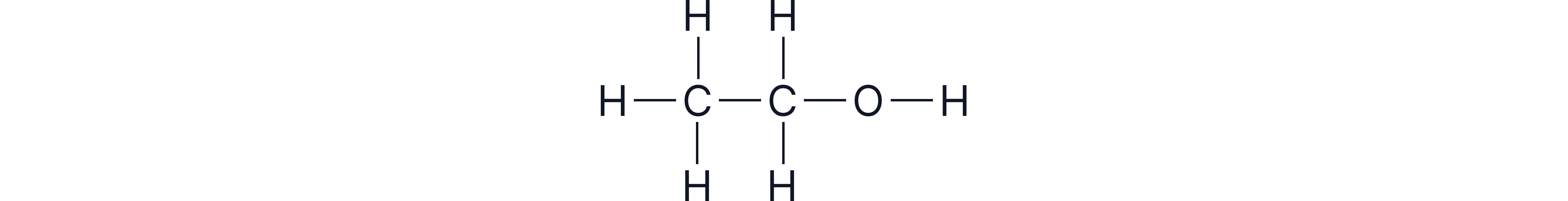
Chemical formula (e.g., C₂H₅OH)
Structural formula (e.g., CH₃CH₂OH)
Displayed formula
Key Reminder
Always include the -OH group when writing formulas - leaving it out changes the compound completely.
Recognising Alcohols
You can identify an alcohol because:
It contains -OH
Its name ends in -ol
Example
CH₃CH₂CH₂OH → This contains an –OH group so it an alcohol and the name ends in -ol. As there are three carbon atoms in the molecule we use the prefix-prop and the suffix-ol to give the name propanol.
Properties and reactions of First Four Alcohols
Physical and Chemical Properties
1. Combustion
Alcohol + oxygen → carbon dioxide + water
Example (for ethanol):

An illustration of a small beaker containing an alcohol fuel producing a blue flame, representing its clean combustion.
2. Solubility
The first four alcohols dissolve completely in water because the –OH group forms hydrogen bonds with water.
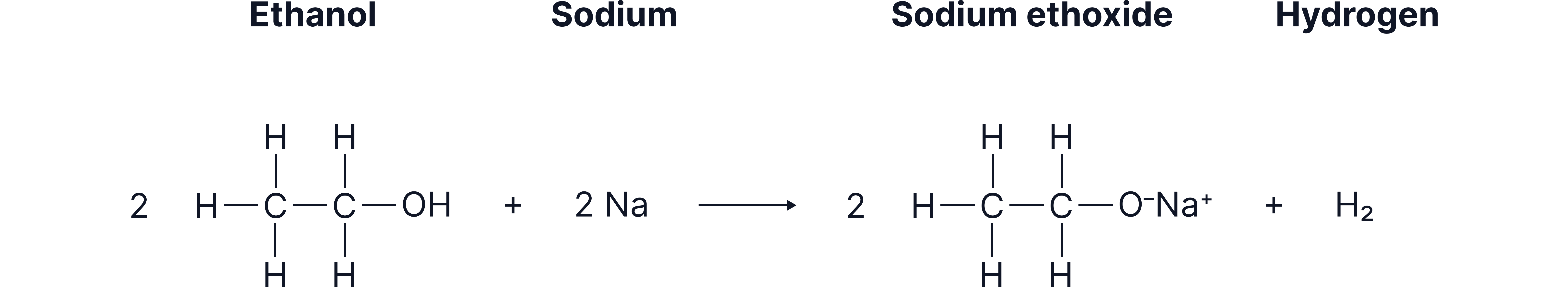
3. Reaction with Sodium
They form hydrogen gas and sodium alkoxides (similar to water, but less vigorous).
4. Oxidation
Alcohols can be oxidised in air (or with oxidising agents) to form carboxylic acids.
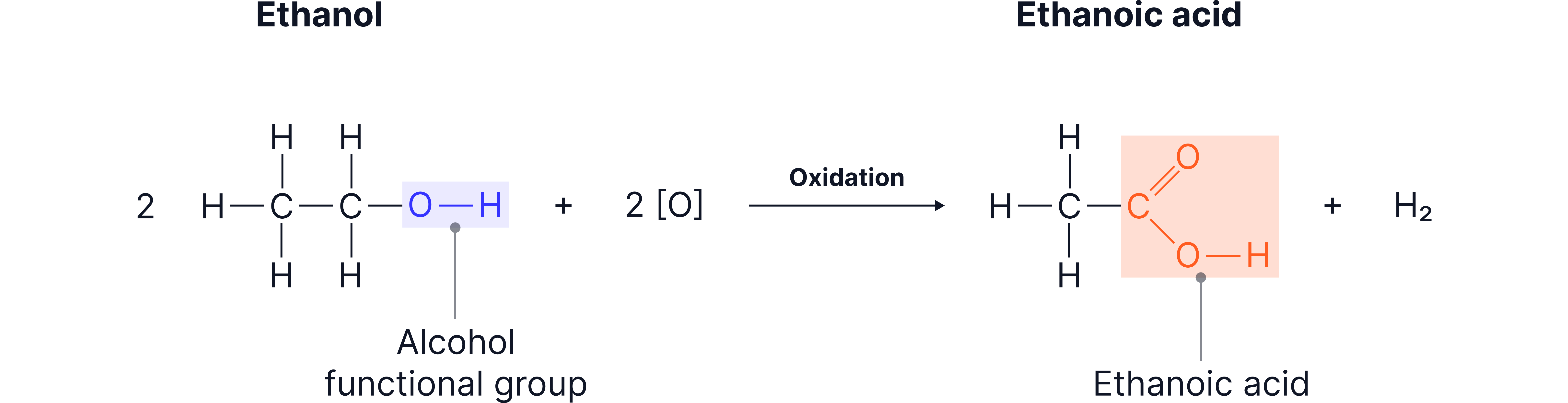
Caption: “Ethanol converting into a ethanoic acid when exposed to oxygen, illustrating loss of hydrogen and gain of oxygen.”
Uses of Alcohols
Alcoholic Drinks
Ethanol is the type of alcohol found in beverages (produced by fermentation).
Solvents
Alcohols can dissolve both:
Water-soluble substances
Substances that water cannot dissolve (e.g., oils)
This dual solubility makes them useful in perfumes, inks and cleaning agents.
Fuels
Ethanol burns with a clean flame.
Used in spirit burners and blended into car fuel to reduce pollution.
Producing Ethanol by Fermentation
The Fermentation Reaction
Yeast enzymes convert sugar into ethanol and carbon dioxide:
Conditions Needed
Temperature around 37 °C
Slightly acidic solution
Anaerobic (no oxygen)
If too hot or too cold enzymes denature or work slowly
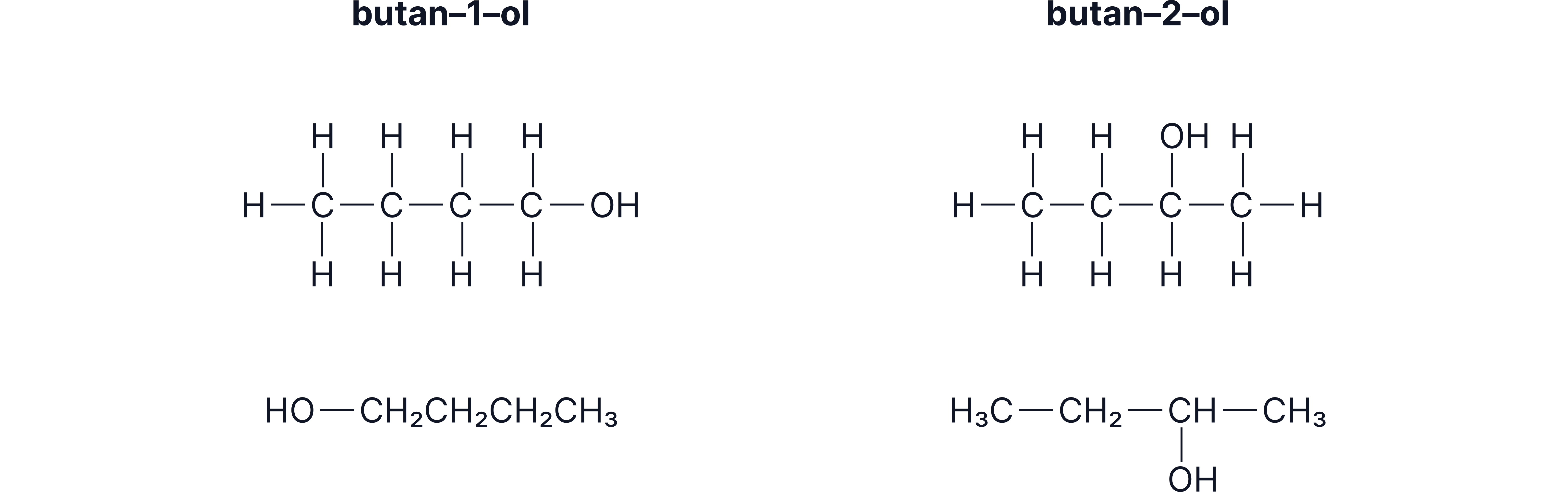
Isomerism: Butan-1-ol vs Butan-2-ol
Some alcohols have more than one structural form because the -OH group can attach to different carbons.
Butan-1-ol: –OH on carbon 1
Butan-2-ol: –OH on carbon 2
Check for Understanding
Recall
a) What functional group is in alcohols?
Answer
–OH
b) State the general formula for an alcohol.
Answer
CₙH₂ₙ₊₁OH
c) What are the conditions for fermentation?
Answer
~37°C; anaerobic; yeast.
Application
d) Write the word equation for combustion of ethanol.
Answer
Ethanol + oxygen → carbon dioxide + water.
e) Name CH₃CH₂CH₂OH.
Answer
Propanol.
Challenge (HT)
f) Explain structural isomerism using pentan-1-ol and pentan-2-ol.
Answer
Same molecular formula (C₅H₁₁OH); different structure/connectivity of atoms; –OH position differs.