Carboxylic Acids (Triple only)
Dr. Davinder Bhachu
Teacher

Contents
Carboxylic Acids (Triple only)
Introduction: What Are Carboxylic Acids?
Carboxylic acids are a family of organic compounds that all contain the carboxyl group, written as –COOH.
This functional group is what gives the molecules their acidity and their characteristic reactions.
Breaking down the –COOH group
- The carbon atom forms:
- A double bond with an oxygen atom.
- A single bond with an –OH group.
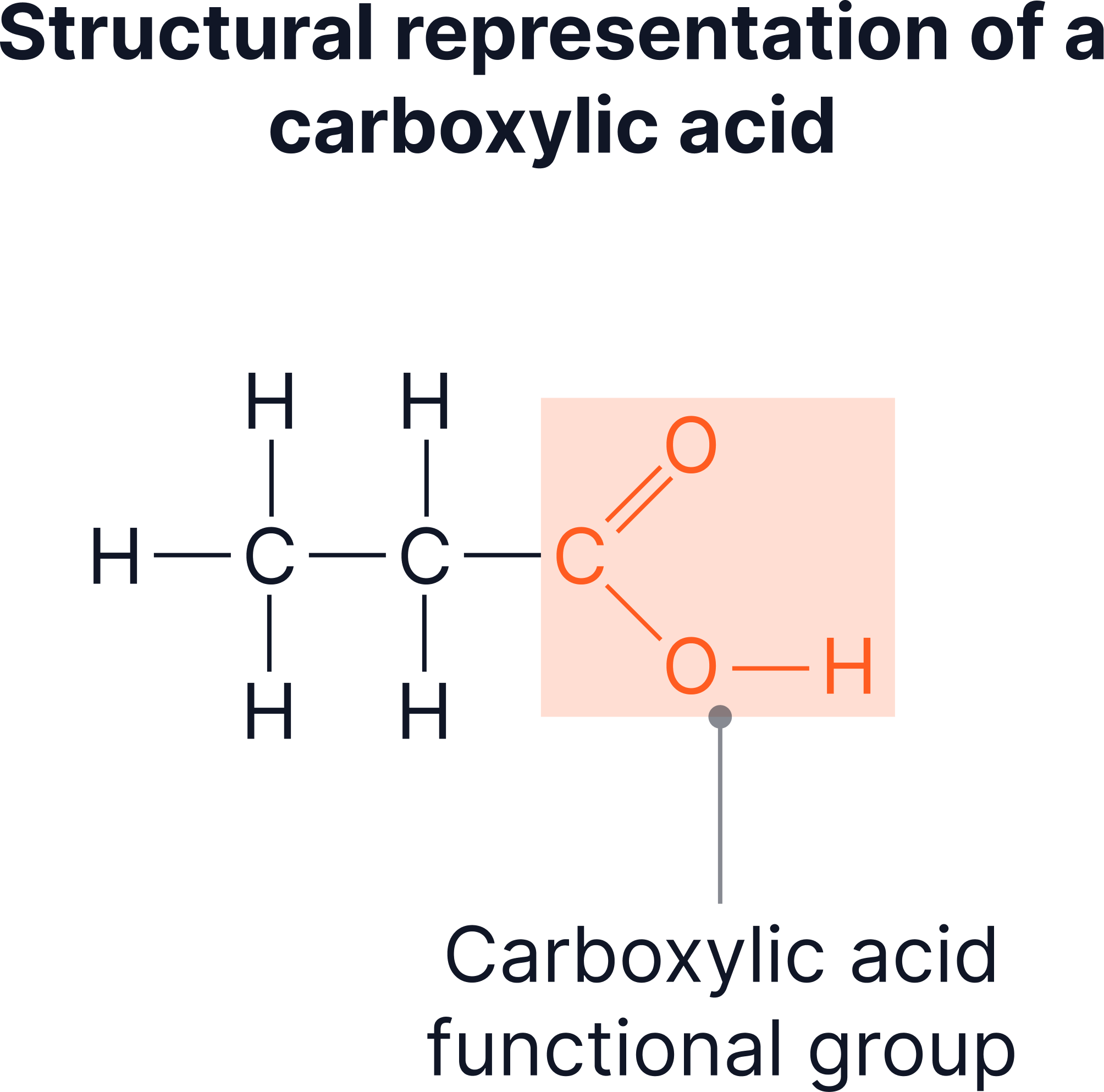
The carboxyl functional group consists of a carbon atom bonded to one oxygen by a double bond and to an –OH group by a single bond. This combination is responsible for the acidic behaviour of carboxylic acids.
Naming The First Four Carboxylic Acids
Names of carboxylic acids end in “-anoic acid” and begin with the standard chain prefixes you already know (meth-, eth-, prop-, but-).
| Name | Formula | Structure (displayed formula) |
| Methanoic acid | HCOOH | 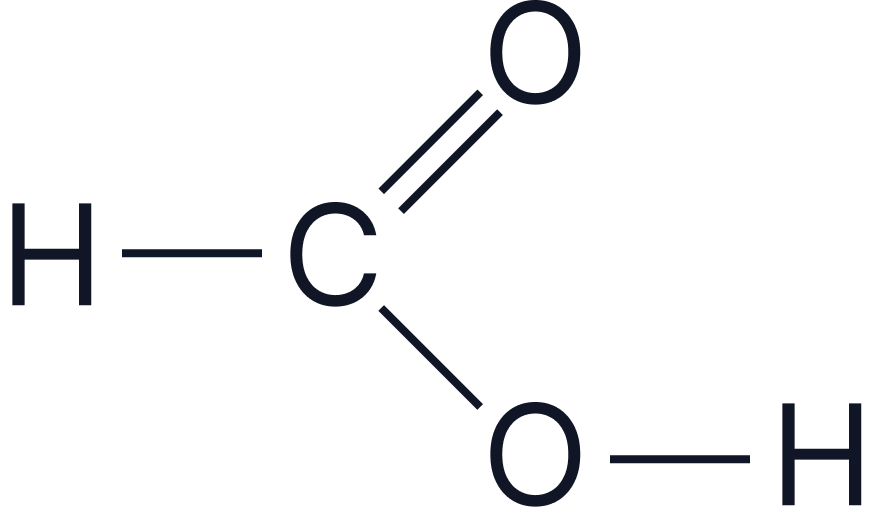 |
| Ethanoic acid | CH₃COOH | 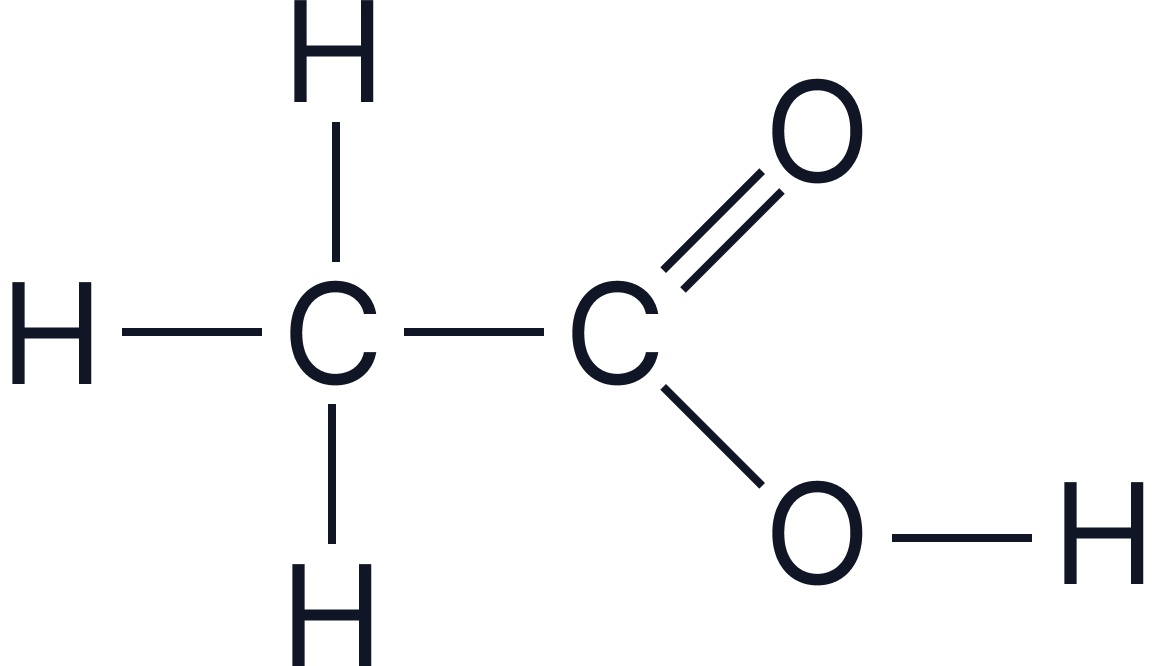 |
| Propanoic acid | C₂H₅COOH | 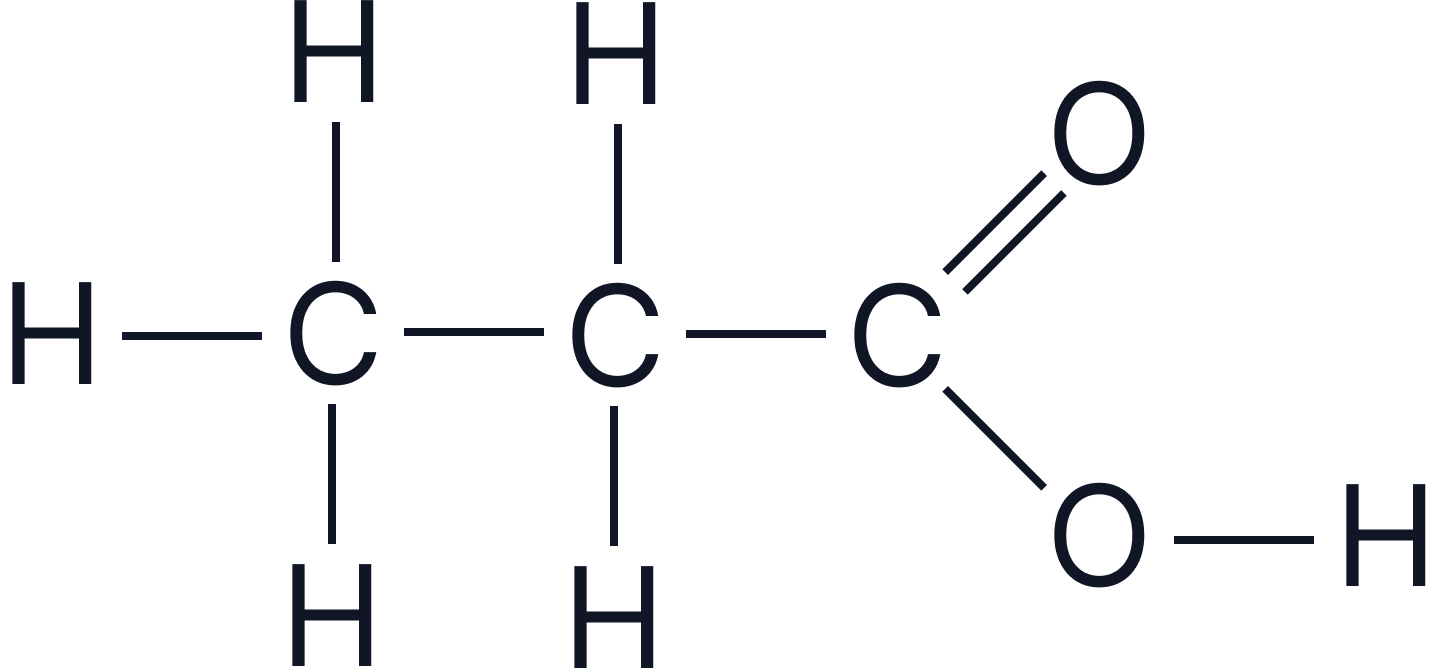 |
| Butanoic acid | C₃H₇COOH | 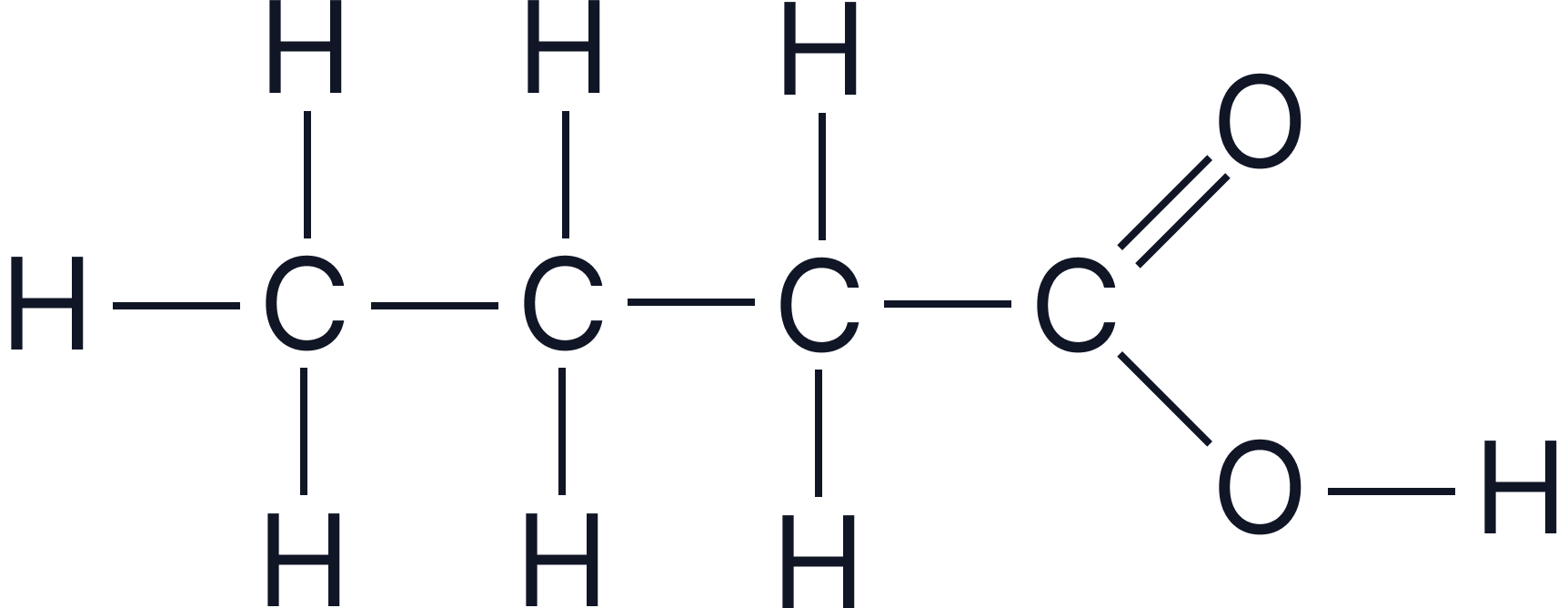 |
A comparison of the displayed structures of the four simplest carboxylic acids, showing how the –COOH group attaches to progressively longer carbon chains.
Identifying Carboxylic Acids
Key idea:
When writing or recognising formulas for carboxylic acids, the –COOH group must remain visible.
For example, ethanoic acid must be written as CH₃COOH, not simply rearranged into an empirical formula.
How to spot one quickly
Look for:
- A –COOH functional group
or - A name ending in “-anoic acid”
Example
Given the structure shown, you can identify the molecule as a carboxylic acid if:
The –COOH group is present, and
The name ends appropriately, such as “2-methylpropanoic acid”.
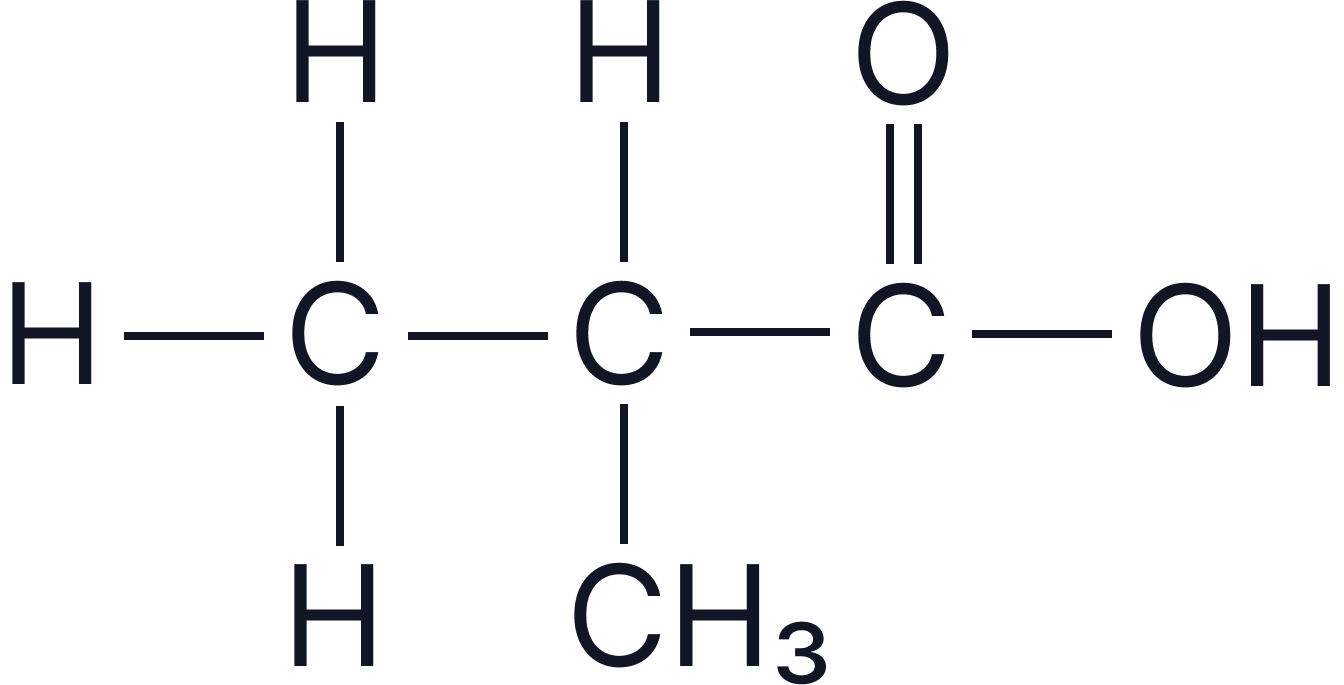
The structure of a branched carboxylic acid (2-methylpropanoic acid). The molecule contains a visible –COOH group and a branching methyl group on the second carbon.
Properties And Reactions Of Carboxylic Acids
Carboxylic acids share a set of characteristic behaviours due to the –COOH group.
Reaction with Carbonates
They react with metal carbonates to produce:
- Carbon dioxide
- Water
- A salt (called a carboxylate) ending in “-anoate”
Example Reaction:
Ethanoic acid + sodium carbonate → carbon dioxide + water + sodium ethanoate
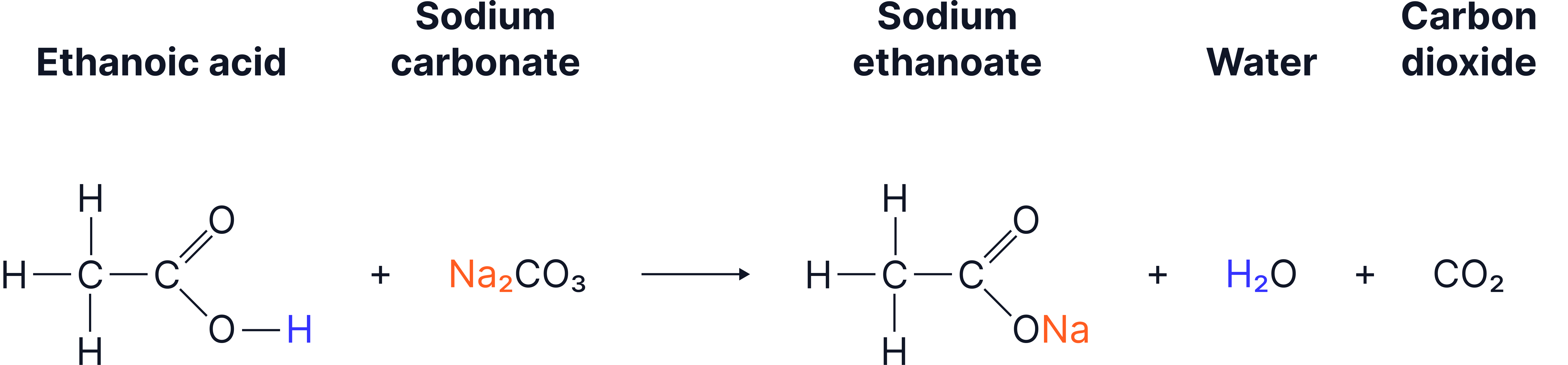
Ethanoic acid reacting with sodium carbonate to generate carbon dioxide gas, liquid water, and the salt sodium ethanoate.
Solubility
Carboxylic acids dissolve in water, and when they do, some molecules release H⁺ ions, making the solution acidic.
Weak Acids
Carboxylic acids are weak acids because they only partly ionise in water.
Therefore, their solutions have higher pH values than solutions of strong acids at the same concentration.
Weak acids occur naturally in foods-here, citric acid contributes to the sour taste of citrus fruits and provides a mildly acidic environment.
Esters
Esters contain the functional group –COO–.
They form when a carboxylic acid reacts with an alcohol, usually in the presence of a strong acid catalyst such as concentrated sulfuric acid.
General Reaction
alcohol + carboxylic acid ⇌ ester + water
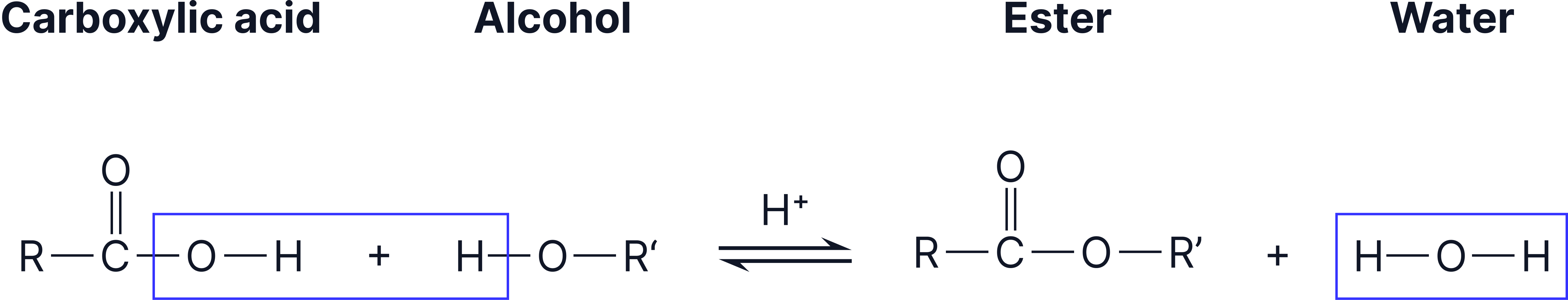 Reaction showing how an alcohol and a carboxylic acid combine under acidic conditions to produce an ester and water.
Reaction showing how an alcohol and a carboxylic acid combine under acidic conditions to produce an ester and water.
Example: Making Ethyl Ethanoate
Ethanol + ethanoic acid —(acid catalyst)→ ethyl ethanoate + water
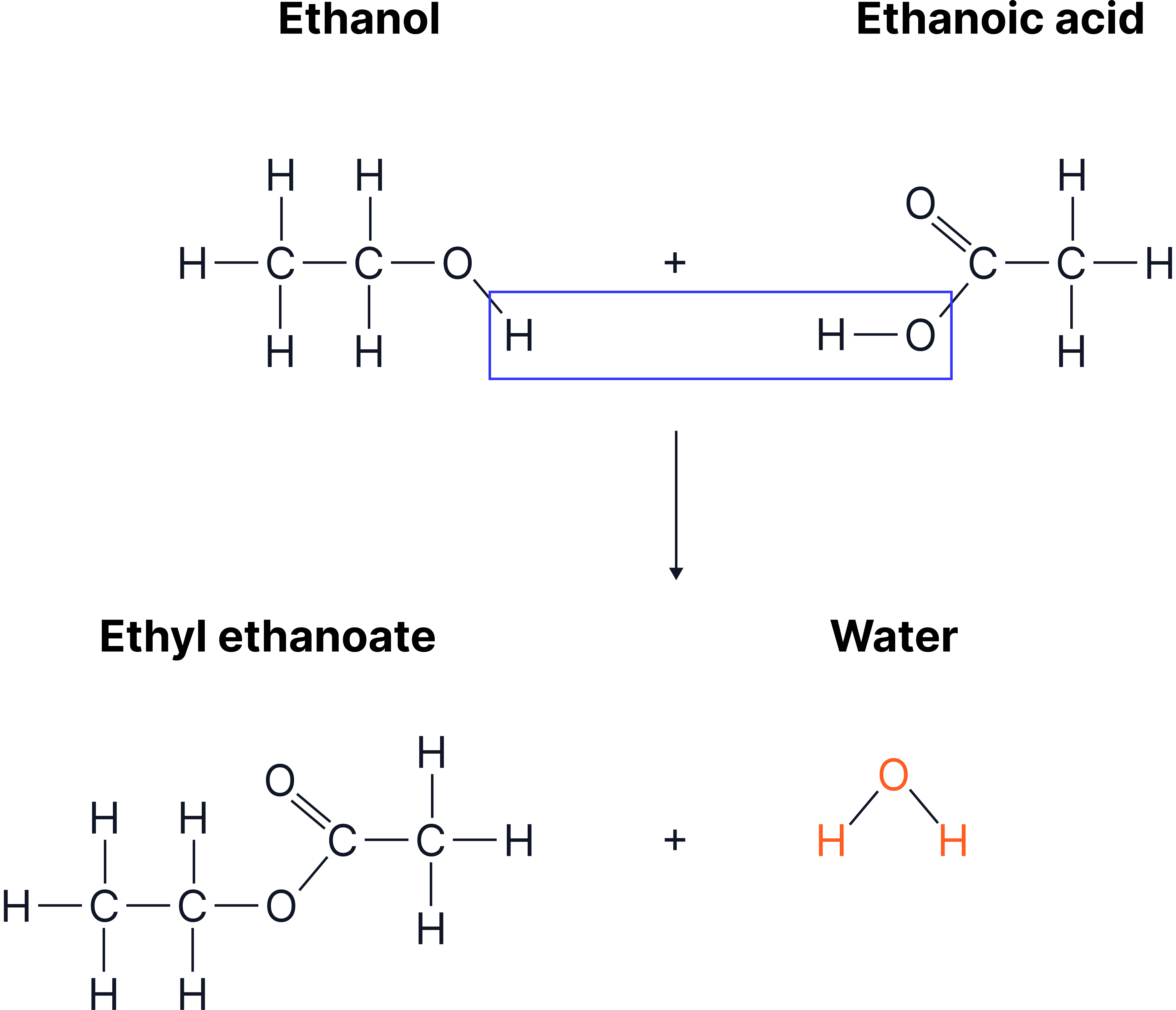
Formation of ethyl ethanoate from ethanol and ethanoic acid. The –OH group of the acid and a hydrogen atom from the alcohol are removed to form water, while the remaining fragments join to create the ester.
How Naming Esters Are Built
General structure:
Alcohol part + Carboxylic acid part
Rule:
- First part (ending in –yl) → comes from the alcohol
- Second part (ending in –anoate) → comes from the carboxylic acid
This is the opposite of how the molecules are joined, so the name must be read carefully.
Step-by-Step Naming Guide
STEP 1 - Identify the alcohol used to make the ester
Remove the –anol ending and replace it with –yl.
|
Alcohol |
Ester prefix |
|
methanol |
methyl |
|
ethanol |
ethyl |
|
propanol |
propyl |
|
butanol |
butyl |
STEP 2 - Identify the carboxylic acid
Remove the –oic acid ending and replace it with –anoate.
|
Acid |
Ester ending |
|---|---|
|
methanoic acid |
methanoate |
|
ethanoic acid |
ethanoate |
|
propanoic acid |
propanoate |
|
butanoic acid |
butanoate |
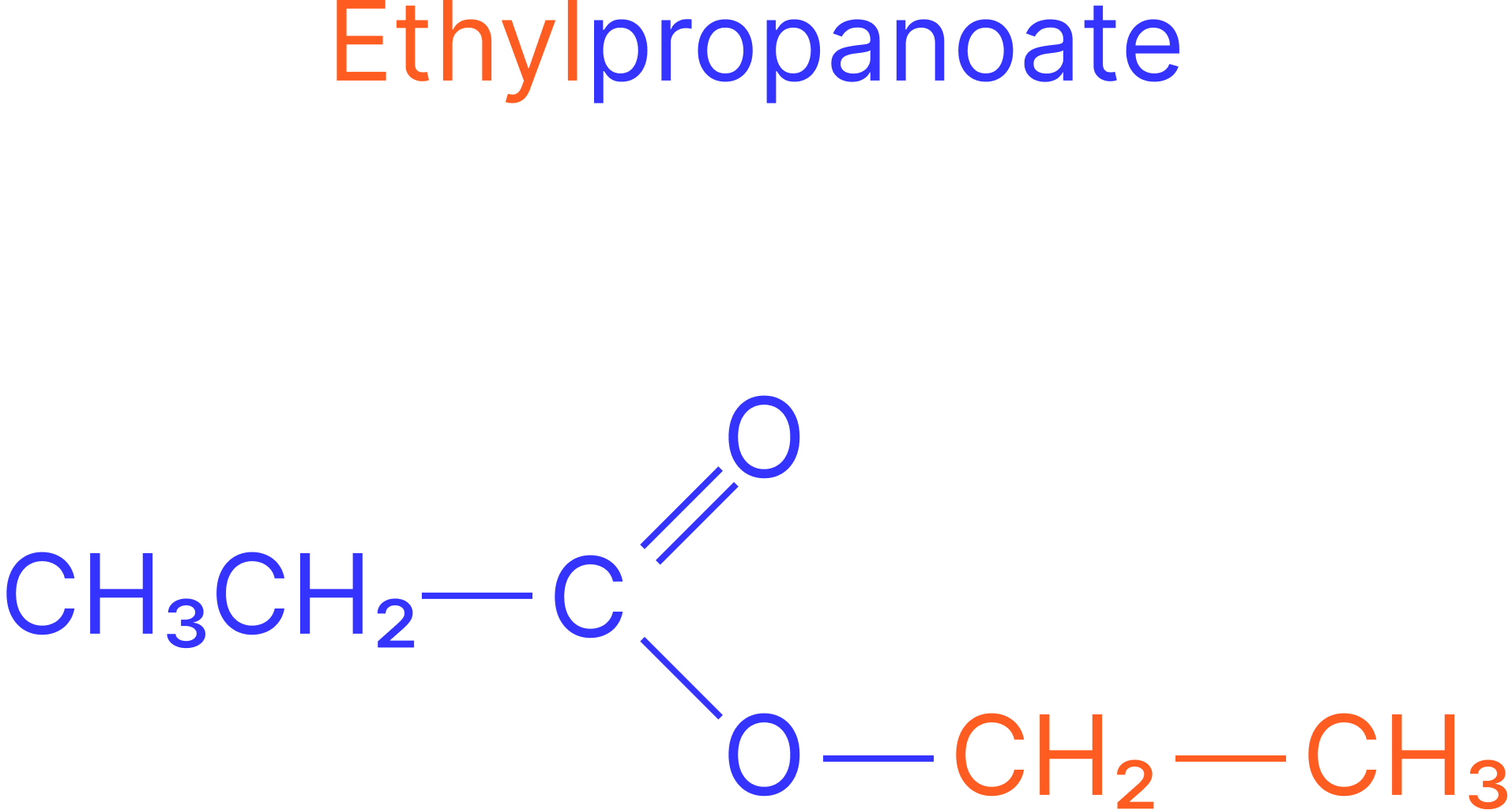
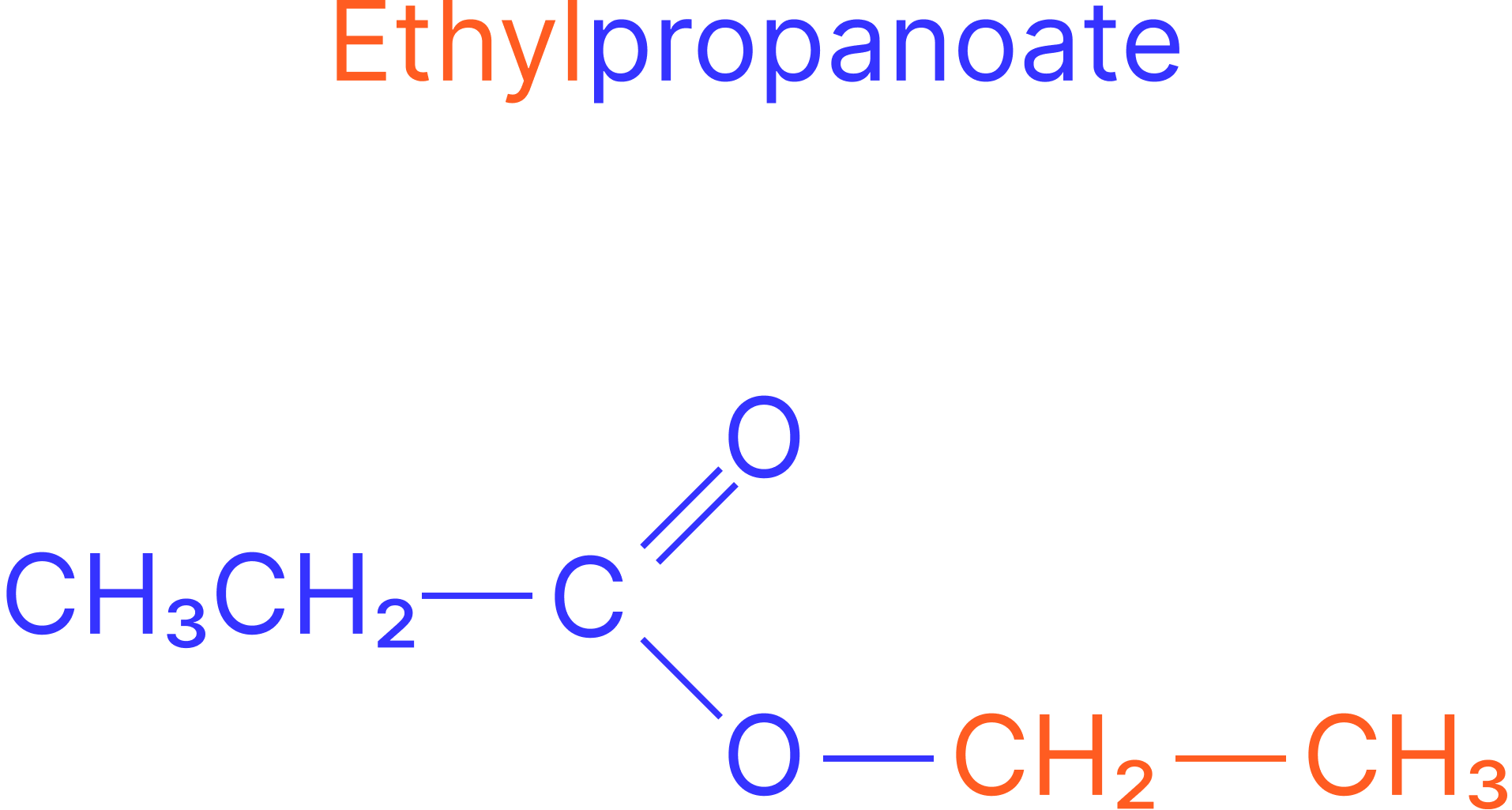
Example:
Alcohol: ethanol
Acid: propanoic acid
Check Your Understanding
Recall
a) What functional group is present in carboxylic acids?
Answer:
–COOH
b) What suffix is used in naming carboxylic acids?
Answer:
–anoic acid
c) What gas forms when reacting with carbonates?
Answer:
Carbon dioxide
Application
d) Write the products formed in this reaction: ethanoic acid + sodium carbonate.
Answer:
Sodium ethanoate + carbon dioxide + water
e) Name ester formed from methanol and butanoic acid.
Answer:
Methyl butanoate
Challenge (HT)
f) Explain how esterification occurs and why it is reversible.
Answer:
Alcohol + acid reacts; condensation reaction happens; water formed; equilibrium reached; reversible as water can hydrolyse (break apart) the ester back into a carboxylic acid and alcohol under acidic conditions
Practice Question
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!