Alloys, Corrosion and its prevention
Emmanuel Opoku
Teacher

Contents
Alloys And Corrosion
What Are Alloys?
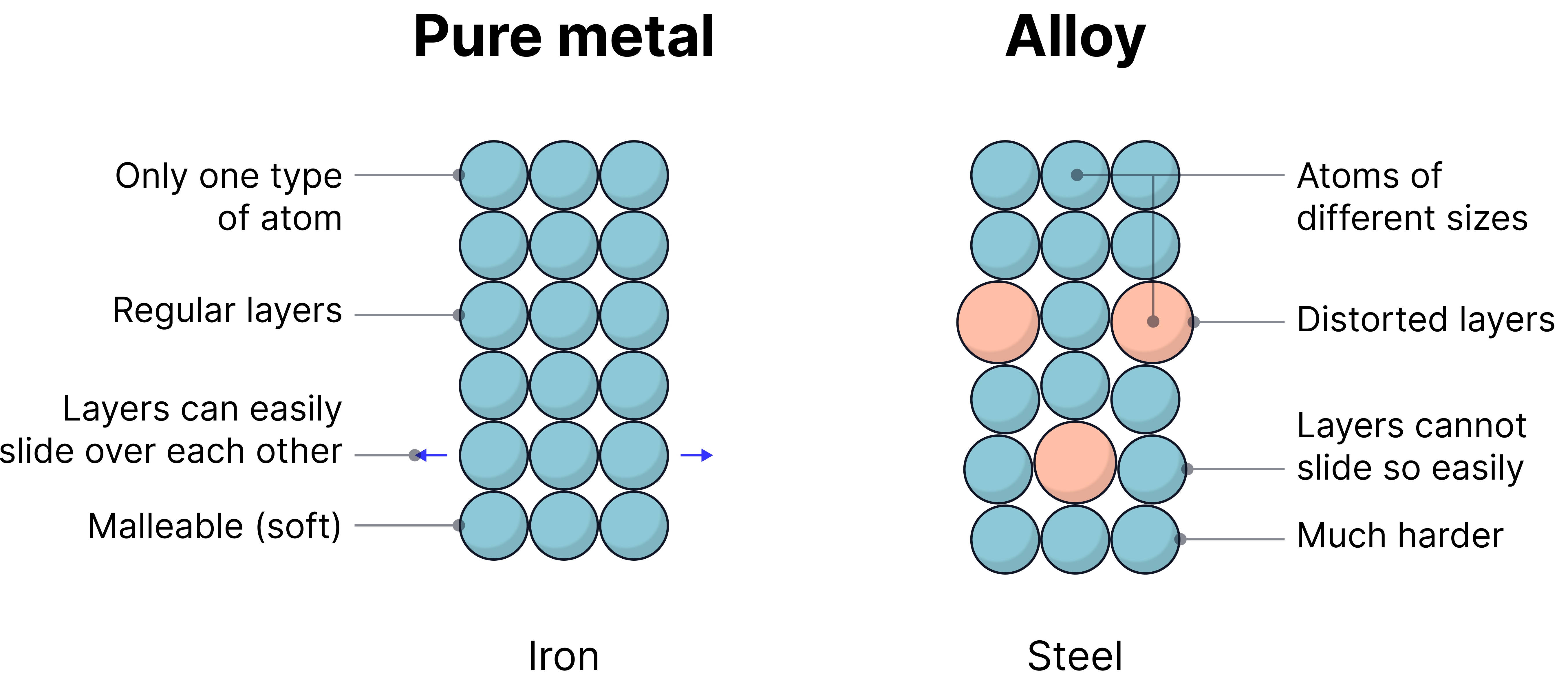
- Alloys are mixtures of two or more elements, where at least one is a metal.
- They are made to improve properties such as strength, hardness, corrosion resistance, and malleability.
- Pure metals have layers of atoms that slide over each other easily, making them soft and malleable.
- When mixed with other elements (becomes an alloy), these layers become distorted, so the metal becomes harder as these layers can no longer slide past each other easily.
Common Alloys And Their Uses
|
Alloy |
Composition |
Properties |
Uses |
|---|---|---|---|
|
Bronze |
Copper + Tin |
Hard, corrosion-resistant |
Statues, medals, decorative objects |
|
Brass |
Copper + Zinc |
Malleable, low friction |
Water taps, musical instruments |
|
Steels |
Iron + Carbon (and other metals) |
Vary with composition |
Tools, car bodies, bridges |
|
Aluminium alloys |
Aluminium + Magnesium |
Low density, strong |
Aircraft manufacture |

Types Of Steel And Their Uses
|
Type of Steel |
Composition |
Properties |
Uses |
|---|---|---|---|
|
High-carbon steel |
Iron + ~2% Carbon |
Very hard, brittle |
Cutting tools |
|
Low-carbon steel |
Iron + <0.3% Carbon |
Soft, easily shaped |
Car panels, construction |
|
Stainless steel |
Iron + Chromium (+ Nickel) |
Resistant to corrosion |
Cutlery, containers |

high carbon steel low-carbon steel stainless steel
Steel’s strength, durability, and versatility make it a key material in everyday life.
Gold Alloys And Carats
- Pure gold (24 carat) is too soft for jewellery.
- Mixed with other metals (e.g. copper, silver, zinc) to increase hardness.
- The carat system shows the fraction of pure gold in the alloy.
![]()
Example:
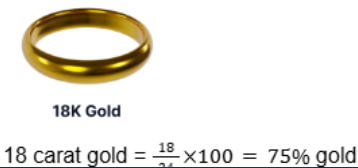
Corrosion And Rusting
- Corrosion is the gradual destruction of a material (usually reactive metals) by reacting with substances in the environment.
- Rusting is a type of corrosion that only happens to iron or steel.
- Rusting requires both air (oxygen) and water:
Iron + oxygen + water → hydrated iron(III) oxide (rust)
- Rust flakes off, exposing fresh iron, so corrosion continues until the metal is destroyed.
Aluminium also reacts with oxygen, but its oxide forms a protective layer that prevents further corrosion.
Rusting Experiments
The experiment shown in the image helps to prove the conditions needed for iron to rust.
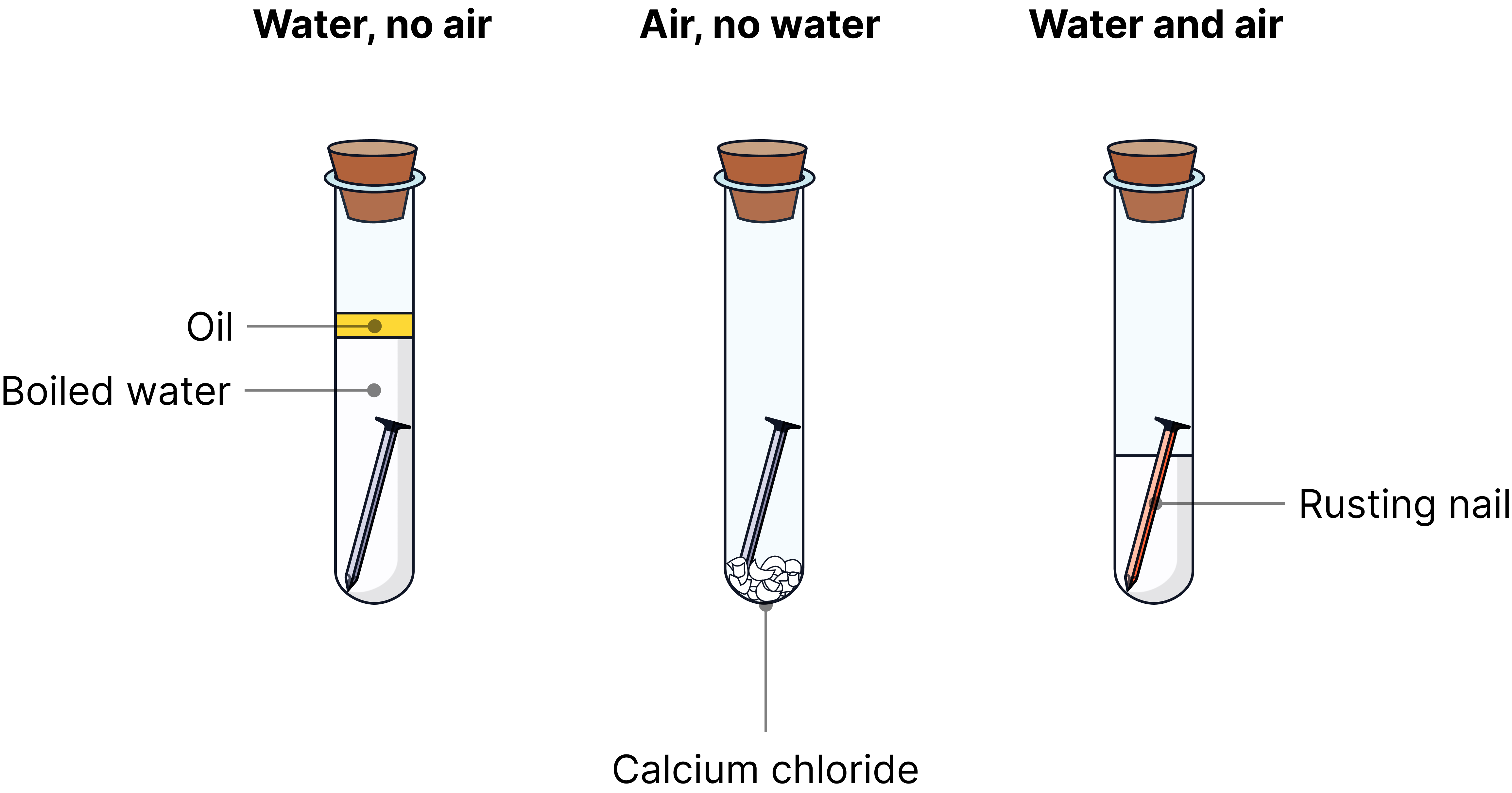
|
Test Tube |
Conditions |
Observations |
Explanation |
|---|---|---|---|
|
1 – Water, No Air |
Air is excluded by the layer of oil |
No rust forms |
The oil prevents oxygen from reaching the water. Rusting cannot occur without air (oxygen). |
|
2 – Air, No Water |
Water is removed by the calcium chloride (drying agent) |
No rust forms |
The calcium chloride absorbs any moisture, so rusting cannot occur without water. |
|
3 – Water and Air |
Both air and water present |
Rust forms on the nail |
Both air (oxygen) and water are needed for rusting. Hydrated iron(III) oxide (rust) forms on the surface of the nail. |
A drying agent is a substance that absorbs water to keep other substances dry, also called a desiccant (similar to small silica gel bags found when buying new bags/trainers to keep our moisture and keep them smelling fresh).
Preventing Corrosion
1. Creating a Barrier
- Painting or plastic coating prevents air and water from reaching the metal.
- Oiling or greasing protects moving parts.
- Electroplating uses electrolysis to coat a metal (e.g. iron) with a thin layer of another metal (e.g. nickel, chromium).
2. Sacrificial Protection
- A more reactive metal (like zinc or magnesium) is attached to the metal being protected (there are normal methods and special methods like galvanising).
- Normal sacrificial protection attaches blocks of a more reactive metal (like magnesium or zinc) to the iron surface - these blocks corrode instead of the iron but do not form a continuous barrier.
3. Mixed Methods Protection
- Galvanising combines both physical barriers and sacrificial protection. By coating the entire surface of iron or steel with a relatively thicker layer (compared to electroplating) of zinc specifically
- The thick coating of zinc acts as a physical barrier to keep out air and water and also provides sacrificial protection by reacting first if the coating is scratched.
Check Your Understanding
Recall
Define an alloy.
Answer:
A mixture of two or more elements, where at least one is a metal
Why are alloys harder than pure metals?
Answer:
The layers of atoms are distorted, disrupting the layers, making it harder for them to slide over each other
What two substances are required for rusting to occur?
Answer:
Both air (oxygen) and water are required
Why doesn’t aluminium corrode like iron does?
Answer:
Aluminium forms a protective oxide layer that prevents further corrosion
What is meant by galvanising?
Answer:
Coating iron or steel with zinc to prevent rusting by sacrificial protection
Explain why high-carbon steel is not used to make car bodies.
Answer:
It is hard but brittle, so it would crack under stress or impact
Apply Your Knowledge
A gold ring is described as 9 carat gold. Calculate the percentage of gold in the ring.
Answer:
924×100=37.5%gold
Challenge (HT Only)
Use the data below to decide which alloy is most suitable for making the frame of a suspension bridge. Justify
your choice using the data below and your knowledge of materials.
|
Alloy |
Density (g/cm³) |
Strength (MPa) |
Corrosion Resistance |
Cost (£/kg) |
Ease of Shaping |
|---|---|---|---|---|---|
|
Stainless Steel |
8.0 |
550 |
Excellent |
2.0 |
Moderate |
|
Aluminium Alloy |
2.7 |
350 |
Good |
1.5 |
Easy |
|
Titanium Alloy |
4.5 |
900 |
Excellent |
8.0 |
Difficult |
Answer:
-
Titanium alloy has the highest strength (900 MPa) and excellent corrosion resistance, ideal for long-lasting outdoor structures like a bridge.
-
However, it is very expensive (£8/kg) and difficult to shape.
-
Aluminium alloy is easy to shape nd quite cheap; however, it is not as strong, which is not ideal for a bridge.
-
Stainless steel offers a good compromise — strong (550 MPa), corrosion-resistant, and more affordable.
-
Therefore, stainless steel is the most suitable material overall.
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!