Ceramics, composites and polymers
Emmanuel Opoku
Teacher

Choosing Materials
Overview
Different materials have unique properties that make them suitable for specific uses. The structure, bonding, and composition of a material determine whether it is strong, flexible, brittle, heat-resistant, or conductive.
You must be able to:
Describe the properties and uses of ceramics, composites, polymers, and metals.
Interpret data to choose the most suitable material for a purpose.
Ceramics
Definition:
Non-metallic solids with high melting points that are insulators of heat and electricity. They are hard, stiff, and brittle.

Clay Ceramics
Made from mouldable clay that hardens when heated in a kiln.
Examples: Pottery, bricks, tiles.
Properties:
Hard and durable once fired.
Can withstand high temperatures.
Brittle (can shatter easily).
Use: Ideal for building materials because they are strong, cheap, and heat-resistant.
Glass
Generally transparent and brittle, can be moulded when molten.
Soda-lime glass: Made by heating sand, sodium carbonate, and limestone.
Borosilicate glass: Made from sand and boron trioxide, melts at a higher temperature, useful for laboratory glassware and cooking.
Composites
Definition:
Made from two or more materials combined to produce a material with improved properties.
Matrix/Binder: Holds the material together.
Reinforcement: Provides strength or rigidity.
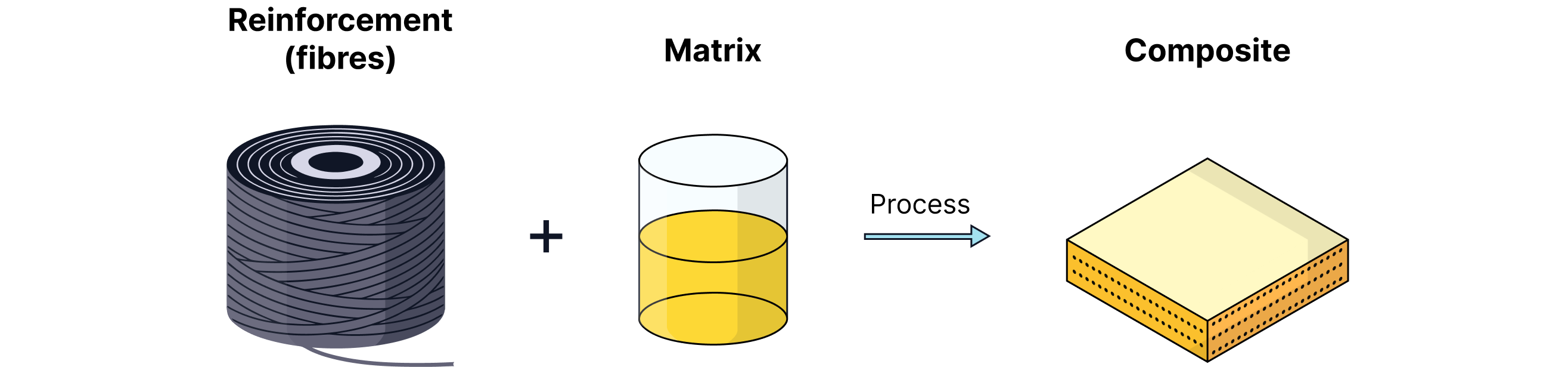
Examples of Composites
Composite | Matrix | Reinforcement | Properties | Uses |
|---|---|---|---|---|
Fibreglass | Polymer | Glass fibres | Low density, strong | Boats, skis |
Carbon fibre | Polymer | Carbon fibres/nanotubes | Very strong, light | Sports cars, aircraft |
Wood | Lignin (organic polymer) | Cellulose fibres | Tough, strong | Building material |
Concrete | Cement | Sand + gravel (aggregate) | Hard, rigid | Construction (buildings, skate parks) |
Advantages:
Combines useful properties (e.g. light and strong).
Resistant to corrosion.
Disadvantages:
Expensive to make.
Cannot be easily recycled.
Polymers
Large molecules made from many small molecules called monomers joined together by strong covalent bonds. They are insulators of heat and electricity and can be flexible or rigid depending on structure.
Effect of Monomers and Conditions
The monomers used and the conditions under which polymers are made affect how the polymer chains are arranged and how strongly they are held together. This changes key properties such as strength, flexibility, density, melting point and biodegradability (as seen by plastic recycling symbols below).

Example (LDPE vs HDPE):
Low-Density Poly(ethene) (LDPE) and High-Density Poly(ethene) (HDPE) are both made from the monomer ethene but have vastly different properties due to the conditions in which they are made.
Polymer Type | Reaction Conditions | Structure | Properties | Example Uses |
|---|---|---|---|---|
Low-Density Poly(ethene) (LDPE) | Moderate temperature (~200 °C), high pressure, and a free radical catalyst | Chains are branched, not tightly packed | Flexible, low melting point | Plastic bags, bottles |
High-Density Poly(ethene) (HDPE) | Lower temperature and pressure with a Ziegler–Natta catalyst | Chains are straight and closely packed | Rigid, strong, higher melting point | Water pipes, crates |
The choice of monomer determines the type of bonding, and the reaction conditions control the chain arrangement, both of which directly influence the strength, flexibility, and melting point of the polymer.
Thermosoftening vs Thermosetting
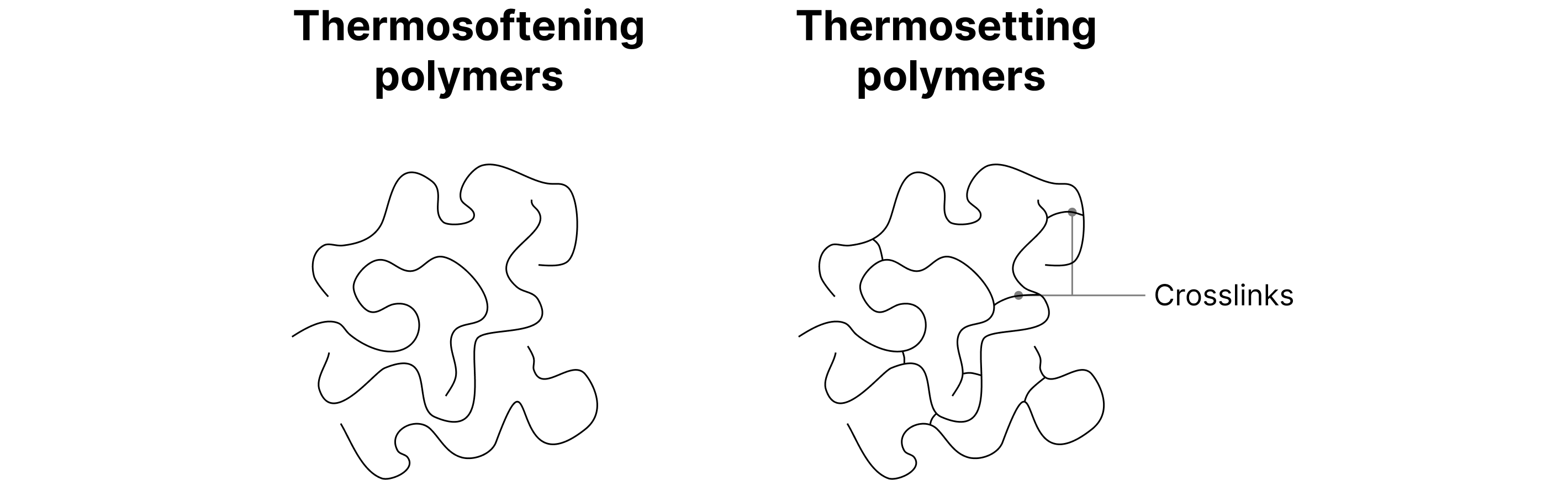
Thermosoftening Polymers
Weak intermolecular forces between chains.
Soften when heated, can be reshaped and cooled.
Example: Poly(ethene), Poly(vinyl chloride).
Thermosetting Polymers
Strong cross-links between chains (covalent/ionic).
Do not melt when heated — instead, they char/burn.
Hard and rigid.
Example: Bakelite, Melamine resin.

Properties:
Malleable (can be shaped).
Ductile (can be drawn into wires).
Good conductors of heat and electricity.
Shiny, strong, and dense.
Examples and Uses:
Copper: Conducts electricity — used in electrical wiring.
Aluminium: Low density, corrosion-resistant — used in aircraft.
Steel (alloy): Strong and durable — used in buildings, cars, tools.
Choosing the Right Material
When choosing materials, consider:
Strength and rigidity
Flexibility
Melting point
Corrosion resistance
Cost and availability
Example 1
Choose from the table the material that would be best suited for making:
1. A kitchen worktop,
2. Window panes,
3. A coffee mug.
Give reasons for each choice.
Material | Cost | Heat Resistance | Transparency | Strength | Porosity (liquid absorption) |
|---|---|---|---|---|---|
A – Glass | Medium | Medium | High | Brittle | Non-porous |
B – Ceramic | Low | High | Opaque | Strong | Non-porous |
C – Plastic (polymer) | Low | Low | Variable | Flexible | Non-porous |
D – Granite (composite) | High | Very High | Opaque | Very strong | Non-porous |
1. D – Granite — The material must withstand heat from hot pans and be strong and non-porous to resist stains.
2. A – Glass — Transparency is essential for windows. It is non-porous and allows light through but strong enough for safety glass.
3. B – Ceramic — Needs high heat resistance for hot drinks, and being non-porous prevents liquid absorption.
Tip: Match the property of each material to the function it needs to perform (e.g., heat resistance for cooking, transparency for viewing).
Example 2
Choose from the table the material that would be best suited for making:
1. Electrical wiring,
2. A bike frame,
3. A rain jacket.
Give reasons for each choice.
Material | Density | Conductivity | Flexibility | Strength | Corrosion Resistance |
|---|---|---|---|---|---|
W – Copper | High | Excellent | Ductile | Strong | Poor |
X – Aluminium alloy | Low | Good | Slightly flexible | Strong | Excellent |
Y – Carbon fibre composite | VVery low | Poor | Rigid | Very strong | Excellent |
Z – Polymer (PVC) | Low | Insulator | Flexible | Weak | Good |
1. W – Copper — Excellent electrical conductivity and ductility make it ideal for wiring.
2. Y – Carbon fibre composite — Very strong yet lightweight; ideal for bike frames where strength-to-weight ratio is crucial.
3. Z – Polymer (PVC) — Flexible, waterproof, and lightweight, making it suitable for clothing like rain jackets.
Exam Tip: Always consider both functional (e.g., conductivity, strength) and practical (e.g., cost, weight, flexibility) properties when choosing materials.
Practice Questions
a) State what ceramics are made from.
a) Ceramics are non-metallic materials made by heating substances like clay or sand at high temperatures.
b) What are the three ingredients used to make soda-lime glass?
b) Sand, sodium carbonate, and limestone.
c) What is meant by a composite material?
c) A composite combines two materials — a matrix (binder) and reinforcement — to produce improved properties.
d) Give one example of a thermosoftening and one of a thermosetting polymer.
d) Thermosoftening: poly(ethene)/poly(vinyl chloride). Thermosetting: bakelite/melamine resin.
Apply
e) Why is clay suitable for making bricks?
e) Clay can be moulded when wet and hardens when heated, making strong, heat-resistant bricks.
f) Explain why borosilicate glass is used for laboratory equipment.
f) It has a high melting point, so it can withstand heating in experiments without breaking.
g) Suggest why carbon fibre composites are used in aircraft instead of metals.
g) Carbon fibre composites are strong but lightweight, reducing fuel use and increasing performance.
h) A polymer has high rigidity and does not soften when heated. What type of polymer is it, and why?
h) It is a thermosetting polymer, as strong cross-links between chains stop it from melting.
Challenge
i) Choose from the table the material that would be best suited for making:
1. The car chassis (frame),
2. The windscreen,
3. The engine block,
4. The interior panels.
Give reasons for each choice.
Material | Density | Strength | Heat Resistance | Corrosion Resistance | Other Properties |
|---|---|---|---|---|---|
A – Aluminium alloy | Low | Strong | High | Excellent | Lightweight, malleable |
B – Carbon fibre composite | Very low | Very strong | Medium | Excellent | Rigid, expensive |
C – Steel | High | Very strong | High | Moderate | Cheap, tough |
D – Glass (laminated) | Medium | Brittle | Medium | Excellent | Transparent, impact-resistant |
E – Polymer (ABS plastic) | Very low | Weak | Low | Excellent | Lightweight, mouldable |
i) The car chassis - B – Carbon fibre composite — Extremely strong yet lightweight, perfect for improving acceleration and fuel efficiency in high-performance cars.
The windscreen - D – Laminated glass — Transparent and impact-resistant, ideal for windscreens as it doesn’t shatter easily on impact.
The engine block - C – Steel — Very strong and heat-resistant, making it suitable for engine blocks that must withstand high temperatures and pressures.
The interior panels - E – Polymer (ABS plastic) — Light, easily moulded, and corrosion-resistant, ideal for interior panels and trim where flexibility and weight reduction are important.