The Haber process
Emmanuel Opoku
Teacher

Contents
The Haber Process
The Haber Process is an industrial method used to manufacture ammonia (NH₃) from nitrogen (N₂) and hydrogen (H₂).
Ammonia is a vital chemical used in:
Fertilisers (to feed crops and support global food production)
Cleaning products
Explosives and pharmaceuticals
Word and Symbol Equation
Nitrogen + Hydrogen ⇌ Ammonia
Conditions:
Temperature: 450°C
Pressure: 200 atmospheres
Catalyst: Iron
The reversible arrow (⇌) shows that this reaction can go both forward and backward - ammonia can decompose back into nitrogen and hydrogen.
Sources of Raw Materials
Substance | Source |
|---|---|
Nitrogen (N₂) | Extracted from the air (≈78% nitrogen). |
Hydrogen (H₂) | Obtained from natural gas (methane) via reaction with steam. |
How the Haber Process Works
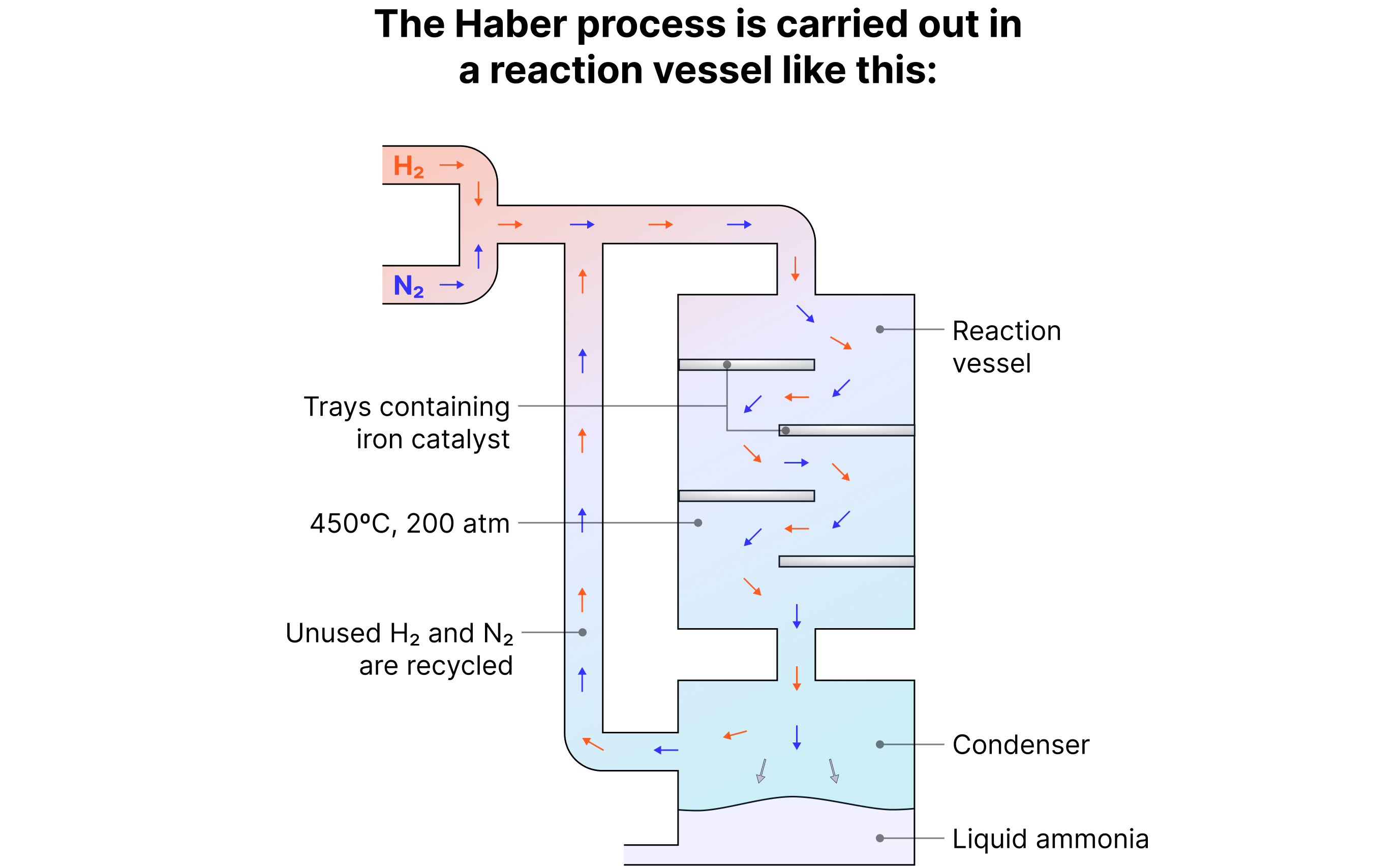
1. Nitrogen and hydrogen gases are purified and mixed in a 1:3 ratio.
2. The gases pass over iron catalyst trays at 450 °C and 200 atm.
3. A reversible reaction occurs, forming ammonia gas.
4. The mixture is cooled - ammonia liquefies and is removed.
5. Unreacted nitrogen and hydrogen are recycled to reduce waste and costs.
Environmental and Economic Considerations
Recycling unreacted gases reduces waste and saves money.
Using a compromise between rate and yield reduces energy use, limiting environmental impact.
Ammonia is crucial for food production, supporting a growing global population.
Explaining Reaction Conditions (HT Only)
The conditions used in the Haber Process are 450 °C, 200 atm, and an iron catalyst are chosen as a compromise between yield, rate of reaction, and cost.
Temperature – 450 °C (Compromise Temperature)
The forward reaction is exothermic, meaning it releases heat:
Increasing temperature shifts the equilibrium backwards (towards nitrogen and hydrogen), reducing yield of ammonia.
However, lower temperatures make the reaction too slow because the gas molecules have less kinetic energy and collide less frequently with enough energy to react.
Therefore, 450 °C is a compromise:
It provides a reasonable rate of reaction.
It still gives an acceptable yield of ammonia (about 15–20%).
It’s better to produce some ammonia quickly than a higher yield very slowly.
Pressure – 200 atm (Compromise Pressure)
The forward reaction forms fewer gas molecules (4 → 2), so increasing pressure shifts equilibrium towards the right, increasing yield of ammonia (Le Chatelier’s Principle).
Very high pressures (e.g., > 1000 atm) would produce a higher yield, but:
The equipment required is extremely expensive.
It increases energy use and safety risks.
Therefore, a pressure of 200 atm gives a good yield while keeping costs and risks manageable.
Doubling the pressure doesn’t double the yield - but it doubles the cost!
Iron Catalyst
The iron catalyst speeds up the rate of reaction by lowering activation energy, helping equilibrium to be reached faster.
It does not change the position of equilibrium or the maximum yield - it only helps the reaction reach equilibrium faster.
Without a catalyst, the temperature would need to be raised to speed up the reaction, which would decrease yield and increase energy costs.
Overall Compromise
Changing Factor | Effect on Yield (due to Le Chateleir’s principle) | Effect on Rate | Economic Effect/Considerations |
|---|---|---|---|
↑ Temperature | ↓ (less ammonia) | ↑ (faster) | More expensive |
↓ Temperature | ↑ (more ammonia) | ↓ (slower) | Cost to lower temperatuer but slower production |
↑ Pressure | ↑ (more ammonia) | ↑ (faster) | Higher costs for sturdier containers and risk of explosion from high pressures |
Use of Catalyst | No change | ↑ (faster) | Lowers Energy cost |
Summary
The Haber Process runs at 450 °C, 200 atm, with an iron catalyst to achieve a balance between a good yield, reasonable speed, and safe, cost-effective operation.
This is a key example of industrial compromise - balancing chemistry with engineering, economics, and safety.
Key idea: Increasing pressure speeds up reactions by increasing collision frequency, but industrial processes must balance this benefit with the cost and safety implications of maintaining very high pressures (same applies to increasing the temperature).
Interpreting Rate from Graphs (HT Only)
Graphs can be used to show how changing conditions (pressure and temperature) affects the rate of reaction in industrial processes such as the Haber Process.
By analysing these graphs, scientists can determine the best operating conditions that give a reasonable yield at a fast rate.
For example, increasing the pressure increases the rate of reaction. This is because gas particles are pushed closer together, leading to more frequent collisions between nitrogen and hydrogen molecules.
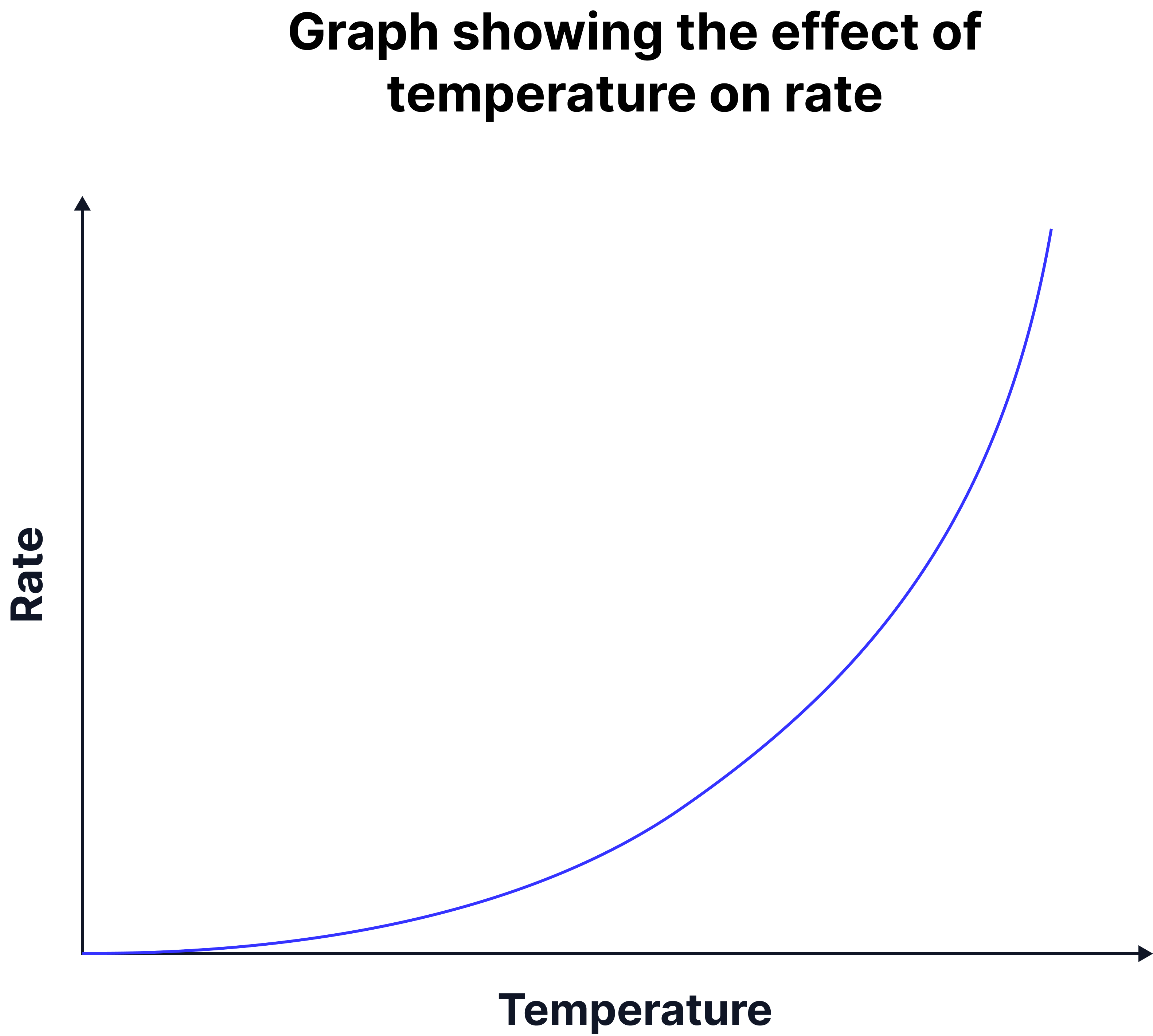
As the graph shows, as pressure increases, the rate of reaction rises sharply at first but eventually levels off.
Beyond a certain point, further increases in pressure have a smaller effect on rate, while greatly increasing costs and safety risks.
Key idea:
Increasing pressure speeds up reactions by increasing collision frequency, but industrial processes must balance this benefit with the cost and safety implications of maintaining very high pressures (same applies to increasing the temperature).
Check Your Understanding
Recall
Write the word equation for the Haber Process.
Answer:
Nitrogen + Hydrogen ⇌ Ammonia
Name the raw materials used and state their sources.
Answer:
Nitrogen from air; Hydrogen from natural gas (methane)
State the purpose of the iron catalyst.
Answer:
The iron catalyst speeds up the rate of reaction but does not affect yield
Explain what the reversible arrow (⇌) means.
Answer:
The reaction is reversible — it can go both forwards (forming ammonia) and backwards (breaking ammonia down)
Apply Your Knowledge (HT Only)
Explain why a high pressure increases the yield of ammonia.
Answer:
The forward reaction produces fewer gas molecules (4 → 2), so high pressure shifts equilibrium to the right producing ammonia (Le Chatelier’s Principle)
Explain why the temperature used in the Haber Process is described as a “compromise”.
Answer:
A lower temperature gives a higher yield because the forward reaction is exothermic. Lowering the temperature shifts the equilibrium to the right, increasing the amount of ammonia. However, a lower temperature but makes the rate of reaction too slow. A higher temperature increases the rate but reduces yield — therefore, 450 °C is a compromise between rate and yield
What happens to unreacted nitrogen and hydrogen?
Answer:
The unreacted nitrogen and hydrogen are recycled back into the reactor to reduce waste and improve efficiency
Challenge Question (HT Only)
The table shows how different conditions affect the rate and yield of ammonia in the Haber Process.
|
Condition |
Rate of Reaction |
Yield of Ammonia (%) |
Cost of Operation |
|---|---|---|---|
|
350 °C, 150 atm |
Slow |
30 |
Low |
|
450 °C, 200 atm |
Moderate |
20 |
Moderate |
|
550 °C, 300 atm |
Fast |
10 |
Very High |
Use the data and your knowledge to evaluate which set of conditions would be most suitable for industrial use.
Answer:
- The best industrial conditions are 450 °C and 200 atm.
- Although the yield (20%) is lower than at 350 °C, the reaction rate is faster, allowing continuous production.
- The pressure is high enough to give a good yield but not so high that costs or safety risks become too great.
- Using an iron catalyst speeds up the reaction without affecting equilibrium, making this the most efficient and economical compromise between rate, yield, and cost.
Important Consideration
Industrial processes aim for the best overall efficiency, not just the highest yield.
The chosen conditions balance rate of reaction, safety, and economic cost for maximum productivity.