Amount of substance in relation to volumes of gases (Triple only)
Lajoy Tucker
Teacher

Contents
The Mole-Volume Relationships for Gasses
Equal numbers of moles gasses occupy the same volume when measured under the same conditions of temperature and pressure.
This means that 1 mole of any gas occupies the same volume as 1 mole of any other gas (as long as temperature and pressure are constant)
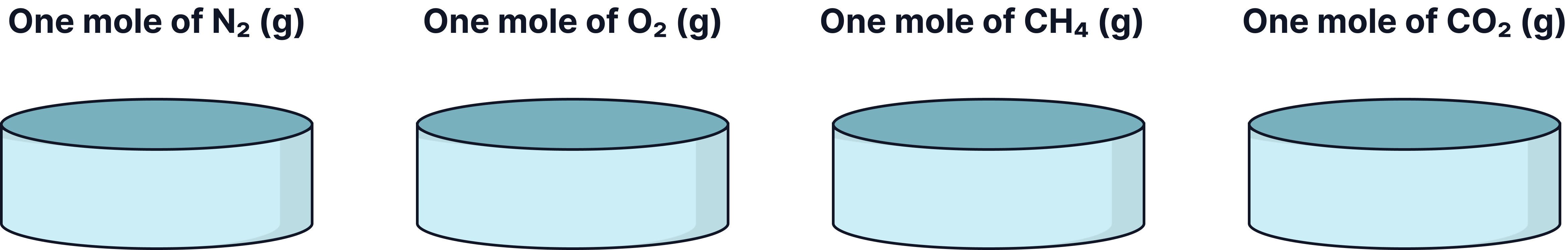
At room temperature and pressure (RTP), define as 20°C and 1 atm,
of gas occupies
()
This relationship is useful for comparing gases and linking mass, moles, and volume in chemical reactions.
Volume of gas at RTP
Moles of gas
Worked Example 1:
Calculate the volume of 4.0g of oxygen gas () at room temperature and pressure .
Step 1: Fine moles
Mr of = 32
Step 2: Convert moles to volume
Worked Example 2:
What mass of ammonia is contained in of the gas at RTP?
Using a Balanced Equation to Find Gas Volumes
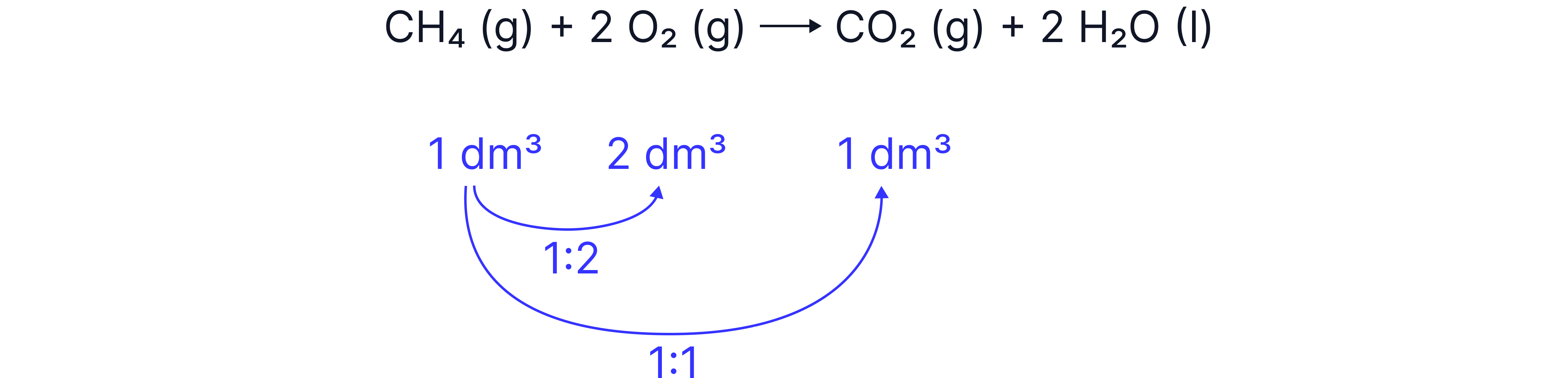
As “equal volumes of all gases, at the same temperature and pressure, have the same number of molecules”,
if both species in the calculation are gases, and the temperature and pressure are the same, we can use the molar ratios in the balanced symbol equation to relate volumes directly INSTEAD of having to calculate moles
At a given temperature and pressure, what volume of oxygen is required to burn 1 of methane and what volume or carbon dioxide is produced?
Check for Understanding
Recall
a) State the volume of 1 mole of any gas at room temperature and pressure (RTP).
Answer:
a) 24 dm³.
b) State the relationship between equal moles of gases and their volumes at the same temperature and pressure.
Answer:
b) Equal moles of gases have equal volumes at the same temperature and pressure.
c) When using a balanced equation, what do gas volumes follow the same ratio as?
Answer:
c) Mole ratios in the balanced equation.
Application
d) Calculate the volume of gas produced at RTP if 2 moles of a gas are formed.
Answer:
d) Volume = moles × 24
Volume = 2 × 24 = 48 dm³.
e) In a reaction, the balanced equation shows 1 mole of gas A produces 2 moles of gas B.
If 24 dm³ of gas A reacts, what volume of gas B will be produced at the same temperature and pressure?
Answer:
e) The ratio of A : B is 1 : 2.
If 24 dm³ of A reacts, the volume of B is 48 dm³.
f) A reaction produces 0.5 moles of a gas at RTP.
Calculate the total volume of gas produced.
Answer:
f) Volume = moles × 24
Volume = 0.5 × 24 = 12 dm³.
Challenge
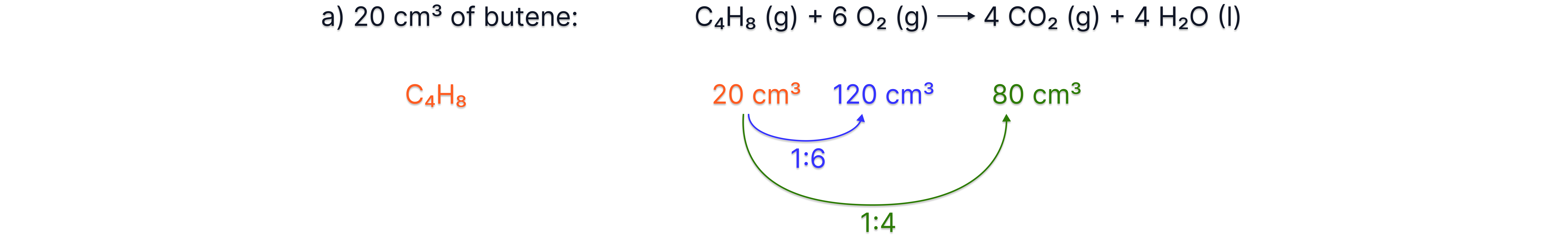
g) In the following reaction, what volume of O₂ is required and what volume of CO₂ is produced from 20m³ od butene?
C₄H₈(g) + 6 O₂(g) → 4 CO₂(g) + 4 H₂O(l)
Answer:
Summary
1 mol of any gas = 24 dm³ at room temperature and pressure (RTP).
Equal moles → equal volumes under identical conditions.
Volume (dm³) = Moles × 24.
Gas volumes follow the same ratios as the mole ratios in balanced equations.