Atmospheric Pollutants
Dr. Davinder Bhachu
Teacher

Air Pollution
What Is Released When Fuels Burn?
When fossil fuels such as petrol, diesel, coal and natural gas burn, they do more than produce energy - they release a range of gases and particles that affect human health and the environment.
Hydrocarbon fuels are made only of carbon (C) and hydrogen (H). When they react with oxygen during combustion, a variety of products can form depending on how much oxygen is available.
Products of Combustion
1. Complete Combustion (Enough Oxygen)
When plenty of oxygen is present, hydrocarbons burn “cleanly.”
Main products:
- Carbon dioxide (CO₂)
- Water vapour (H₂O)
These products form because all carbon and hydrogen atoms are fully oxidised.
Example:
Burning methane (a hydrocarbon) in excess oxygen gives:
methane + oxygen → carbon dioxide + water
2. Incomplete Combustion (Limited Oxygen)
If oxygen supply is restricted, combustion is incomplete and harmful substances are produced.
Possible products:
- Carbon monoxide (CO) – a poisonous gas
- Soot / carbon particles – tiny solid carbon pieces
- Unburned hydrocarbons – fuel molecules that didn’t react fully
- Carbon dioxide and water – still formed, but in lower amounts
Why does this happen?
Not all carbon gets fully oxidised, so some escapes as soot or becomes carbon monoxide rather than carbon dioxide.
3. Other Pollutants from Fossil Fuels
Sulfur dioxide (SO₂)
Produced if the fuel contains sulfur impurities (common in coal and some oils).
Nitrogen oxides (NOₓ)
Formed when nitrogen and oxygen in air react at very high temperatures, such as inside car engines or power-station furnaces.
NOₓ includes:
- NO (nitrogen monoxide)
- NO₂ (nitrogen dioxide)
Problems Caused By Combustion Products
Carbon Particulates (Soot)
Effects on humans
- Can become lodged in the lungs
- Linked to breathing difficulties and long-term respiratory disease
Effects on the environment
- Reflect sunlight away from Earth
- Contribute to reduced global sunlight levels, a process often referred to as global dimming
Carbon Monoxide (CO)
Carbon monoxide is especially dangerous because:
- It attaches to haemoglobin more strongly than oxygen does
- This means the blood can’t carry enough oxygen around the body
- This may lead to fainting, organ damage, coma, or death
- It is colourless and odourless, so it is extremely hard to detect without specialised equipment
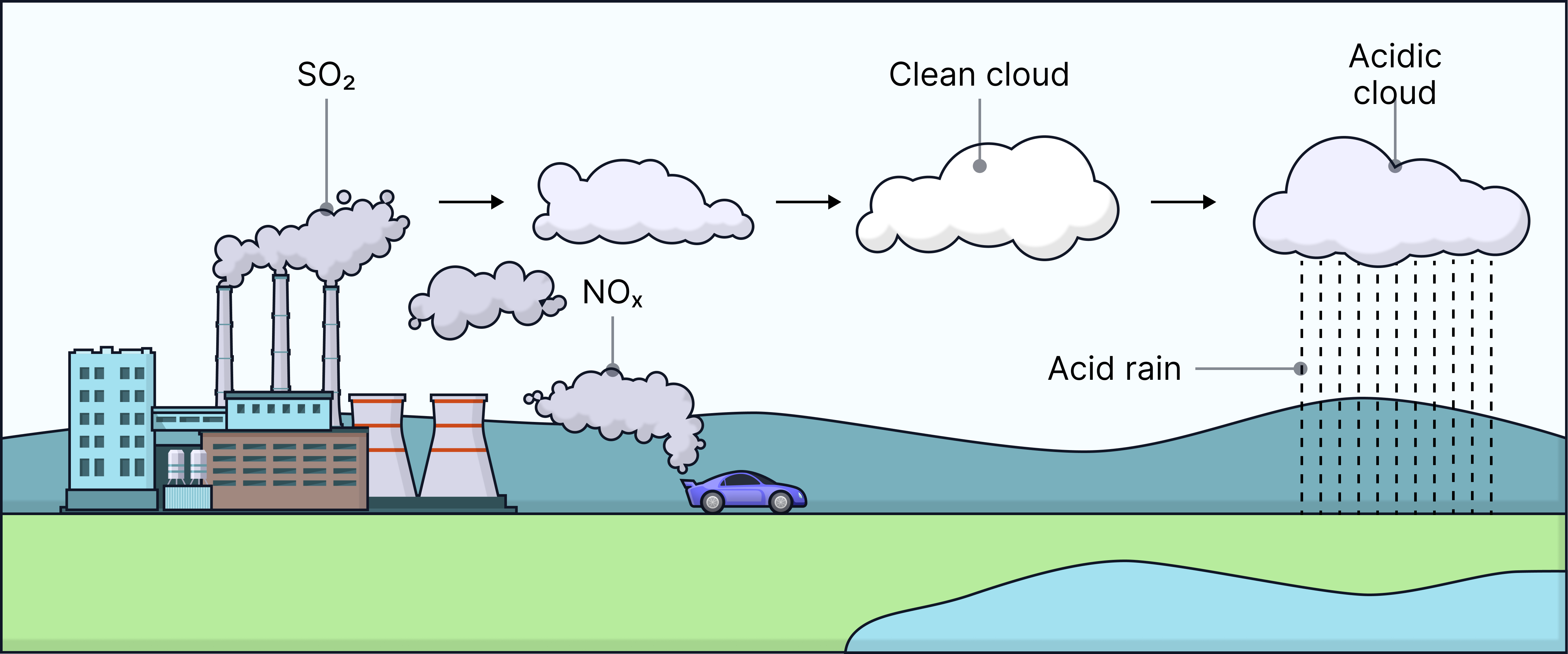
Acid Rain
Acid rain forms when:
- Sulfur dioxide (SO₂) reacts with water in clouds → sulfuric acid
- Nitrogen oxides (NOₓ) react with water in clouds → nitric acid
These acids fall to the ground as acid rain.
Environmental consequences
- Makes lakes and streams acidic → kills fish and aquatic organisms
- Damages trees by weakening leaf surfaces and removing nutrients from soil
- Erodes buildings made from limestone or marble
- Can degrade outdoor sculptures
Health Effects of SO₂ and NOₓ
Even aside from acid rain, breathing in sulfur dioxide or nitrogen oxides can:
- Trigger asthma attacks
- Irritate airways
- Cause long-term respiratory issues
These gases dissolve in the moisture lining the respiratory system, forming mild acids that irritate tissues.
Check Your Understanding
Recall
a) What two substances are formed during the complete combustion of a hydrocarbon?
Answer:
Carbon dioxide; water
b) Name two products of incomplete combustion.
Answer:
Carbon monoxide; soot/carbon particles; unburned hydrocarbons
c) Which gas is produced when fuels containing sulfur impurities burn?
Answer:
Sulfur dioxide
Application
d) Explain why carbon monoxide is dangerous to humans.
Answer:
Carbon monoxide binds to haemoglobin more strongly than oxygen; reduces oxygen transport in blood; can cause unconsciousness or death; colourless and odourless
e) Describe how nitrogen oxides are formed in car engines.
Answer:
Very high temperatures in engines; nitrogen and oxygen from air react; form NO and NO₂
f) Explain how sulfur dioxide leads to acid rain and describe one environmental impact.
Answer:
Sulfur dioxide reacts with water in clouds; forms sulfuric acid; acid rain falls; damages plants / acidifies lakes / kills aquatic life / erodes stone
Challenge (HT Only)
g) Explain how sulfur dioxide and nitrogen oxides form acids in the atmosphere and analyse why this makes limestone buildings particularly vulnerable.
Answer:
Sulfur dioxide forms sulfuric acid; nitrogen oxides form nitric acid; acids dissolve in rainwater; limestone contains calcium carbonate; acids react with carbonates; forms soluble salts/carbon dioxide; surface erodes over time
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!