Atom Economy (Triple Only)
Lajoy Tucker
Teacher

Introduction & Definitions
Percentage Atom Economy: A measure of the proportion of atoms in the reactants that are converted into the desired product(s).
Importance:
Used to assess the efficiency and sustainability of a chemical process.
Higher atom economy = less waste, more sustainable and cost-effective.
Note: the total of all reactants is the same as that of the products due to the law of conversation of mass.
Basic Priciples
Atom economy is based purely on the balanced chemical equation (not on experimental yield).
It does not depend on reaction conditions or experimental success.
It focuses on how many of the atoms in the reactants end up in the desired product.
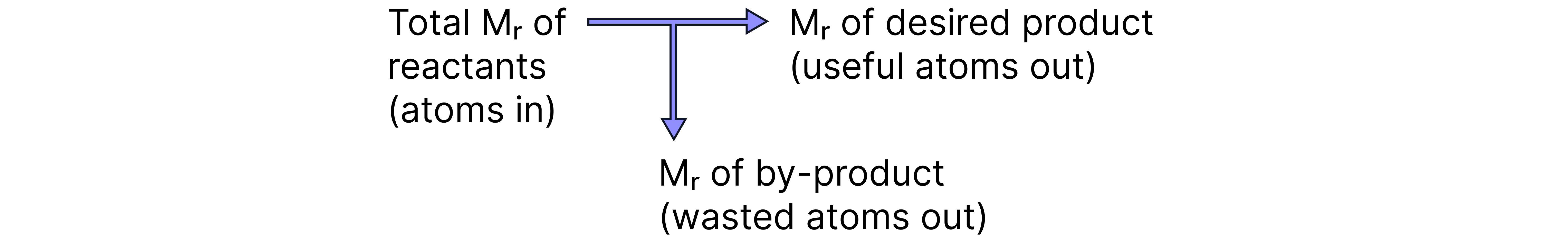
Explanations
Why Atom Economy Matters
Economic: Less waste means lower disposal costs and higher profits.
Environmental: Less pollution and waste production.
Ethical: Green chemistry principles promote sustainability and minimal resource use.
Choosing Reactions
Reactions with fewer by-products and only one main product tend to have higher atom economy.
Addition reactions generally have 100% atom economy, since all atoms become part of the product.
Substitution and elimination reactions usually have lower atom economies due to by-products.
Worked Examples
Example 1 – High Atom Economy Reaction
Reaction:
Mr Values:
Addition reaction No waste, high efficiency.
Example 2 – Lower Atom Economy Reaction
Reaction:
Desired product:
Total of reactants = 106 + 2(36.5) = 179.0
Less efficient due to waste products
Note that the values must be multiplied by the coefficients to account for the total atoms in and out.
Check for Understanding
Recall
a) What does the law of conservation of mass state about the total relative formula mass (Mr) of reactants and products?
Answer:
a) The total Mr of the reactants is equal to the total Mr of the products / mass is conserved.
b) In atom economy, what is meant by the desired product?
Answer:
b) The useful product formed in the reaction / the product the reaction is intended to make.
c) What is a by-product in a chemical reaction?
Answer:
c) A product formed in a reaction that is not the desired/useful product / a waste product.
Application
d) Calculate the atom economy for the following reaction
Answer:
e) A chemical reaction forms a useful product and a waste by-product. Explain how the atoms from the reactants are distributed between these two products.
Answer:
d) Atoms from the reactants are rearranged to form both the desired product and the by-product.
f) A company uses a process with a low atom economy. Suggest one way the company could reduce the environmental impact of the waste products.
Answer:
f) Sell the waste products.
Use waste products for another process.
Recycle or reuse the by-products.
g) Explain why finding alternative uses for by-products can reduce the economic impact of a chemical process.
Answer:
g) By-products can be sold or reused.
This reduces waste disposal costs.
This improves the economic efficiency of the process.