Atoms, elements and compounds
Lajoy Tucker
Teacher

Contents
Atoms – The Building Blocks of Matter
All substances are made of atoms.
An atom is the smallest part of an element that can exist on its own.
Atoms have a nucleus containing protons and neutrons, with electrons moving in shells around it.
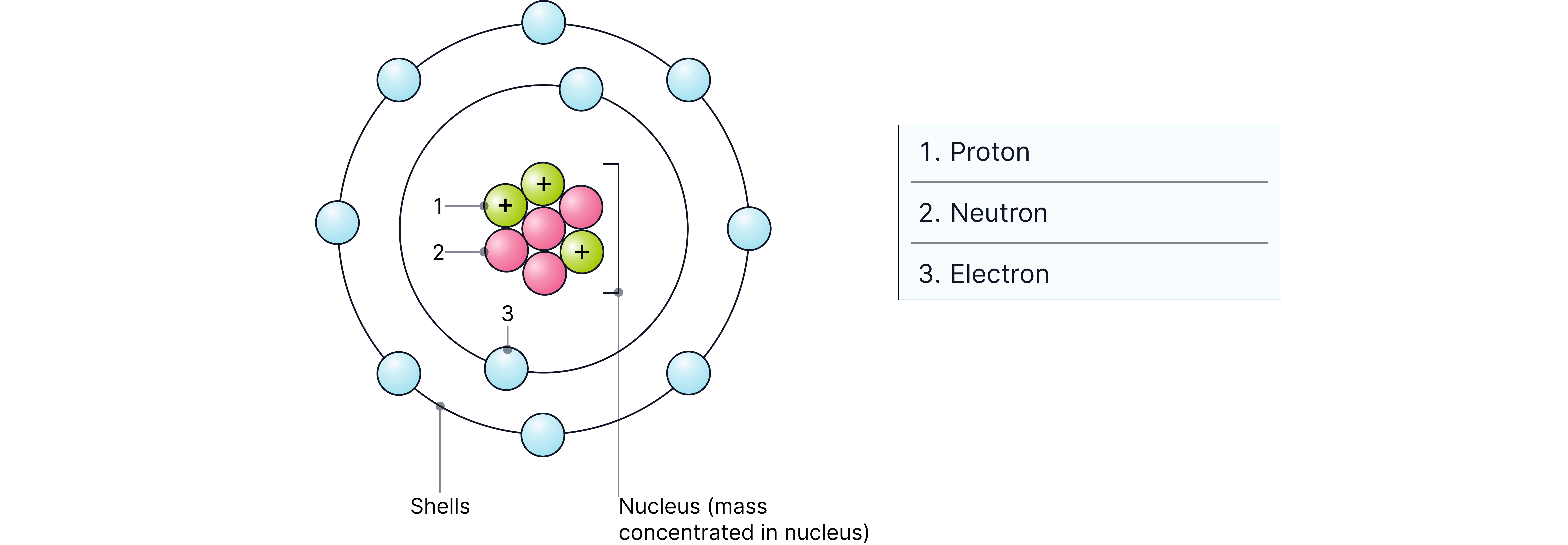
Atoms are extremely small, typically about 1 × 10⁻¹⁰ m in diameter.
Elements
An element is a pure substance made up of only one type of atom.
Each element is represented by a chemical symbol, for example:
O – oxygen
Na – sodium
Cl – chlorine
There are about 100 known elements, all arranged in the periodic table.
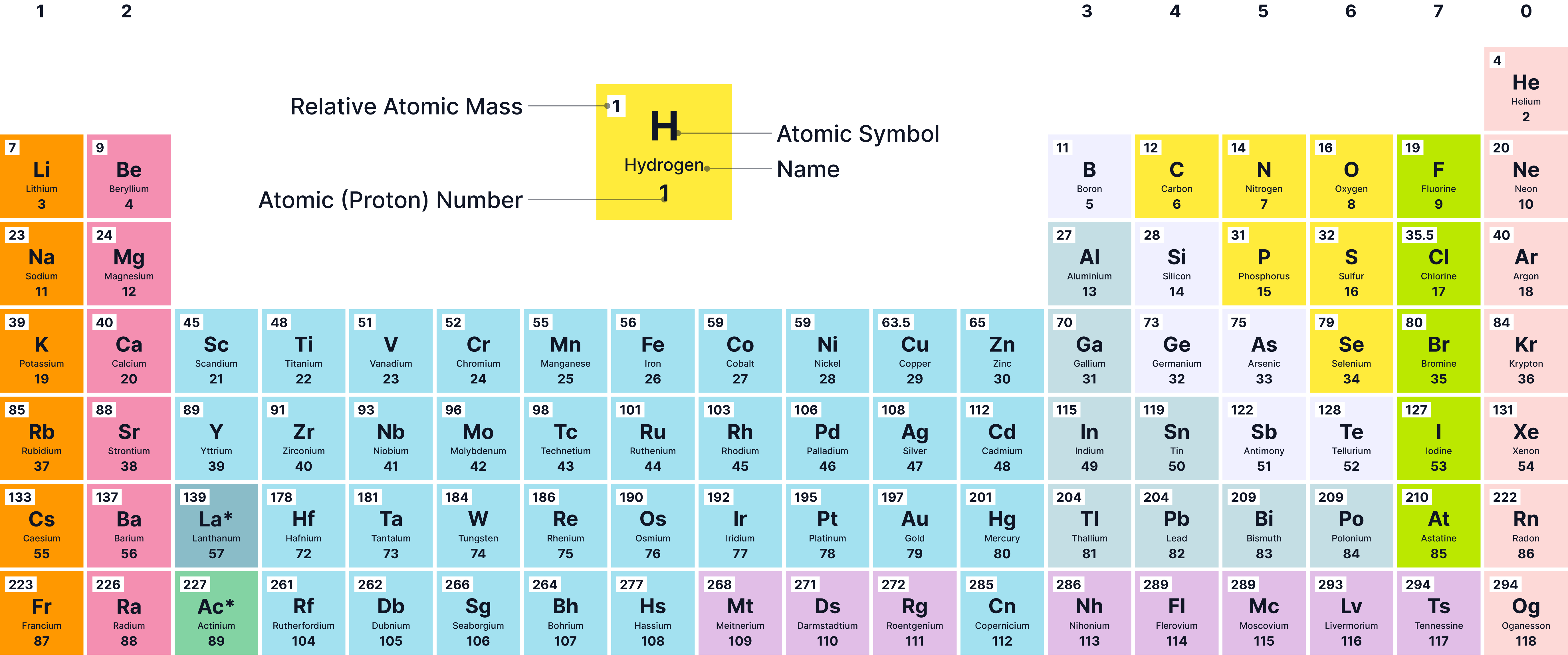
Elements cannot be broken down into simpler substances by chemical means.
Attention! All element symbols must start with a capital letter. If there is more than one letter, the second letter is lowercase
e.g. magnesium is Mg, NOT mg or MG
Diatomic Elements
Some elements exist naturally as molecules made up of two atoms joined together.
These are called diatomic elements.
Each molecule contains two atoms of the same element bonded together.
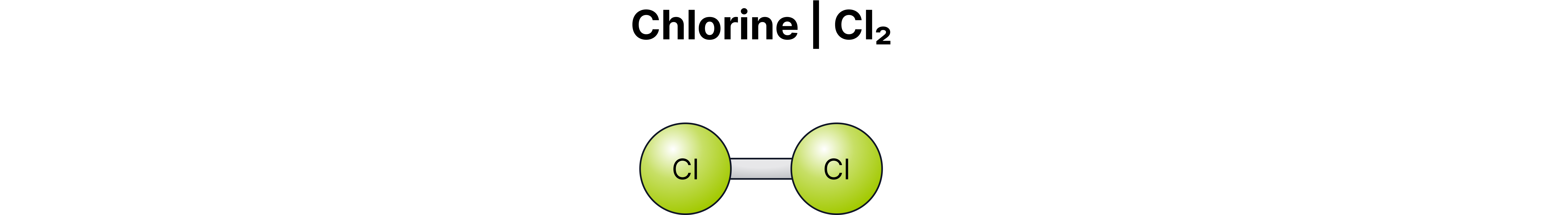
Diatomic Element | Symbol / Formula |
Hydrogen | H₂ |
Nitrogen | N₂ |
Oxygen | O₂ |
Fluorine | F₂ |
Chlorine | Cl₂ |
Bromine | Br₂ |
Iodine | I₂ |
Compounds
A compound forms when two or more elements are chemically combined in fixed proportions.
The compound’s properties are different from the elements it contains.
Compounds are represented by chemical formulae that show the symbols and ratios of atoms present.
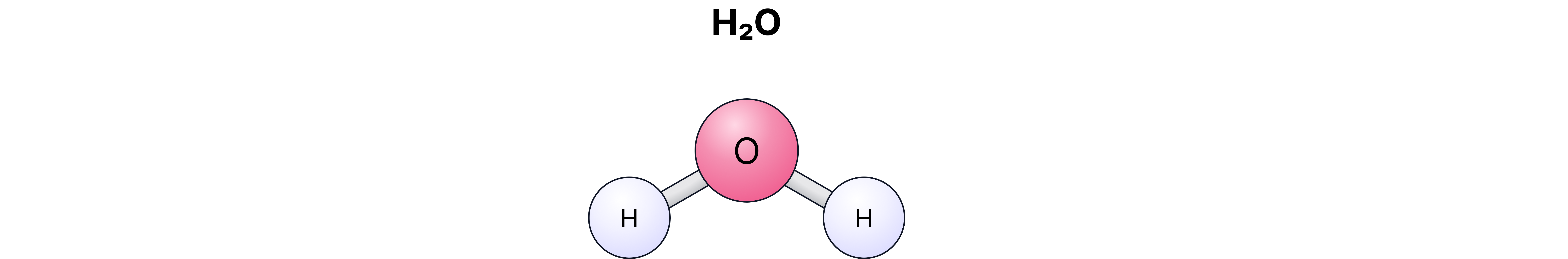
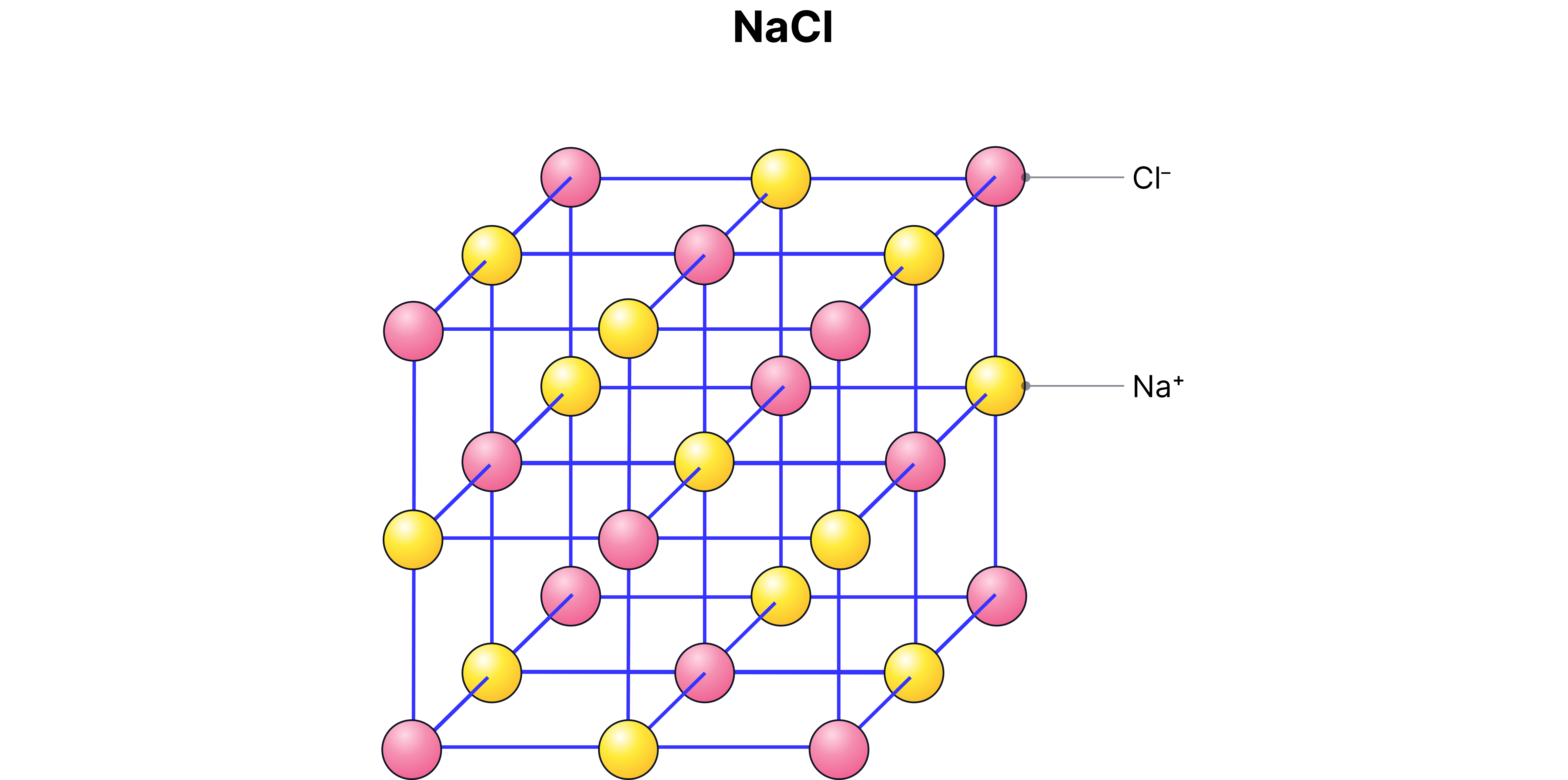
Examples:
– water (2 hydrogen atoms, 1 oxygen atom)
– carbon dioxide (1 carbon atom, 2 oxygen atoms)
– sodium chloride (1 sodium atom, 1 chlorine atom)
Compounds can only be separated into elements by chemical reactions, not by physical methods.
Compounds are typically named based on the elements they contain:
Type of Elements | Naming Pattern | Example |
Metal + Non-metal | Name ends in -ide | Sodium + chlorine → sodium chloride |
Metal + Non-metal + Oxygen | Name ends in -ate or -ite | Copper + sulfur + oxygen → copper sulfate |
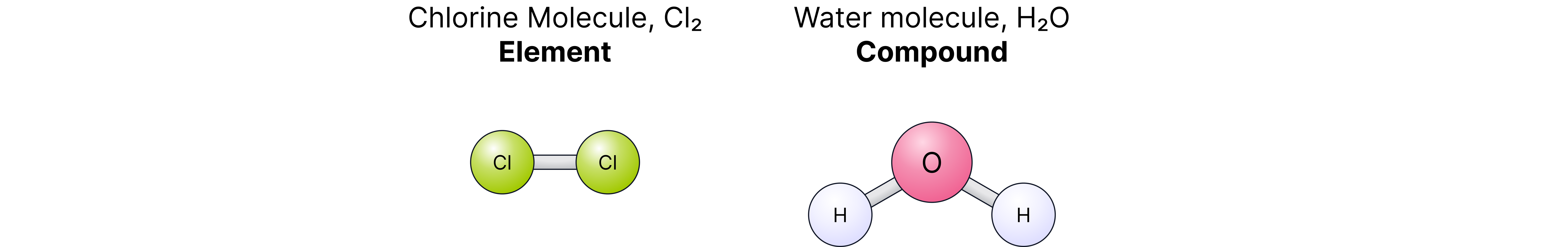
Molecules
A molecule includes a few atoms covalently bonded together.
Molecules can contain one type of atom and be an element OR contain more than one type of atom and be a compound.
Check for Understanding
Recall
a) What is an atom?
Answer:
a) An atom is the smallest part of an element that can exist.
b) What is an element?
Answer:
b) An element is a pure substance made of only one type of atom.
c) What is a compound?
Answer:
c) A compound is a substance formed when two or more different elements are chemically combined in fixed proportions.
Application
d) Explain why O₂ is considered an element.
Answer:
d) O₂ is considered an element because it contains only one type of atom, which is oxygen.
e) Which of the following is the correct chemical symbol for iron?
Fe
FE
fe
fE
Answer:
e) Fe
Challenge (HT)
f) A substance has the formula CO₂. Explain why this substance is a compound rather than an element.
Answer:
f) CO₂ is a compound because it contains more than one type of element. It contains carbon and oxygen atoms that are chemically combined.
g) Explain why a substance can be called a molecule and an element at the same time.
Answer:
g) A substance can be called both a molecule and an element because:
A molecule is two or more atoms chemically bonded together.
An element is a substance made of only one type of atom.
So, if a molecule is made of two or more atoms of the same element, it fits both definitions.