Mixtures
Lajoy Tucker
Teacher

Contents
Introduction
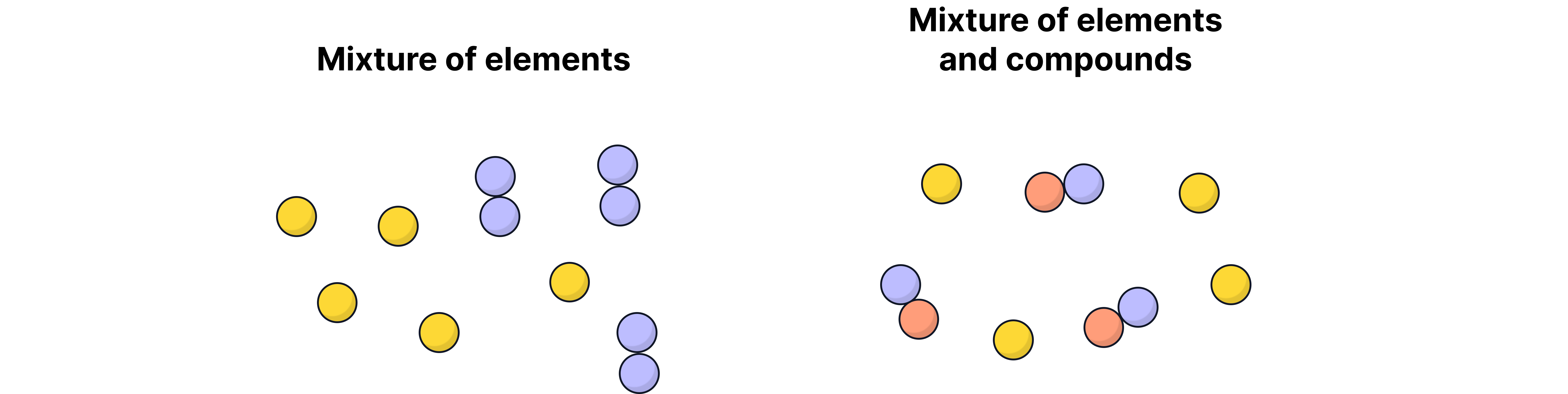
A mixture consists of two or more elements or compounds that are not chemically combined together.
The chemical properties of each substance in a mixture remain unchanged because no chemical bonds are formed or broken between them.
Mixtures are common in everyday life, e.g. air (a mixture of gases), sea water (water + salts), and alloys like brass (copper + zinc).
Key Features of Mixtures
Feature | Description |
Chemical bonding | No new chemical bonds are formed between substances. |
Properties | Each component keeps its own chemical properties. |
Separation | Can be separated by physical methods (no chemical reactions). |
Separating Mixtures – Physical Methods
Process | Used to Separate | Principle / How It Works | Example |
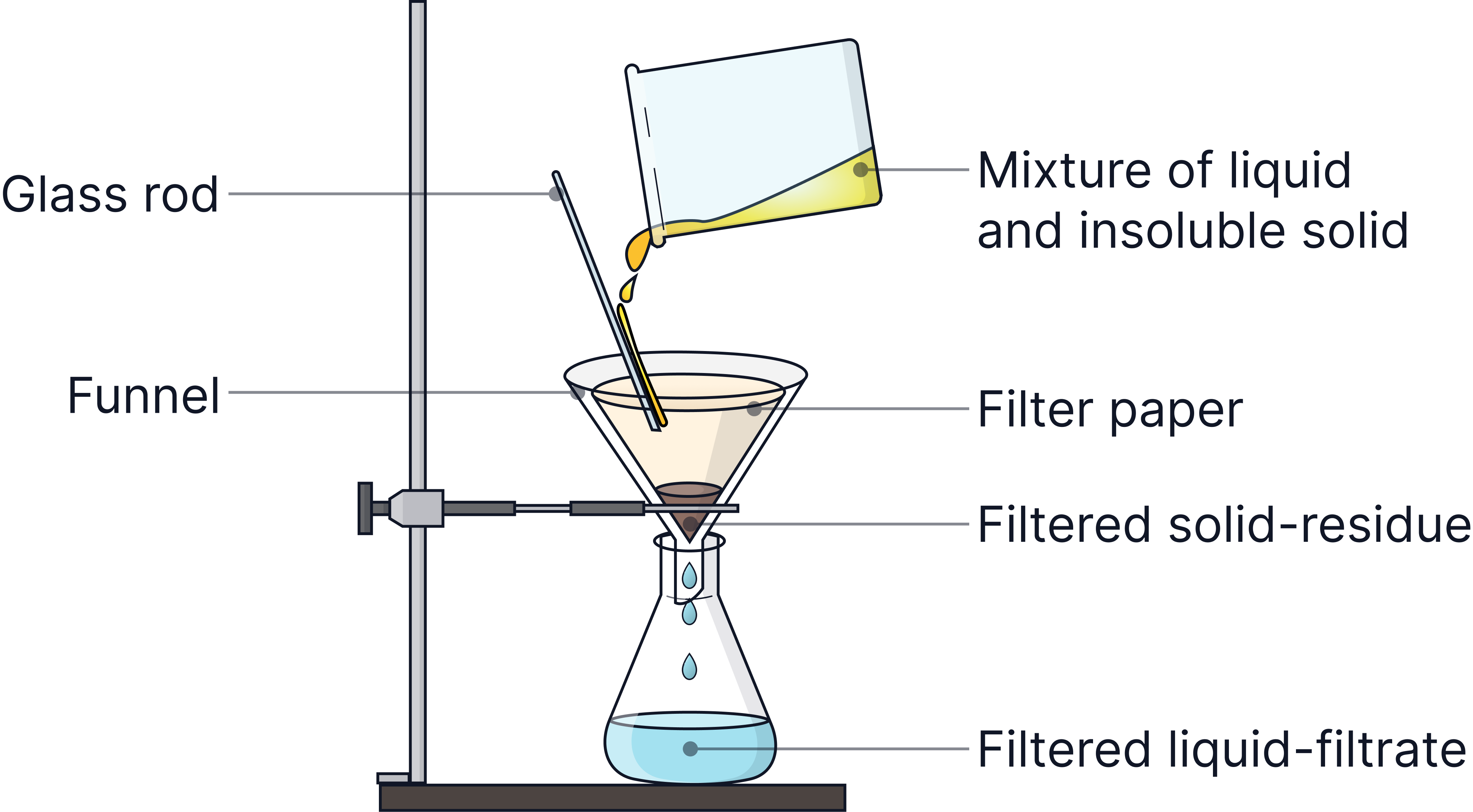
Filtration | Insoluble solid from a liquid | The solid cannot pass through the filter paper | Sand from water |
Crystallisation | Soluble solid from a solution | The solvent is evaporated, leaving crystals of solute.
| Salt from salt solution |
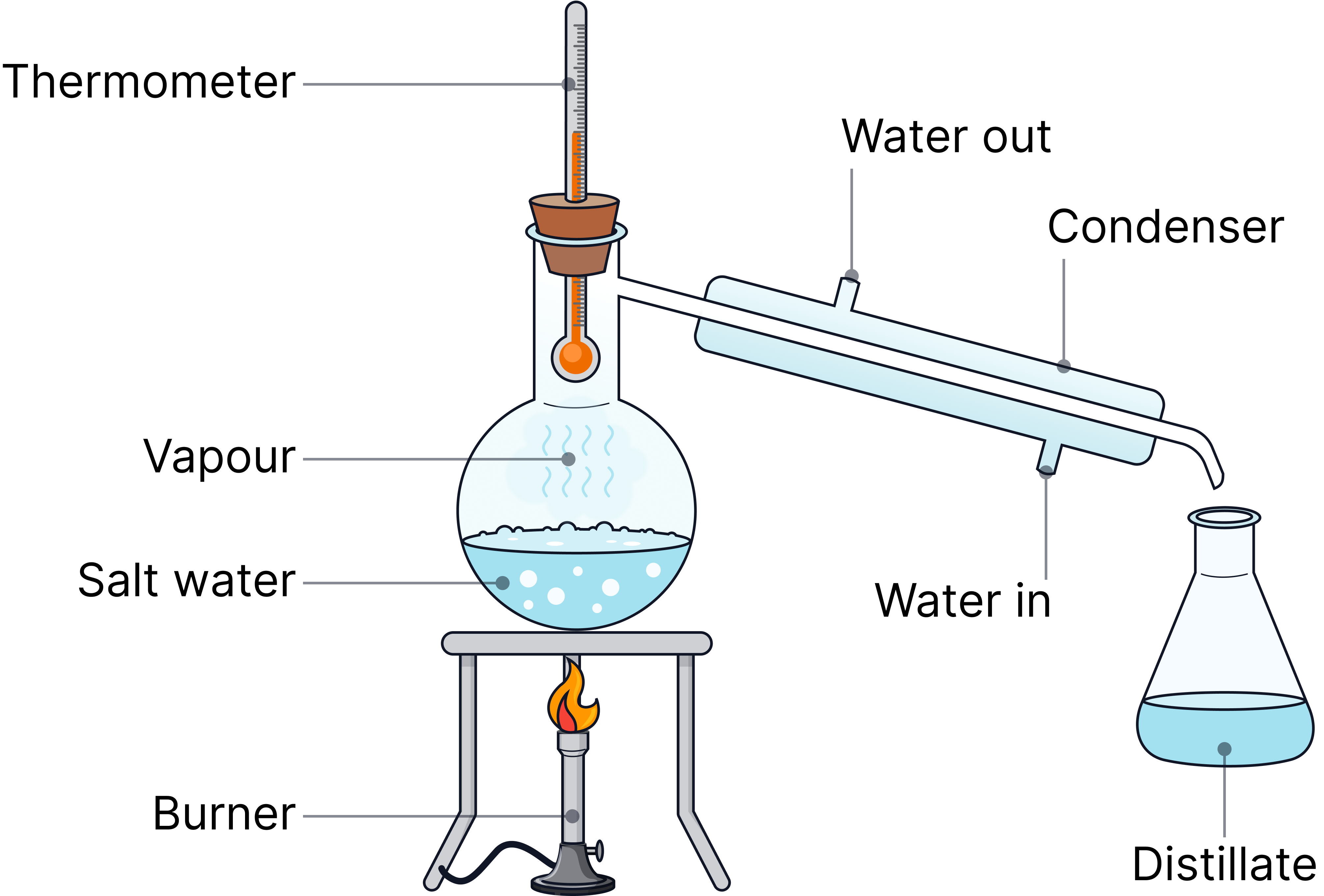
Simple distillation | Solvent from a solution | The solvent boils, evaporates, then condenses into a separate container.
| Water from salt water |
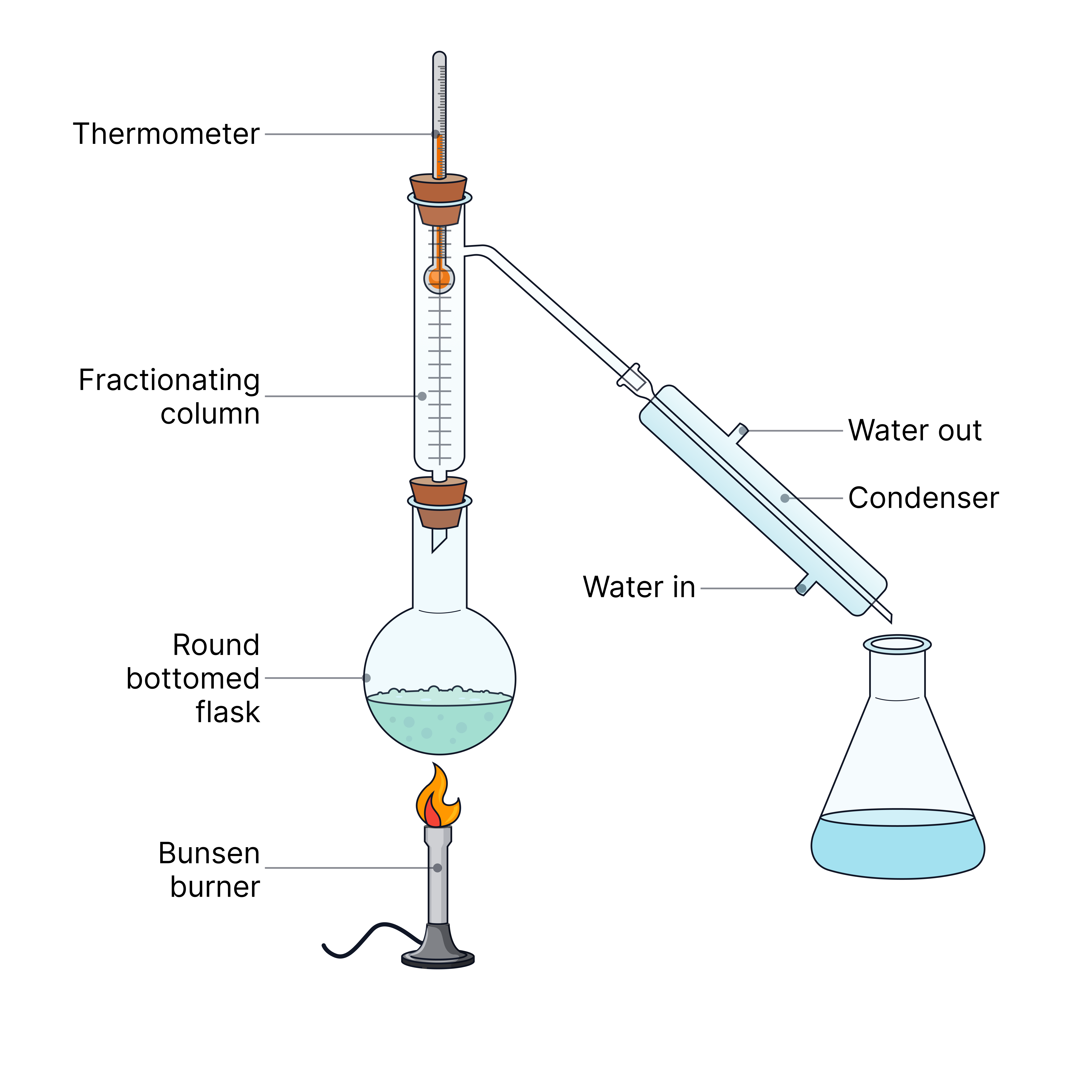
Fractional distillation | Two or more liquids with different boiling points | The mixture is heated; liquids boil and condense at different temperatures
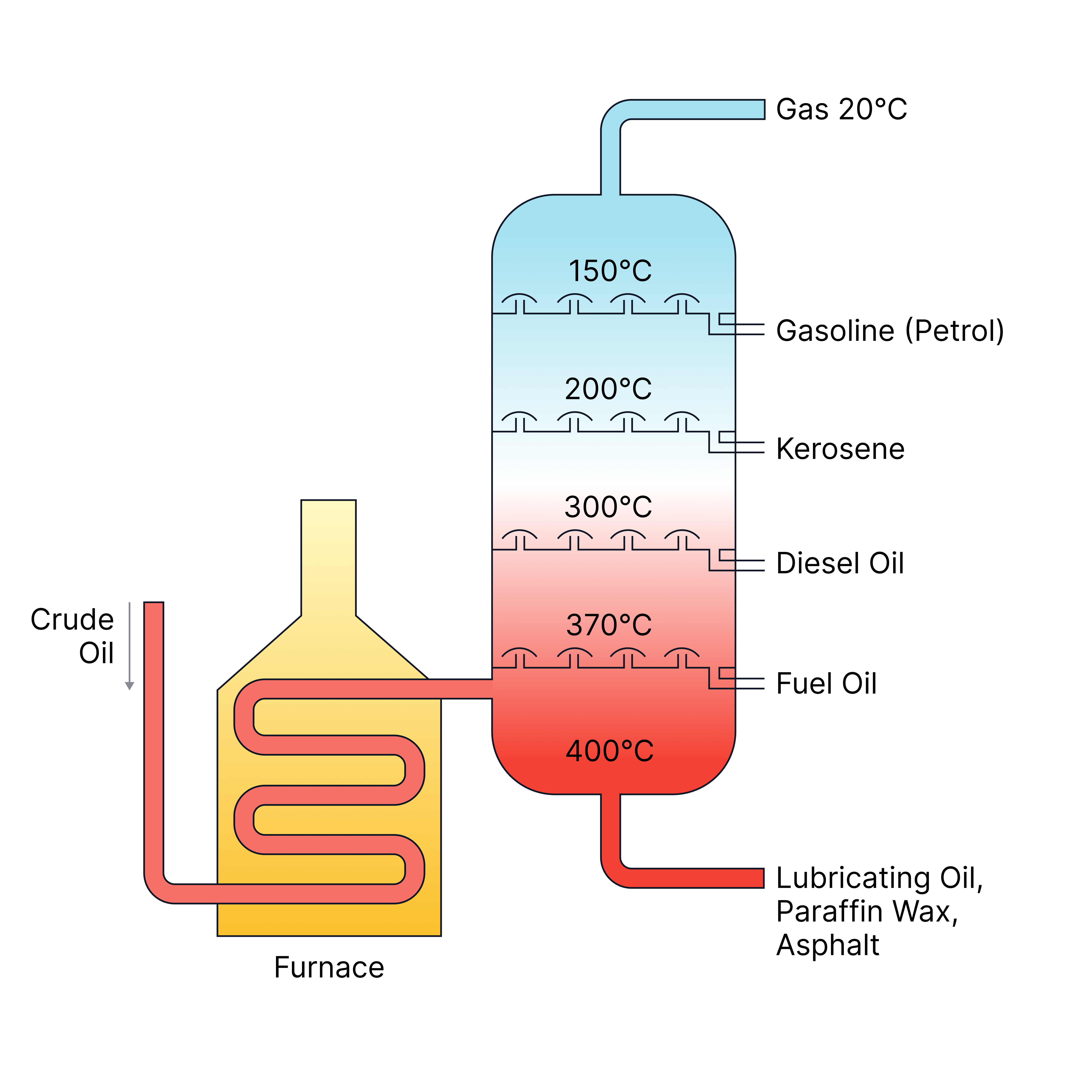
| Ethanol from water, or crude oil fractions.
|
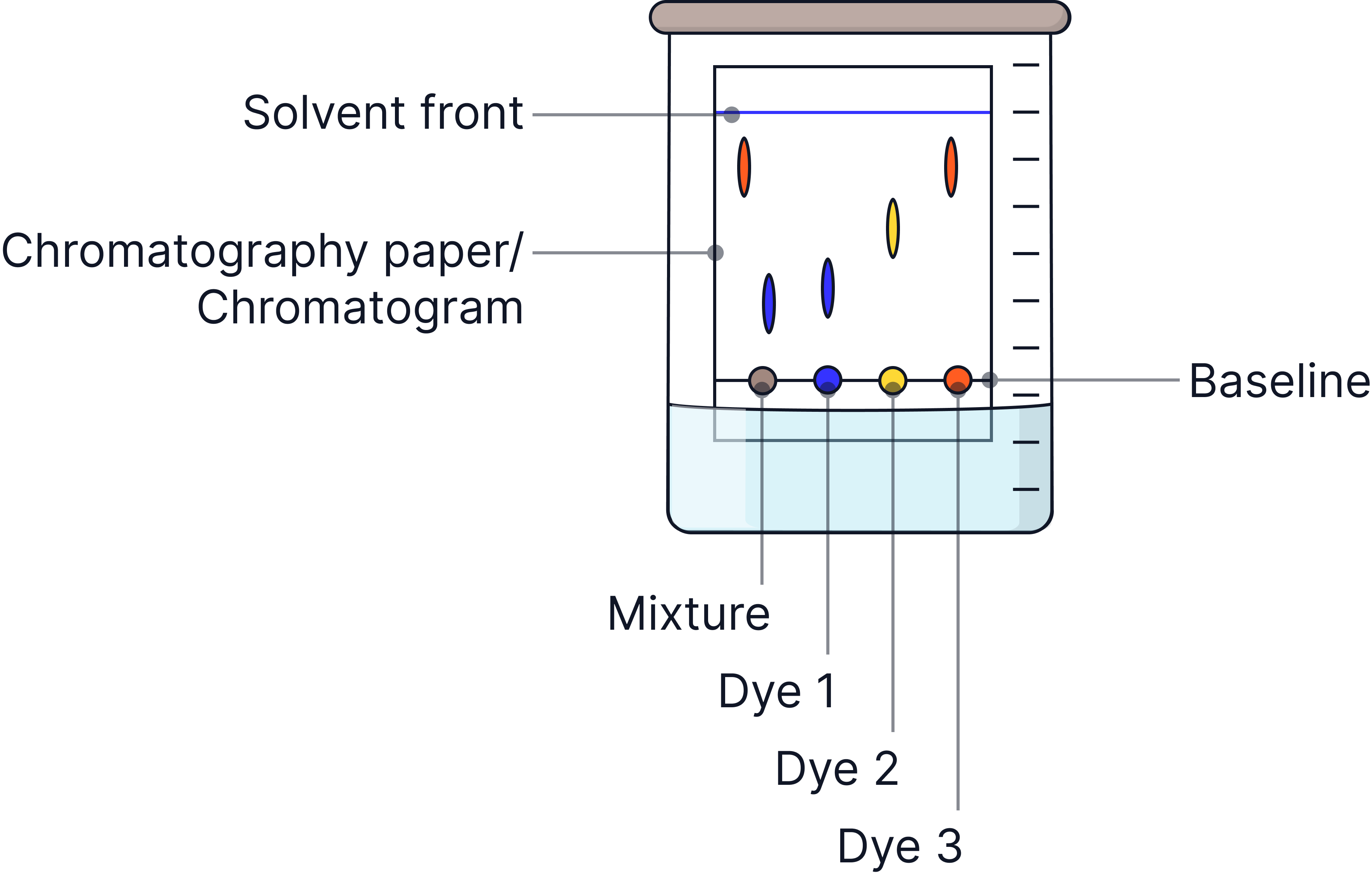
Chromatography | Substances dissolved in a solvent (e.g. dyes, inks) | Different substances move at different speeds through the stationary phase.
| Colours in ink, food dyes |
Chromatography is covered in detail in Topic 8
When selecting a method, consider:
Whether the substances are solids, liquids, or gases.
Whether they are soluble or insoluble.
Whether their boiling points or solubilities are different.
Check for Understanding
Recall
a) What is a mixture?
Answer:
a) A mixture is two or more elements or compounds that are not chemically combined together.
b) Do substances in a mixture chemically react with each other?
Answer:
b) No. The substances are not chemically bonded.
c) Name one example of a mixture found in everyday life.
Answer:
c) Acceptable answers include air, seawater, or alloys such as brass.
d) Name one physical method used to separate mixtures.
Answer:
d) Acceptable answers include filtration, crystallisation, distillation, or chromatography.
Application
e) Explain why mixtures can be separated using physical processes.
Answer:
e) Mixtures can be separated because the substances are not chemically bonded and have different physical properties such as boiling point or solubility.
f) Which separation technique would you use to obtain pure water from seawater, and why?
Answer:
f) Use simple distillation, because water has a lower boiling point than the dissolved salts and can be evaporated and condensed separately.
g) Which separation technique would you use to obtain ethanol from a mixture of ethanol and water, and why?
Answer:
g) Fractional distillation as these will be liquids with similar, but different boiling points.
Challenge
h) Describe how you could separate a mixture of sand and salt.
Answer:
h) Add water to dissolve the salt. Filter the mixture so the sand remains as residue. Evaporate the filtrate so the salt crystallises.
Summary
A mixture contains substances not chemically joined together.
The properties of each substance stay the same.
Mixtures can be separated using physical methods like filtration, crystallisation, distillation, and chromatography.
Always choose a separation method based on physical differences such as boiling point, particle size, or solubility.