Development of the model of the atom
Emmanuel Opoku
Teacher

Contents
Changing Scientific Models
Scientific ideas change over time as new evidence is discovered.
Each new experiment gives scientists a better understanding of the atom’s structure.
The atomic model evolved through several stages — from indivisible spheres, to the plum pudding model, to the nuclear model, and finally the modern atomic model.
Model | Scientist | Year | Description |
Solid sphere
| Dalton | 1803 | Atoms were thought to be tiny, solid spheres that could not be divided or broken down. |
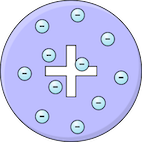
Plum pudding
| Thomson | 1904 | The atom was thought to be a ball of positive charge with negative electrons embedded within it — like plums in a pudding. This model suggested that positive and negative charges were spread evenly throughout the atom. |
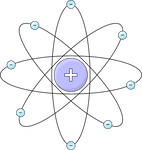
Nuclear
| Rutherford | 1911 | The atom has a small, dense, positively charged nucleus at its centre. Electrons move around the nucleus in the surrounding empty space. |
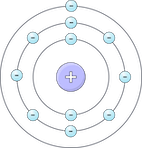
Planetary
| Bohr | 1913 | Electrons orbit the nucleus at specific distances or energy levels. |
The Alpha Particle Scattering Experiment
Evidence to support the nuclear model was gathered in 1909 by Ernest Rutherford, assisted by Hans Geiger and Ernest Marsden, with the alpha particle scattering experiment.
They fired alpha particles (positively charged) at thin gold foil and recorded how they were deflected.
If the plum pudding model was correct, all the alpha particles would pass straight through as the positive charge of the atom would be too spread out to repel them.
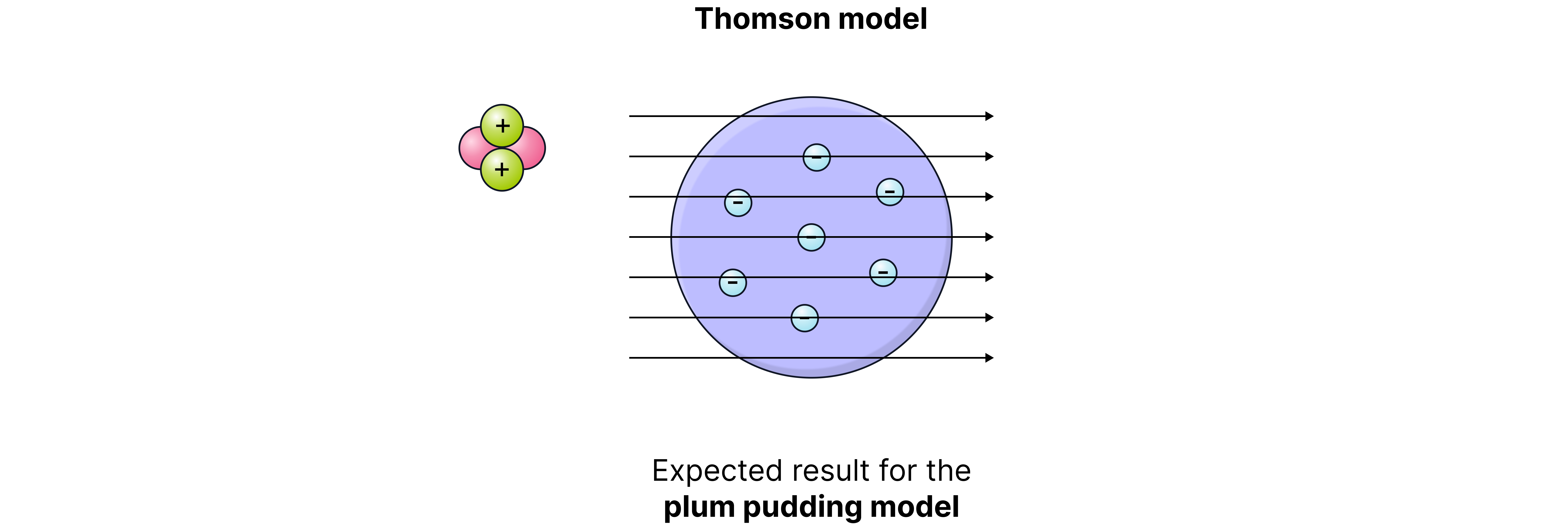
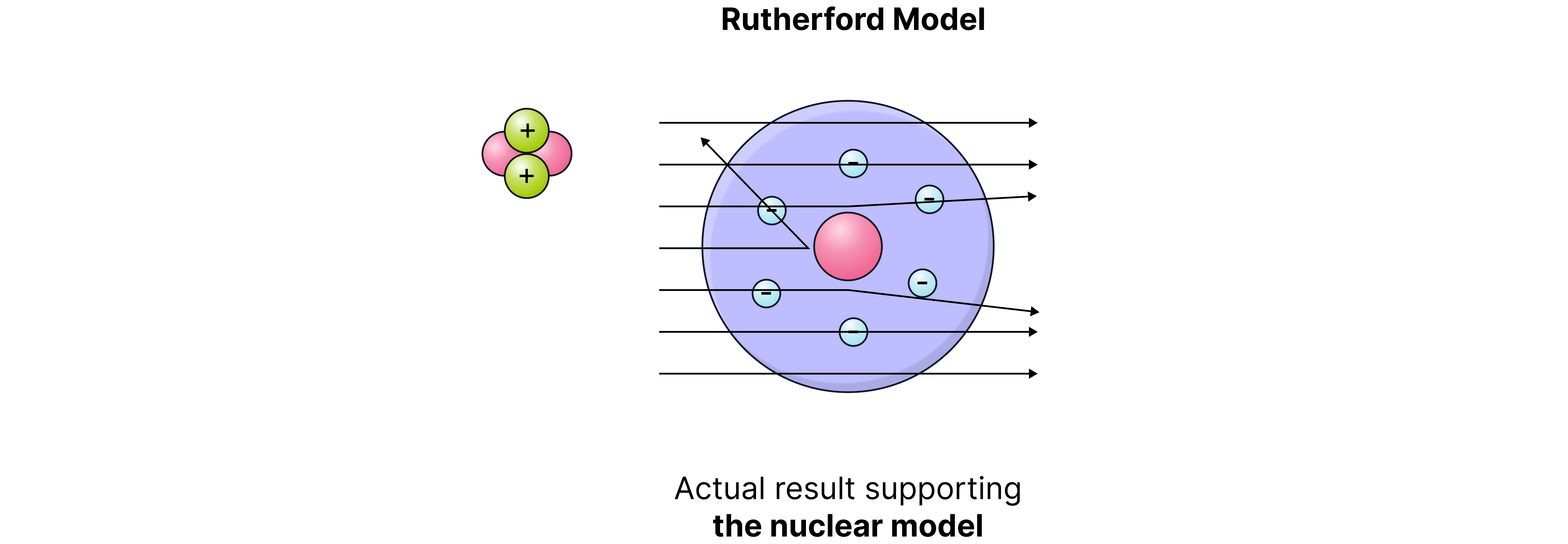
However, the evidence below was collected:
Observation (Evidence) | What is showed |
Most alpha particles passed straight through the foil. | Atoms are mostly empty space. |
Some alpha particles were deflected slightly. | The positive charge is concentrated in a small central area. |
A few particles were deflected backwards. | Most of the mass is concentrated in a tiny nucleus. |
Discovery of Protons and Neutrons
Later experiments showed that the positive charge in the nucleus was carried by smaller particles called protons.
Around 1932, James Chadwick provided experimental evidence for neutrons, neutral particles within the nucleus.
This discovery completed the modern atomic model — with protons and neutrons in the nucleus and electrons in shells around it.
Subatomic particle | Year discovered |
Electron | 1897 |
Proton | 1919 |
Neutron | 1932 |
Check for Understanding
Recall
a) What was the name of the model that described the atom as a ball of positive charge with electrons embedded in it?
Answer:
a) The plum pudding model.
b) Which scientist carried out the alpha particle scattering experiment?
Answer:
b) Ernest Rutherford.
c) In which part of the atom are protons and neutrons found?
Answer:
c) The nucleus.
Application
d) Most alpha particles passed straight through the gold foil in the scattering experiment. What did this suggest about the structure of the atom?
Answer:
d) It suggested that most of the atom is empty space.
e) Some alpha particles were deflected at large angles during the experiment. What did this suggest about the centre of the atom?
Answer:
e) It suggested there is a small, dense, positively charged nucleus at the centre of the atom.
Challenge (HT)
f) Explain how the results of the alpha particle scattering experiment caused scientists to replace the plum pudding model with the nuclear model.
Answer:
f) Most alpha particles passed straight through the foil showing atoms are mostly empty space. Some particles were deflected or bounced back showing there must be a small, dense, positively charged nucleus. This evidence contradicted the plum pudding model and led to the nuclear model.
g) State three differences between the plum pudding model and the planetary model
Answer:
g) Planetary model has a central nucleus, plum pudding does not (positive charge is spread out).
Planetary model has electrons in shells, plum pudding has electrons randomly distributed.
Planetary model shows mostly empty space, plum pudding does not.
Summary
Early models viewed the atom as indivisible or a solid sphere.
Thomson’s plum pudding model introduced electrons in a positive “pudding”.
Rutherford’s scattering experiment proved the atom has a dense, charged nucleus.
Bohr introduced electron orbits (shells), explaining atomic behaviour.
Chadwick discovered neutrons, completing the modern nuclear model.
Each stage of discovery shows how new evidence leads to changes in scientific models over time.