Atomic structure
Lajoy Tucker
Teacher

Contents
Definition
The atomic model has evolved over time as scientific discoveries have improved our understanding of atomic structure. Modern atomic theory describes atoms as consisting of a dense, positively charged nucleus surrounded by negatively charged electrons in specific energy levels.
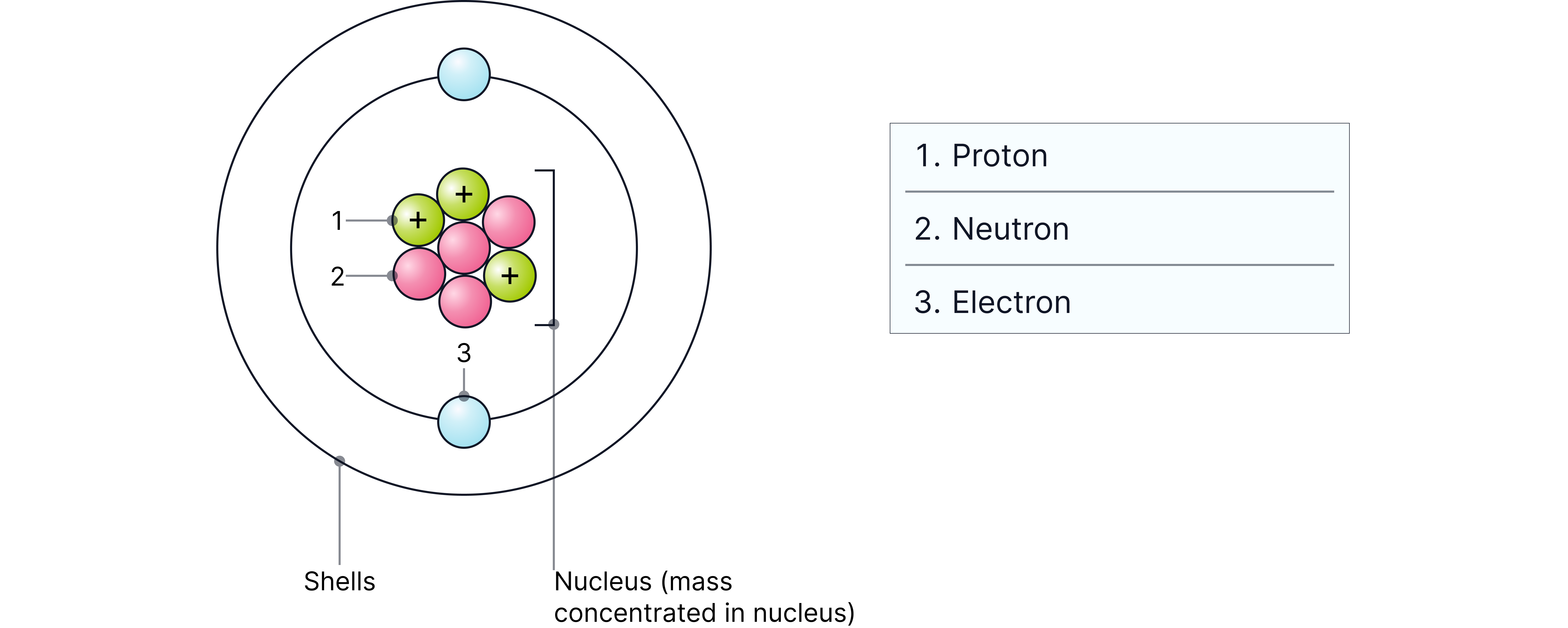
Subatomic Particles
Protons determine the atomic number and the identity of the element.
Neutrons contribute to the mass of the atom but do not affect chemical properties.
Electrons are responsible for chemical bonding and reactivity.
Particle | Relative Charge | Relative Mass | Location |
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | 1/1840 | Orbitals around the nucleus |
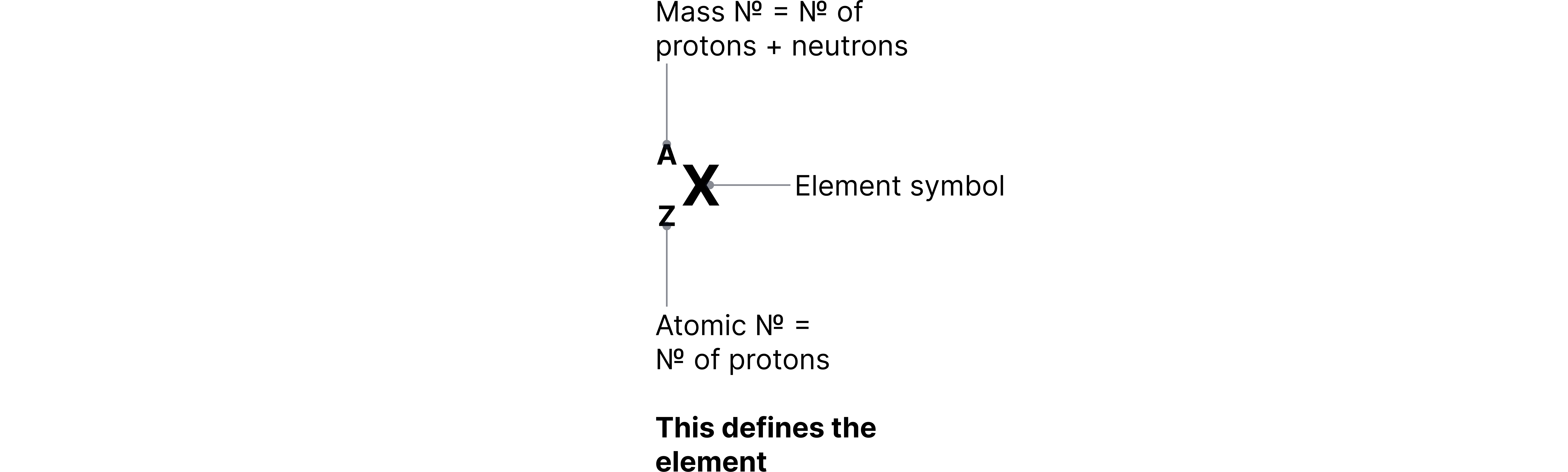
Atomic Number and Mass Number
Atomic Number (Z) = Number of protons in the nucleus.
Mass Number (A) = Total number of protons and neutrons in the nucleus.
Number of Neutrons = Mass Number – Atomic Number.
Example 1
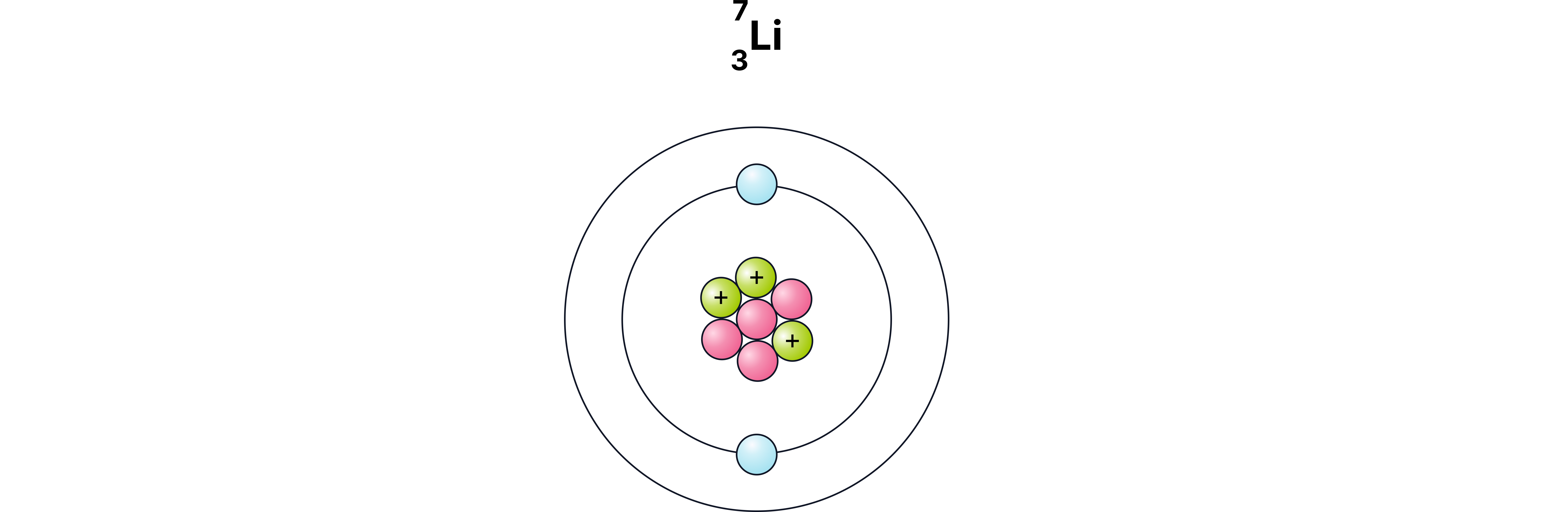
Number of protons + |
| 3 |
Number of electrons - (in a neutralatom) |
| 3 |
Number of neutrons |
| 4 |
Example 2
Determine the number of protons, neutrons, and electrons in
Number of protons = Atomic number = 11
Number of neutrons = Mass number - Atomic number = 23 - 11 = 12
Number of electrons = Number of protons (in a neutral) atom = 11
Understanding the size of an Atom
Atoms are extremely small - their size is around 0.1 nonemetres (nm) in diameter.
1 nanometer =
Therefore, the diameter of an atom ~
Comparing Sizes
Object | Approximate size (metres) |
Football | 0.2 m |
Human hair | 1 × 10⁻⁴ m |
Red blood cell | 7 × 10⁻⁶ m |
Bacterium | 1 × 10⁻⁶ m |
Virus | 1 × 10⁻⁸ m |
Atom | 1 × 10⁻¹⁰ m |
Nucleus of the atom | 1 × 10⁻¹⁴ m |
The nucleus is about 10,000 times smaller than the atom.
Scale models of atoms
To imagine something so small, scientists use scale models.
If an atom were enlarged so you could see it:
A hydrogen atom (0.1 nm) scaled up by 10¹⁰ times would be 1 metre across.
The nucleus would then be about the size of a small marble inside a football stadium.

Check for Understanding
Recall
a) What are the two subatomic particles found in the nucleus?
Answer:
a) Protons; neutrons
b) Which subatomic particle determines the identity of an element?
Answer:
b) Proton
c) What is the atomic number?
Answer:
c) Number of protons in the nucleus.
d) What is the mass number?
Answer:
d) Total number of protons and neutrons in the nucleus.
Application
e) An atom has a mass number of 27 and an atomic number of 13. Calculate the number of neutrons.
Answer:
e) Neutrons = mass number − atomic number
27 − 13 = 14
f) Explain why neutrons do not affect the chemical properties of an atom.
Answer:
f) Neutrons have no charge; chemical reactions involve electrons; therefore neutrons do not affect chemical properties
g) An atom has a diameter of (1 × 10-10) m. A model of the atom is 0.1 m wide. Calculate the scale factor of the model.
Answer:
g) Scale factor = model size ÷ actual size
0.1 ÷ (1 × 10-10) = (1 × 109 )
Challenge (HT)
h) The nucleus of an atom is approximately (1 × 10-14) m across. Using the scale factor from question g, calculate how large the nucleus would appear in the model.
Answer:
h) Model size = actual size × scale factor
((1 × 10-14 ) × (1 × 109) = 1 × 105 m
= 0.01 mm
i) An electron’s mass is said to be negligible. What does this mean in relation to relative atomic mass?
Answer:
i) The mass of an electron is very small compared to protons and neutrons.
Therefore, electrons do not significantly contribute to the relative atomic mass.
Relative atomic mass is calculated using only the masses of protons and neutrons.
j) Explain why an atom is neutral overall.
Answer:
j) An atom contains equal numbers of protons and electrons.
Protons have a positive charge (1+) and electrons have a negative charge (1-).
These charges cancel each other out, resulting in no overall charge.