Isotopes and Relative atomic mass
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Mass Number (A): The total number of protons and neutrons in the nucleus of an atom.
Atomic Number (Z): The number of protons in an atom; it defines the element.
Neutrons: Neutral subatomic particles in the nucleus. The number of neutrons can vary between atoms of the same element, forming isotopes.
Determining the Number of Subatomic Particles
For an atom or ion, the number of subatomic particles can be found using:
Protons = Atomic Number (Z)
Neutrons = Mass Number (A) - Atomic Number (Z)
Electrons (for neutral atoms) = Protons
Electrons in Ions = Protons – Charge
Example: Determine the number of protons, neutrons, and electrons in the atom below

Number of protons = 5
Number of neutrons = 11 – 5 = 6
Number of electrons = number of protons = 5
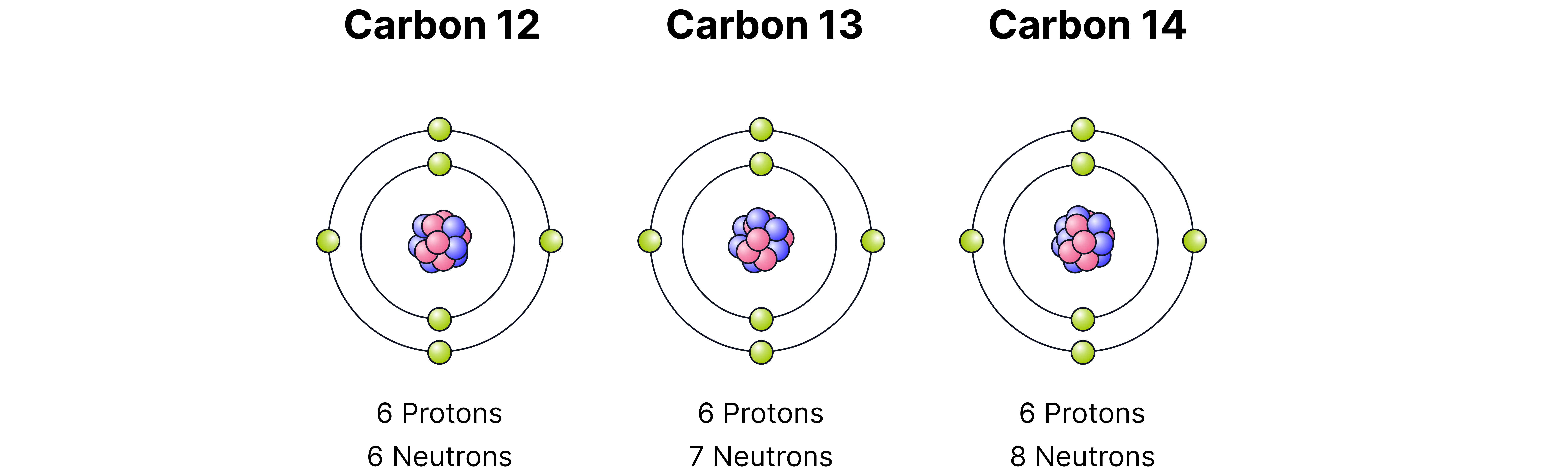
Isotope
Atoms of the same element with the same number of protons but different numbers of neutrons. This means they will have the same atomic number () but a different mass number ().
Example: Carbon-12 (), carbon-13, and carbon-14 () are isotopes of carbon.
Relative Atomic Mass
Isotopes exist in different proportions. For example, approximately 99% of all carbon atoms exist as carbon-12.
The average mass of all isotopes, accounting for their relative abundances, is the relative atomic mass found in the periodic table.
Definition:
The weighted mean mass of an atom of an element, compared to 1/12th the mass of a carbon-12 atom
1/12th the mass of an atom of carbon-12 is defined as 1 and so used as the reference for other atomic masses.
Calculating Relative Atomic Mass ()
= ∑(isotopic mass × relative abundance)/∑(total abundance)
Worked Example 1: Relative Atomic Mass of Chlorine
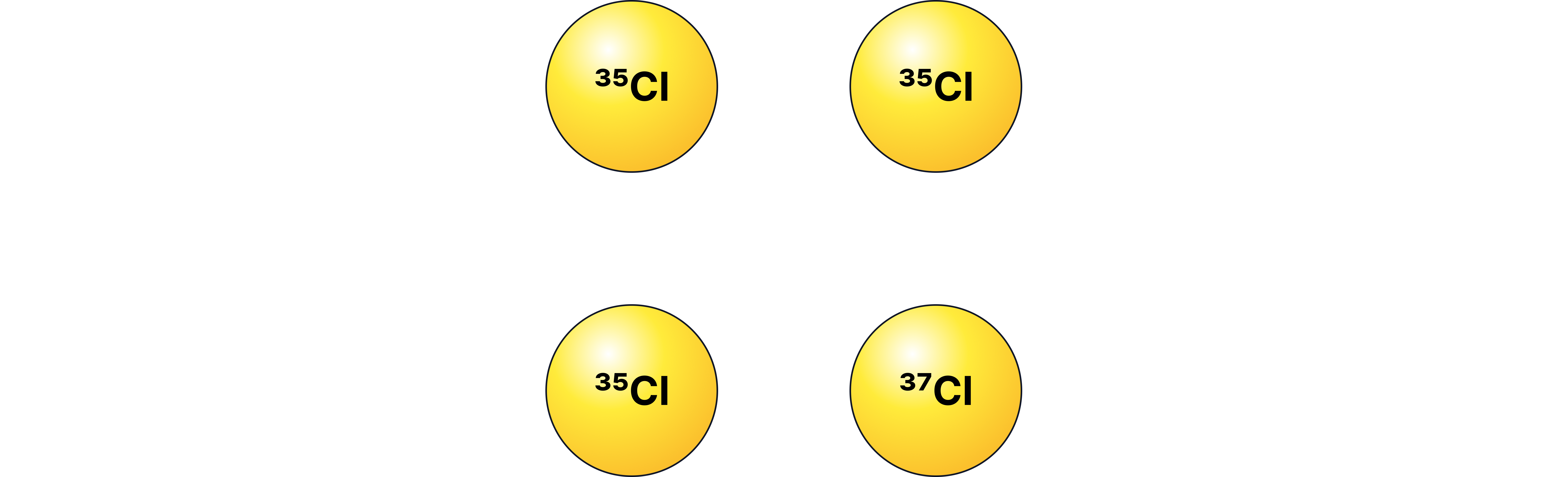
Chlorine has two isotopes: (75%) and (25%).
Calculate the relative atomic mass ().
=(35×75)+(37×25)/100 = 35.5
Thus, the periodic table gives chlorine an atomic mass of 35.5 due to its isotopic composition.
Worked Example 2: Relative Atomic Mass of Magnesium
Magnesium has three isotopes:
(78.6%)
(10.1%)
(11.3%)
Calculate the relative atomic mass ().
Answer:
= (24×78.6)+(25×10.1)+(26×11.3)/100 = 24.3
Check for Understanding
Recall
a) What is an isotope?
Answer:
a) Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
b) What does the mass number represent?
Answer:
b) The mass number is the total number of protons and neutrons in the nucleus.
c) What does relative atomic mass mean?
Answer:
c) The weighted mean mass of an atom of an element compared with 1/12 of the mass of a carbon-12 atom.
Application
d) Explain why carbon-12 and carbon-14 are isotopes of the same element.
Answer:
d) They both have the same number of protons but different numbers of neutrons.
e) Chlorine has two isotopes, Cl-35 (75%) and Cl-37 (25%). Calculate the relative atomic mass of chlorine.
Answer:
e) Ar = (35 × 75 + 37 × 25) ÷ 100 = 35.5.
Challenge (HT)
f) Lithium has two isotopes, Li-6 (7.5%) and Li-7 (92.5%). Calculate the relative atomic mass of lithium to two decimal places.
Answer:
f) Ar = (6 × 7.5 + 7 × 92.5) ÷ 100 = 6.93.
Exam tip: Make sure that if asked about subatomic particles you include the number of protons and neutrons and do NOT just refer to atomic number and mass number.
Summary
Mass number (A) = Protons + Neutrons.
Isotopes have the same number of protons but different neutrons.
Relative atomic mass (Ar) is calculated using isotopic masses & abundances.