Electronic structure
Lajoy Tucker
Teacher

Contents
Introduction
The electronic structure of an atom shows how its electrons are arranged in energy levels (also called shells) around the nucleus.
Electrons fill the lowest available energy levels first – this means the shells closest to the nucleus are filled before the outer ones.
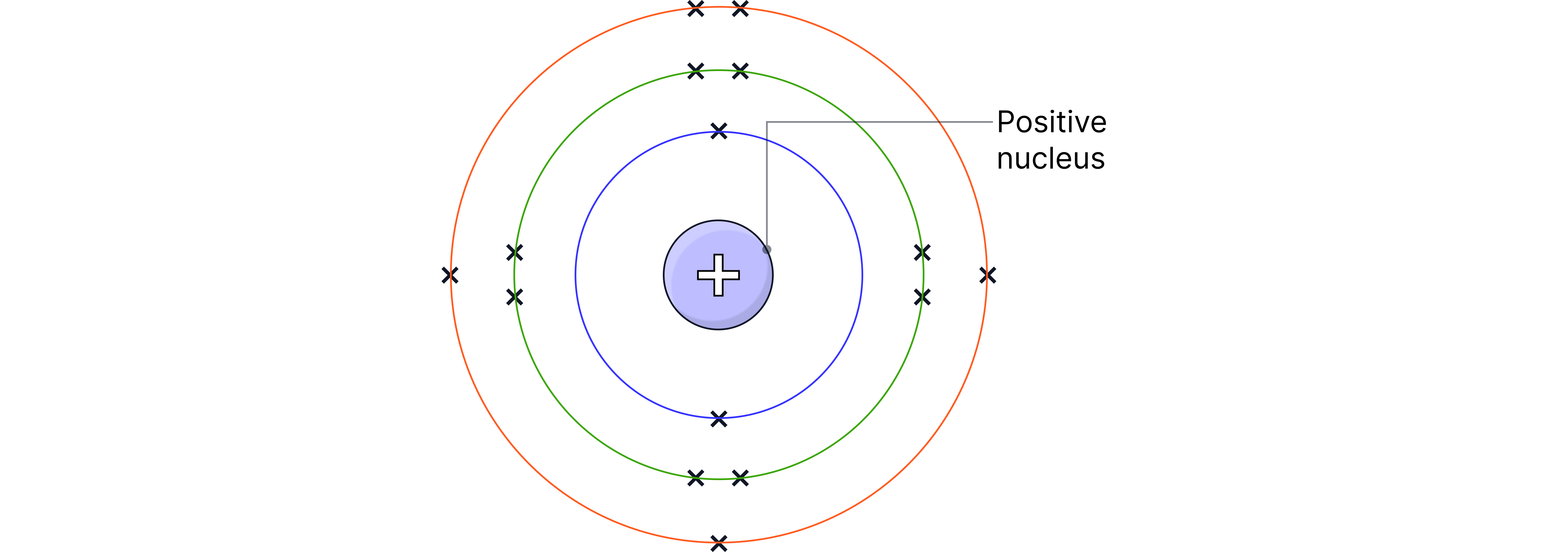
Energy Levels (Shells)
Each shell can hold a limited number of electrons:
1st shell: up to 2 electrons
2nd shell: up to 8 electrons
3rd shell: up to 8 electrons (for the first 20 elements)
When one shell is full, the next electron goes into the next shell.
Representing Electronic Structure
The electronic structure of an atom can be shown in two ways:
a) As numbers (numerical form)
This shows how many electrons are in each shell.
b) As a diagram (shell diagram)
Electrons are shown as dots or crosses on concentric circles (shells) around the nucleus.
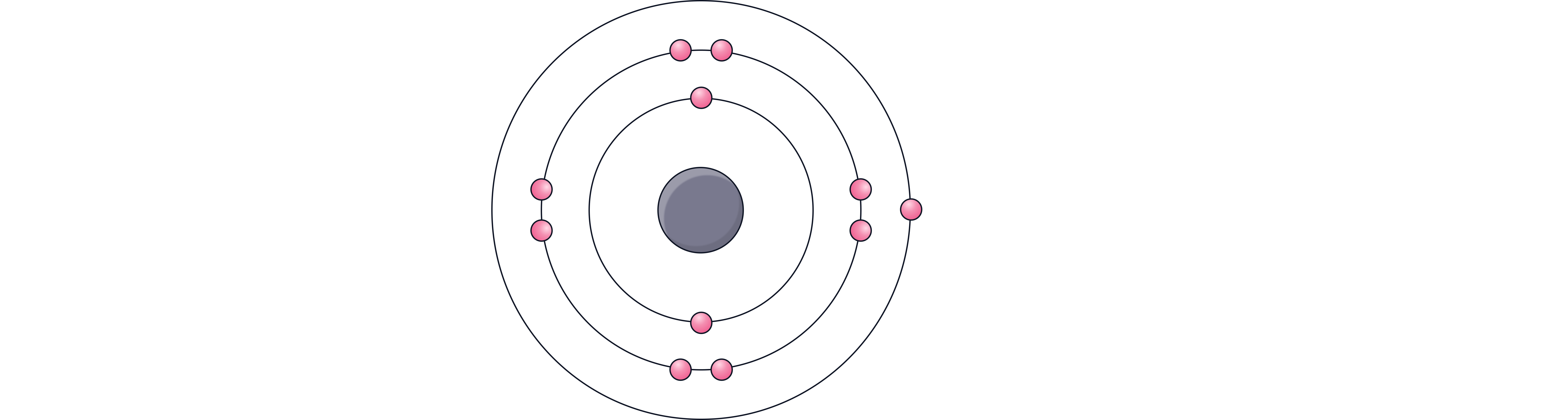
Example 1 – Sodium (atomic number 11):
11 electrons in total
1st shell – 2 electrons
2nd shell – 8 electrons
3rd shell – 1 remaining electron
Arrangement: 2,8,1
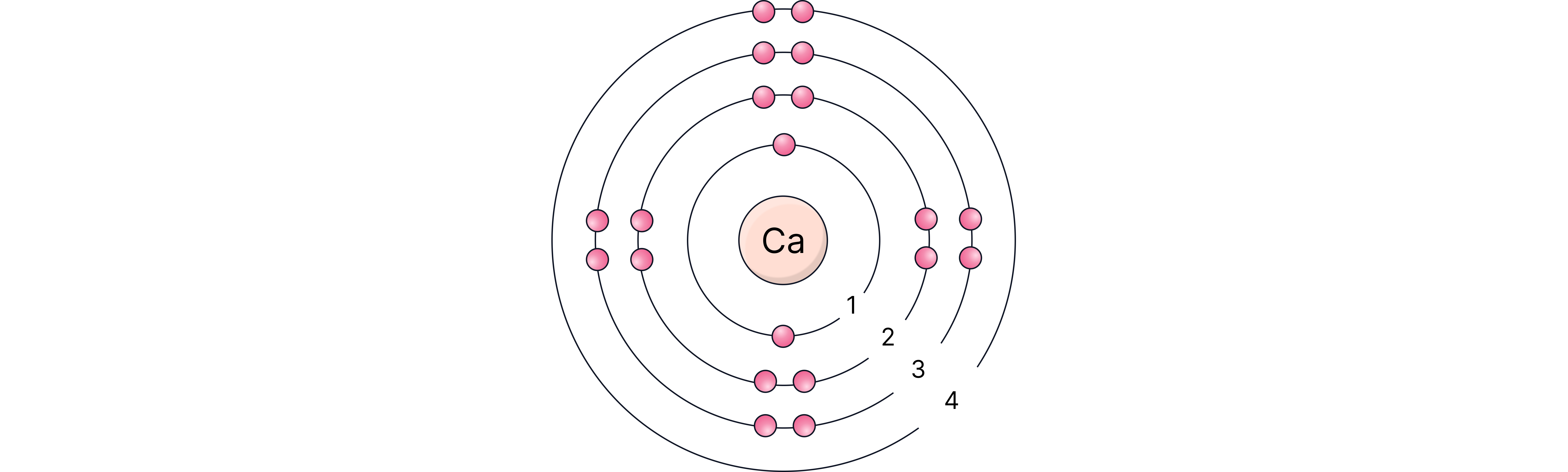
Example 2 – Calcium (atomic number 20):
20 electrons in total
1st shell – 2 electrons
2nd shell – 8 electrons
3rd shell – 8 electrons
4th shell – 2 remaining electrons
Arrangement: 2,8,8,2
Periodic Table
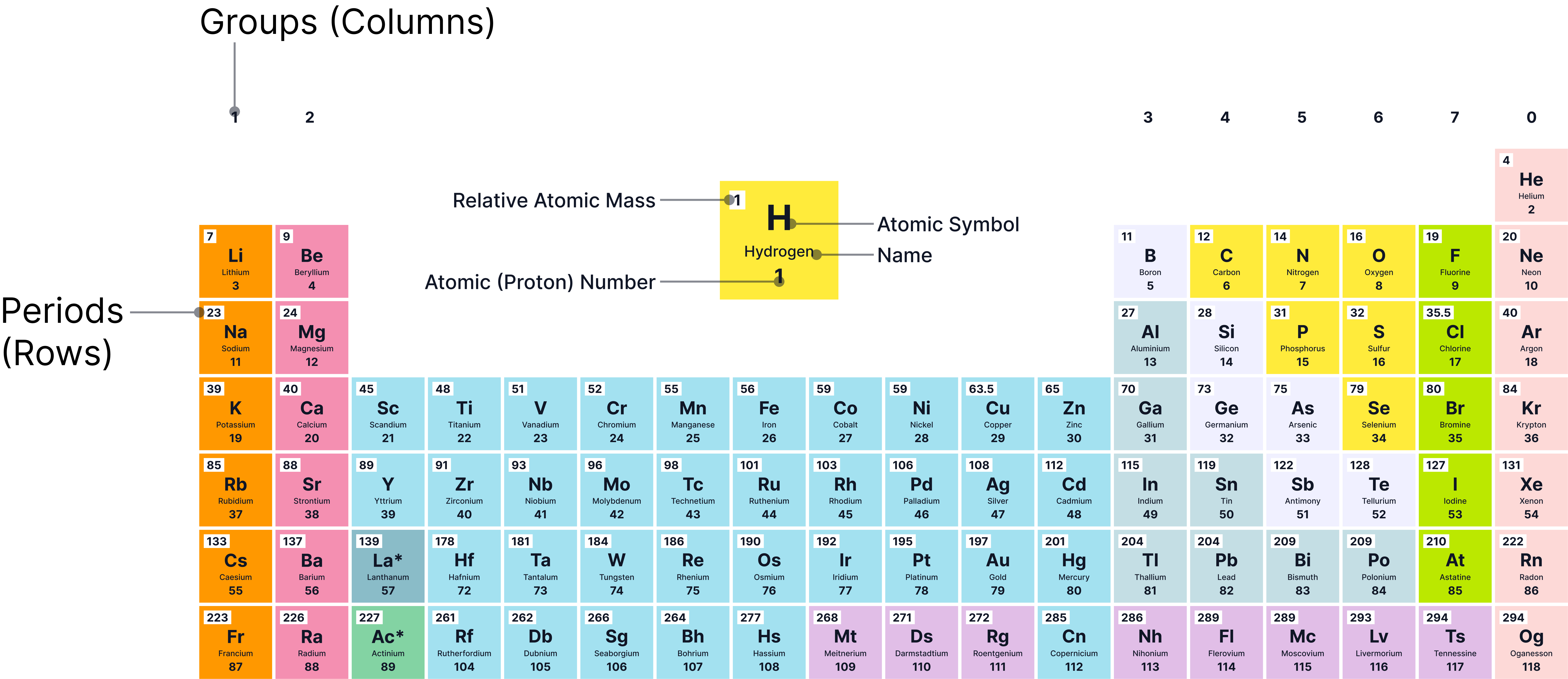
The period number shows the number of shells an atom has.
The group number shows the number of electrons in the outer shell (for Groups 1–7).
Examples:
Lithium (Li) → 2,1 → Group 1, Period 2
Oxygen (O) → 2,6 → Group 6, Period 2
Sodium (Na) → 2,8,1 → Group 1, Period 3
Why It Matters
The number of outer electrons determines how an element reacts.
Atoms with full outer shells (like noble gases) are stable and unreactive.
Atoms with 1 or 2 outer electrons (like metals) tend to lose them.
Atoms with 6 or 7 outer electrons (like non-metals) tend to gain or share electrons.
Check for Understanding
Recall
a) What does the electronic structure of an atom show?
Answer:
a) The electronic structure shows how electrons are arranged in shells (energy levels) around the nucleus.
b) How many electrons can the first electron shell hold?
Answer:
b) 2 electrons.
c) What does the group number tell you about an atom (for Groups 1 - 7)?
Answer:
c) The number of electrons in the outer shell.
Application
d) Write the electronic structure for oxygen (atomic number 8).
Answer:
d) 2,6.
e) An element has the electronic structure 2,8,1. Identify its group and period in the periodic table.
Answer:
e) Group 1 (1 electron in the outer shell) and Period 3 (3 occupied shells).
Challenge (HT)
f) Explain why elements in the same group of the periodic table have similar chemical properties.
Answer:
f) Elements in the same group have the same number of electrons in their outer shell, so they react in similar ways to achieve a full outer shell.
Summary
Electrons occupy the lowest available energy levels first.
Electronic structure can be shown as numbers (e.g. 2,8,1) or a diagram.
The group number = outer shell electrons, and period number = number of shells.
The arrangement of electrons explains patterns in reactivity across the periodic table.