Bond energies (HT only)
Emmanuel Opoku
Teacher

Contents
Energy in Chemical Bonds
Key idea:
Energy is taken in to break bonds (endothermic).
Energy is given out when new bonds form (exothermic).
The overall energy change shows whether a reaction is exothermic or endothermic.
Conservation of Energy
Energy cannot be created or destroyed, it is only transferred.
Process | Flow of Energy | Type of Change |
Breaking bonds | Taken in | Endothermic |
Making bonds | Given out | Exothermic |
Worked Examples
Example 1 - Forming Water
Question:
Calculate the energy change for the reaction below.
Displayed formula:

Bond Energies:
Bond | Bond Energy (kJ/mol) |
H–H | 436 |
O=O | 498 |
O–H | 464 |
STEP 1: Calculate total energy needed to break bonds and total energy released when making bonds
To do this, you need to count all the bonds in the reactants (bonds broken) and all the bonds in the products (bonds made). This is where the displayed formula comes in handy.
Remember to take the coefficients (stoichiometry) into account – a table is useful here.
BONDS BROKEN
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
H–H | 436 | 2 | (436 x 2) = 872 |
O=O | 498 | 1 | (498 x 1) = 498 |
Total Energy In | 1370 |
BONDS MADE
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
O–H | 464 | 4 | (464 x 4) = 1856 |
Total Energy Out | 1856 |
STEP 2: Calculate the overall energy change (ΔH)
This is an example of an exothermic reaction where energy is released to surroundings (indicated by a negative overall energy change)
Example 2 - Reaction of Hydrogen and Bromine
Question:
Calculate the overall energy change (ΔH) for the reaction between hydrogen and bromine:
Displayed formula:

Bond Energies:
Bond | Bond Energy (kJ/mol) |
H–H | 436 |
Br–Br | 193 |
H-Br | 366 |
STEP 1: Calculate total energy needed to break bonds and total energy released when making bonds
BONDS BROKEN
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
H–H | 436 | 1 | (436 x 1) = 436 |
Br-Br | 193 | 1 | (193 x 1) = 193 |
Total Energy In | 629 |
BONDS MADE
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
H–Br | 366 | 2 | (366 x 2 ) = 732 |
Total Energy Out | 732 |
STEP 2: Calculate the overall energy change (ΔH)
This is another example of an exothermic reaction where energy is released to surroundings (indicated by a negative overall energy change)
Example 3 - Reaction of Ethene and Bromine
Question:
Calculate the overall energy change (ΔH) for the reaction below.
Displayed formula:
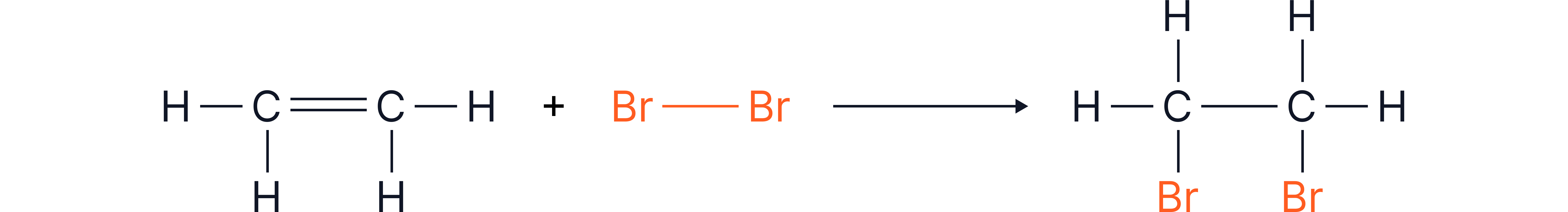
Bond Energies:
Bond | Bond Energy (kJ/mol) |
C=C | 614 |
Br–Br | 193 |
C–C | 347 |
C–Br | 276 |
STEP 1: Calculate total energy needed to break bonds and total energy released when making bonds
BONDS BROKEN
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
C=C | 614 | 1 | (614 x 1) = 614 |
Br–Br | 193 | 1 | (193 x 1) = 193 |
Total Energy In |
|
| 807 |
BONDS MADE
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
C=C | 347 | 1 | (347 x 1) = 347 |
Br–Br | 276 | 2 | (276 x 2) = 552 |
Total Energy Out |
|
| 899 |
STEP 2: Calculate the overall energy change (ΔH)
This reaction is also exothermic reaction where energy is released to surroundings (indicated by a negative overall energy change)
Example 4 - Combustion of Methane
Question:
Calculate ΔH for the combustion of methane:
Displayed formula:
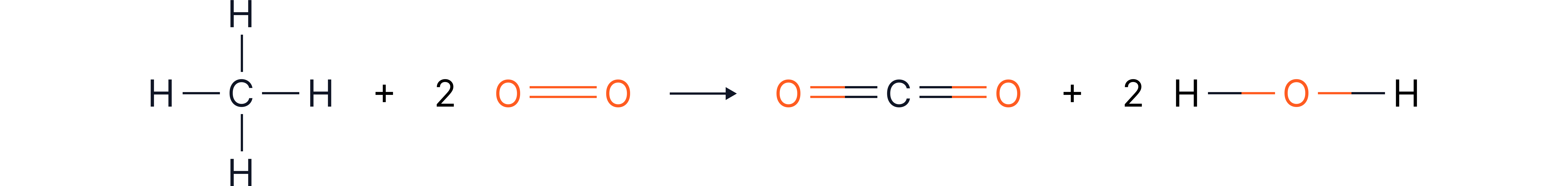
Bond Energies Provided:
Bond | Bond Energy (kJ/mol) |
C–H | 413 |
O=O | 498 |
C=O | 805 |
O–H | 464 |
STEP 1: Calculate total energy needed to break bonds and total energy released when making bonds
BONDS BROKEN
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
C–H | 413 | 4 | (413 x 4) = 1652 |
O=O | 498 | 2 | (498 x 2) = 996 |
Total Energy In |
|
| 2648 |
BONDS MADE
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
C–O | 805 | 2 | (805 x 2) = 1610 |
O=H | 464 | 4 | (464 x 4) = 1856 |
Total Energy Out |
|
| 3466 |
STEP 2: Calculate the overall energy change (ΔH)
This reaction is strongly exothermic, indicated by a large, negative value for the overall energy change (ΔH) - usually the case for combustion reactions
Challenging Example
Finding an Unknown Bond Energy
Question:
Find the N–N bond energy in this reaction, given
Displayed formula:
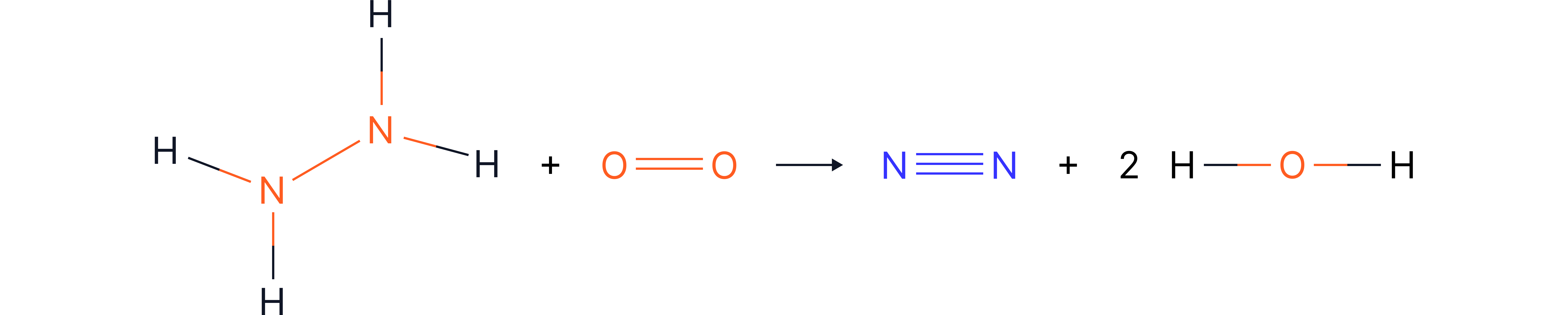
Bond Energies:
Bond | Bond Energy (kJ/mol) |
N–H | 388 |
O=O | 498 |
N=N | 944 |
O–H | 463 |
N-N | x |
STEP 1: Calculate total energy needed to break bonds and total energy released when making bonds
BONDS BROKEN
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
N–N | x | 1 | (1x) = x |
N–H | 388 | 4 | (388 x 4) = 1552 |
O=O | 498 | 1 | (498 x 1) = 498 |
Total Energy In | x + 2050 |
BONDS MADE
Bond | Bond Energy (kJ/mol) | No. of Bonds | Energy (kJ/mol) |
N–N | 944 | 1 | (944 x 1) = 944 |
O–H | 463 | 4 | (563 x 4) = 1852 |
Total Energy Out | 2796 |
STEP 2: Calculate missing bond by substituting overall energy change given in question and rearranging to find x (missing bond enthalpy)
N–N bond energy = 113 kJ/mol
Interpreting overall energy change (ΔH)
Sign of overall energy change (ΔH) | Meaning | In terms of bonds | Reaction Type |
Positive (+) | Energy taken in from surroundings overall | More energy needed to break bonds than is released forming bonds | Endothermic |
Negative (–) | Energy given out to surroundings | More energy released forming bonds than is needed to break them | Exothermic |
Key Takeaways
Concept | Explanation |
Breaking bonds | Energy taken in → endothermic |
Making bonds | Energy given out out → exothermic |
Overall | = Bonds broken – Bonds made |
Exothermic Reactions | ΔH is a negative value |
Endothermic Reactions | ΔH is a positive value |
Check Your Understanding
Recall
a) What is meant by bond energy?
Answer
a) Bond energy = energy required to break 1 mole of a specified bond in gaseous molecules.
b) Why is breaking bonds endothermic?
Answer
b) Breaking bonds is endothermic because energy must be absorbed to overcome the attractive forces in the bond.
c) Write the formula for ΔH.
Answer
c) (bonds broken)(bonds made).
d) What does a positive ΔH value show?
Answer
d) Positive overall endothermic (more energy absorbed than released).
Apply
e) Calculate the overall energy change,, for:
Displayed (structural) formulas:
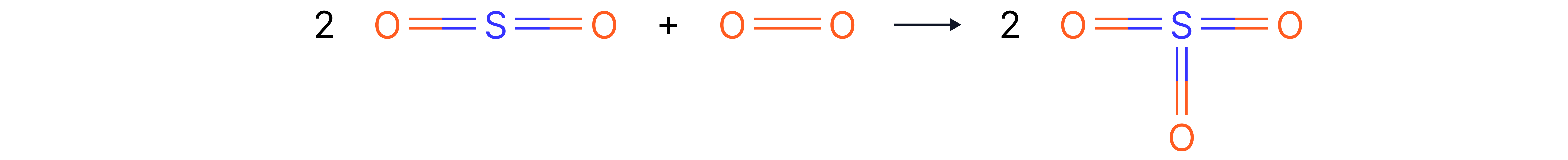
Bond energies supplied
Bond | Bond enthalpy / |
S=O | 523 |
O=O | 495 |
Answer
Count Bonds broken and made and find totals of each
Bond | No. | ||
Broken | |||
S=O (in) | 523 | 4 | |
O=O | 495 | 1 | 495 |
broken | 2587 | ||
Made | |||
S=O (in ) | 523 | 6 | |
made | 3138 |
Exothermic overall.
Challenge
f) Propane () combusts completely in oxygen as follows:
Displayed formula:
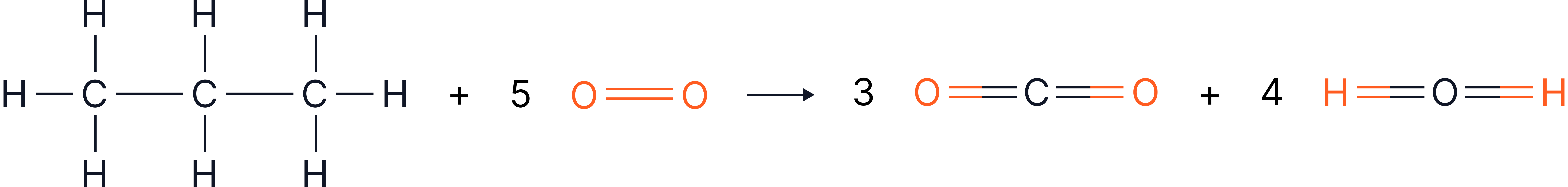
Given that the overall energy change for this reaction is , calculate the bond enthalpy for a C=O bond in carbon dioxide.
Bond Enthalpies
Bond | Bond enthalpy / |
C–H | 413 |
C–C | 348 |
O=O | 495 |
O–H | 463 |
C=O | ? (to find) |
Answer
Combustion of propane:
Count bonds and totals
Bond | No. | ||
Bonds Broken | |||
C–C | 348 | 2 | 696 |
C–H | 413 | 8 | 3304 |
O=O | 495 | 5 | 2475 |
broken | 6475 | ||
Bonds Made | |||
C=O | x | 6 | 6x |
O–H | 463 | 6 | 3704 |
made | 6x + 3704 |
Solve with brokenmade
(to 3 s.f)