Exothermic and endothermic reactions
Lajoy Tucker
Teacher

Contents
Conservation of Energy
Key idea:
Energy is conserved in all chemical reactions - it cannot be created or destroyed, only transferred.
If energy is given out to the surroundings by a chemical reaction (the system), the products have less energy than the reactants.
If energy is taken in from the surroundings by a chemical reaction (the system), the products have more energy than the reactants.
Exothermic Reactions
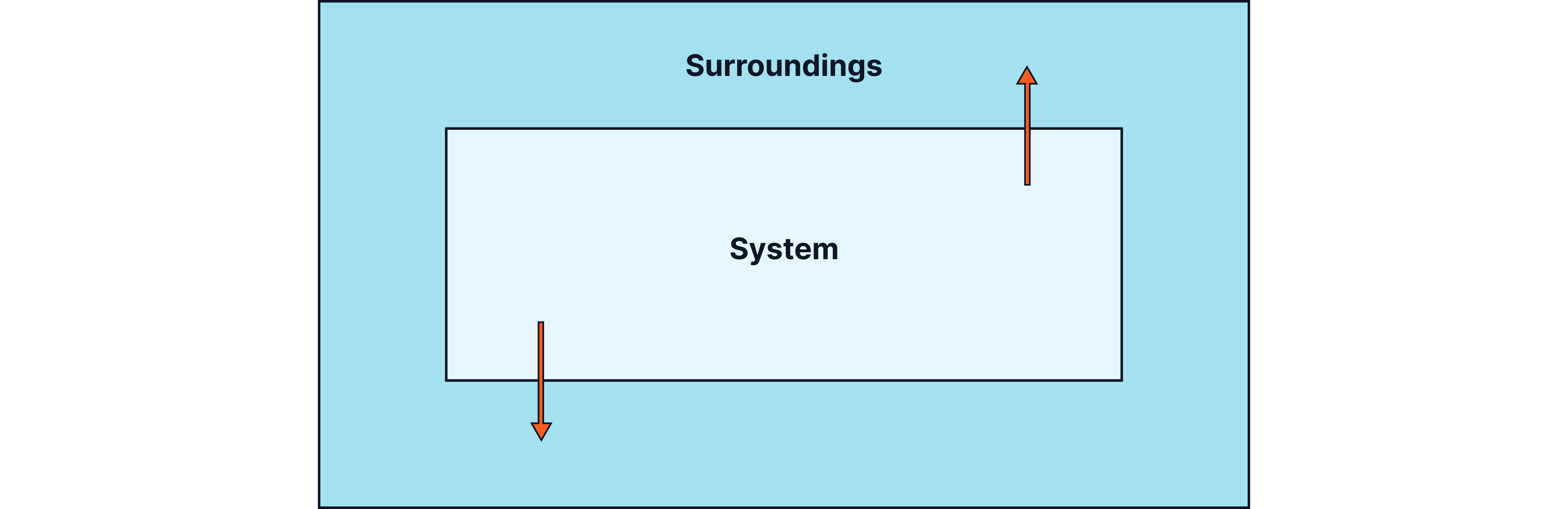
Definition:
An exothermic reaction transfers energy to the surroundings, causing the temperature of the surroundings to increase.
Examples:
Combustion (e.g. burning fuels)
Neutralisation (acid + alkali)
Everyday Uses:
Self-heating cans
Hand warmers
In Terms of Bonds (HT ONLY):
Energy released from forming new bonds is greater than the energy needed to break existing bonds.
Energy released (forming bonds) > Energy absorbed (breaking bonds)
Endothermic Reactions
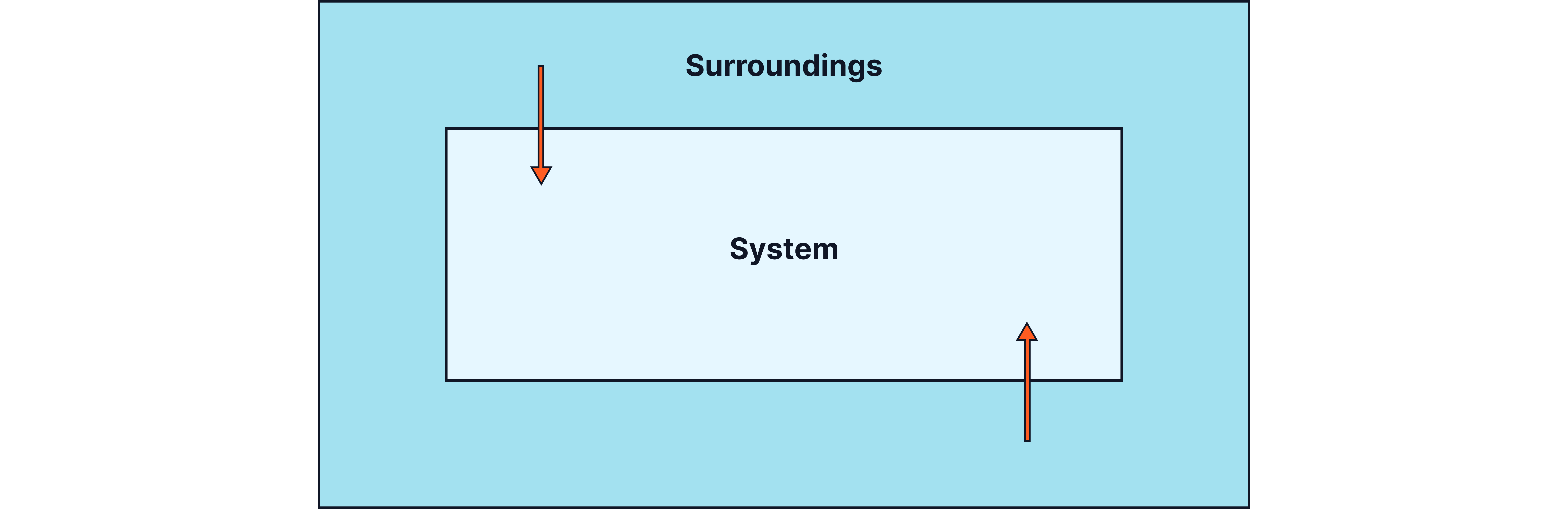
Definition:
An endothermic reaction takes in energy from the surroundings, so the temperature of the surroundings decreases.
Examples:
Thermal decomposition (e.g. calcium carbonate → calcium oxide + carbon dioxide)
Photosynthesis
Everyday Uses:
Instant cold packs (sports injury packs)
In Terms of Bonds (HT ONLY):
Energy needed to break bonds is greater than the energy released when new bonds form.
Energy absorbed (breaking bonds) > Energy released (forming bonds)
Energy Profile Diagrams
Definition:
An energy profile diagram (reaction profile diagram) shows the relative energies of the reactants and products over time (reaction progress/coordinate), the activation energy and the overall energy change .
Exothermic Reaction Profile
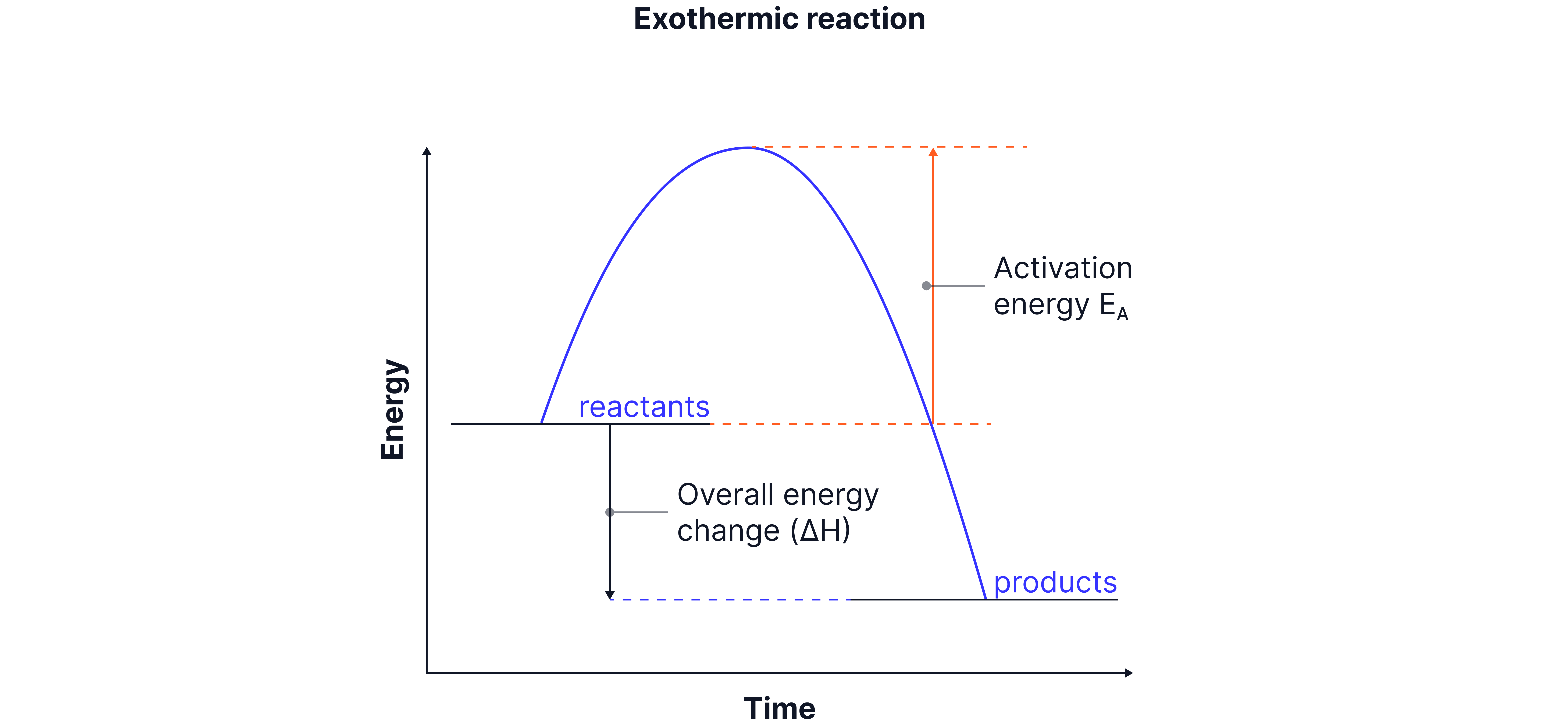
Products are lower in energy than reactants.
Energy is given out to the surroundings.
(negative overall energy change)
Overall energy change is less than zero
Diagram features:
Downward curve.
Activation energy shown from reactants to the top of curve (single-headed arrow).
Downward arrow from reactants to products shows energy given out.
Endothermic Reaction Profile
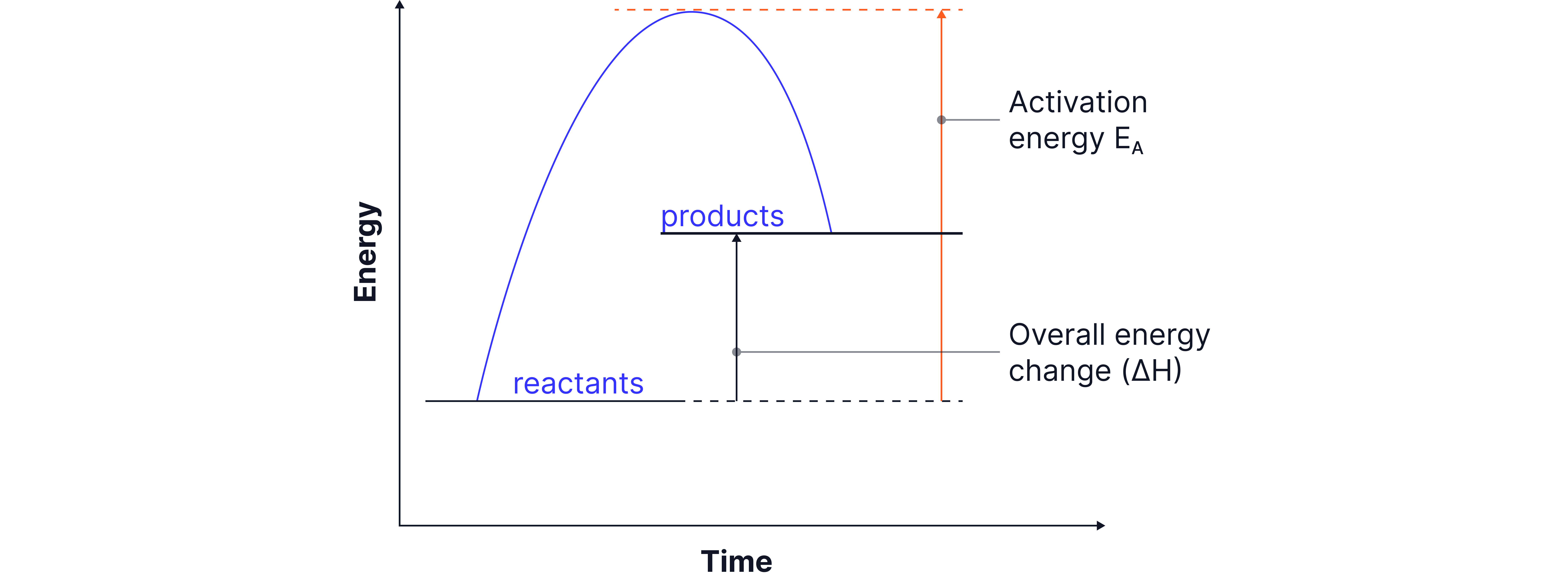
Products are higher in energy than reactants.
Energy is taken in from the surroundings.
(positive energy change)
Overall energy change is more than zero
Diagram features:
Activation energy shown from reactants to the top of curve (single-headed arrow).
Upward arrow from reactants to products shows energy taken in.
Key Terms
Term | Definition |
Activation Energy (Eₐ) | Minimum energy that reactant particles must have to react when they collide. |
Overall energy change (ΔH) | Difference between energy of reactants and products. |
Exothermic Reaction | A reaction that gives out to the surroundings (causes a temperature increase) |
Endothermic Reaction | A reaction that takes in energy from the surroundings (causes a temperature decrease) |
Comparing Exothermic and Endothermic
Feature | Exothermic | Endothermic |
Energy transfer | Gives out energy to surroundings | Takes energy in from surroundings |
Temperature of surroundings | Increases | Decreases |
Relative Energies of reactants vs products | Reactants higher in energy | Products higher in energy |
ΔH value | Negative (-) | Positive (+) |
Common Examples | Combustion, neutralisation, hand warmers | Thermal decomposition, photosynthesis, sports injury packs |
Check Your Understanding
Recall
a) What happens to temperature in an exothermic reaction?
Answer
a) It increases - energy is transferred to the surroundings.
b) Define activation energy.
Answer
b) The minimum energy particles need to collide and react.
c) Give one example of an exothermic and one of an endothermic reaction.
Answer
c) Exothermic: combustion; Endothermic: thermal decomposition.
d) In an energy profile diagram, where is activation energy shown?
Answer
d) From reactants to top of the curve.
Apply
e) Describe how the energy of the products compares with the reactants in:
i) an exothermic reaction
ii) an endothermic reaction.
Answer
e)
i) Products have less energy (energy released).
ii) Products have more energy (energy absorbed).
f) A student mixes citric acid with sodium hydrogencarbonate - the temperature falls.
What type of reaction is this?
Explain your reasoning.
g) Draw and label an energy profile diagram for an exothermic reaction.
Include: reactants, products, activation energy, and overall energy change.
Answer
f) Endothermic - temperature fell, so energy absorbed.